Task-positive Functional Connectivity of the Default Mode
Network Transcends Task Domain
Amanda Elton and Wei Gao*
Astratto
■ The default mode network (DMN) was first recognized as a
set of brain regions demonstrating consistently greater activity
during rest than during a multitude of tasks. Originally, this net-
work was believed to interfere with goal-directed behavior
based on its decreased activity during many such tasks. More
recently, Tuttavia, the role of the DMN during goal-directed
behavior was established for internally oriented tasks, in which
the DMN demonstrated increased activity. Tuttavia, the well-
documented hub position and information-bridging potential
of midline DMN regions indicate that there is more to uncover
regarding its functional contributions to goal-directed tasks,
which may be based on its functional interactions rather than
its level of activation. An investigation of task-related changes
in DMN functional connectivity during a series of both internal
and external tasks would provide the requisite investigation for
examining the role of the DMN during goal-directed task perfor-
mance. In this study, 20 participants underwent fMRI while per-
forming six tasks spanning diverse internal and external
domains in addition to a resting-state scan. We hypothesized
that the DMN would demonstrate “task-positive” (cioè., positively
contributing to task performance) changes in functional connec-
tivity relative to rest regardless of the direction of task-related
changes in activity. Infatti, our results demonstrate significant
increases in DMN connectivity with task-promoting regions
(per esempio., anterior insula, inferior frontal gyrus, middle frontal gyrus)
across all six tasks. Inoltre, canonical correlation analyses in-
dicated that the observed task-related connectivity changes were
significantly associated with individual differences in task perfor-
mance. Our results indicate that the DMN may not only support
a “default” mode but may play a greater role in both internal and
external tasks through flexible coupling with task-relevant brain
regions. ■
INTRODUCTION
The default mode network (DMN; Raichle et al., 2001)
was originally identified based on an observation by
Shulman, Fiez, Corbetta, Buckner, and Miezin (1997) Quello
a common set of brain regions demonstrated decreased
blood flow across a range of visual processing tasks. Questo
finding led the authors to posit that this set of regions
represents a “default mode of brain function.” Although
originally considered a “task-negative” network and be-
lieved to interfere with goal-directed actions, theories
of its functional roles have extended to include the sup-
port of internally based mentation processes (Kelly,
Uddin, Biswal, Castellanos, & Milham, 2008; Sonuga-
Barke & Castellanos, 2007; Weissman, Roberts, Visscher,
& Woldorff, 2006; Fox et al., 2005) and general monitor-
ing of the internal and/or external world (Gao, Gilmore,
Alcauter, & Lin, 2013; Gilbert, Dumontheil, Simons, Frith, &
Burgess, 2007; Hahn, Ross, & Stein, 2007; Gilbert, Simons,
Frith, & Burgess, 2006; Wagner, Shannon, Kahn, & Buckner,
2005). Così, the DMN may promote—rather than interfere
with—certain “task-positive” processes. Nonetheless, IL
mechanisms by which this network contributes to behavior
are not fully understood.
University of North Carolina at Chapel Hill
*Now at Cedars-Sinai Medical Center, Los Angeles, CA.
© 2015 Istituto di Tecnologia del Massachussetts
If the DMN is involved in general monitoring of the en-
vironment, the fulfillment of this function should encom-
pass multimodal information processing including
interactions with higher-order cognitive areas to make
prompt decisions in response to internal and external
stimuli. Infatti, numerous studies from both structural
and functional connectivity perspectives have documented
that midline DMN regions are among the most effi-
ciently wired brain areas, serving as global “hubs” that
bridge different functional systems across the brain
(van den Heuvel & Sporns, 2013; Buckner et al., 2009;
Hagmann et al., 2008). The behavioral relevance of such
connections has been demonstrated by several studies
showing that increased DMN connectivity with regions
of other brain networks during goal-directed task states
facilitates task performance. Per esempio, both Gao and
Lin (2012) and Spreng, Stevens, Chamberlain, Gilmore,
and Schacter (2010) have shown that the DMN increases
functional connectivity with a frontoparietal control sys-
tem during “internally directed” tasks. Tuttavia, DMN
contributions to task performance may not be limited
to internal tasks, as Gao et al. (2013) showed a similar
enhancement of DMN interactions, particularly with re-
gions of the salience network (Seeley et al., 2007), during
an “externally directed” classification task. Così, IL
current literature, composed of several independent
Journal of Cognitive Neuroscience 27:12, pag. 2369–2381
doi:10.1162/jocn_a_00859
D
o
w
N
l
o
UN
D
e
D
F
R
o
M
l
l
/
/
/
/
j
F
/
T
T
io
T
.
:
/
/
H
T
T
P
:
/
D
/
o
M
w
io
N
T
o
P
UN
R
D
C
e
.
D
S
F
io
R
o
l
M
v
e
H
R
C
P
H
UN
D
io
io
R
R
e
.
C
C
T
.
o
M
M
/
j
e
o
D
tu
C
N
o
/
C
UN
N
R
UN
T
R
io
T
io
C
C
l
e
e
–
P
–
D
P
D
2
F
7
/
1
2
2
7
/
2
1
3
2
6
/
9
2
1
3
9
6
5
9
0
/
0
1
4
8
7
8
o
3
C
7
N
8
_
5
UN
/
_
j
0
o
0
C
8
N
5
9
_
UN
P
_
D
0
0
B
8
sì
5
G
9
tu
.
e
P
S
T
D
o
F
N
B
0
sì
7
S
M
e
IO
P
T
e
M
l
io
B
B
e
R
R
UN
2
R
0
2
io
3
e
S
/
j
T
/
F
.
tu
S
e
R
o
N
1
7
M
UN
sì
2
0
2
1
reports, indicates that the DMN flexibly shifts its functional
connections to contribute to the facilitation of task goals,
which may particularly involve interactions with certain
executive control (cioè., “task-positive”) brain regions/net-
works. Nonetheless, a comprehensive characterization of
the task-related changes focusing on DMN functional con-
nectivity across multiple task domains is currently lacking.
In this study, we tested the hypothesis that the DMN
supports goal-directed tasks through changes in func-
tional connectivity independent of whether DMN activity
is increased or decreased. Specifically, we hypothesized
Quello (1) the DMN would exhibit a dynamic reorganization
of its functional connectivity pattern in a task-specific
manner that would include enhanced interactions with
regions traditionally considered to support task perfor-
mance (cioè., “task-positive” regions: bilateral middle fron-
tal gyrus, inferior frontal gyrus, anterior insula, anterior
cingulate cortex; Gao et al., 2013) E (2) changes in
functional connectivity would be positively associated
with individual differences in measures of behavioral per-
formance. Six tasks encompassing diverse functional do-
mains were created to test this hypothesis including four
“external” goal-directed tasks associated with decreased
DMN activity, (cioè., working memory, inhibitory control,
lingua, emotion processing), two “internal” tasks known
to promote DMN activity (cioè., autobiographical memory,
movie watching). Activity changes were detected using
conventional general linear modeling, and task-related
DMN connectivity was characterized using seed-based
functional connectivity and supported with independent
components analysis (ICA). Functional connectivity during
each of these tasks was compared with that of a resting-
state scan, and the relevance of the detected connectivity
changes for behavioral performance was characterized.
The study findings support our hypothesis and reveal
new insights into the complex role of the DMN in normal
brain functioning.
METHODS
Participants
Twenty participants (11 women) between the ages of 21
E 38 years (mean = 28.5 years) were included in this
study, which was approved by the institutional review
board of the University of North Carolina at Chapel Hill.
All participants provided informed consent to participate
after a thorough explanation of study procedures. All par-
ticipants were healthy controls based on the absence of
lifetime psychiatric or neurological disorders.
lected with an EPI sequence on a Siemens Trio 3T MRI
(Erlangen, Germany) and 32-channel head coil (repetition
time = 1500 msec, echo time = 24 msec, flip angle = 70,
matrix = 64 × 64 mm2, voxel size = 4 × 4 × 4 mm3, 29
axial slices). Inoltre, a 5-min T1 MPRAGE scan provided
anatomical data for each participant (matrix = 256 ×
256 mm2, 192 sagittal slices, voxel size = 1 × 1 ×
1 mm3). Data preprocessing was conducted with Analysis
of Functional Neuroimages (AFNI) software and included
slice timing correction, deobliquing, motion correction,
despiking of noise timepoints, alignment to the partici-
pant’s anatomical image, warping to an Montreal Neuro-
logical Institute (MNI) template, removal by regression
of signal from white matter and cerebral spinal fluid as well
as the six motion covariates, linear detrending, Gaussian
smoothing at 8 mm FWHM and scaling to percent signal
change and bandpass filtering (008–0.08 Hz). For BOLD
activation detection, the bandpass filtering step was omit-
ted. Any data sets for which greater than 10% of acquired
volumes exhibited motion (>0.5 mm shift in head motion
in addition to greater than a 0.5% change in BOLD signal
from the previous repetition time) during a particular task
were excluded from analyses of that task. Final analyses
included 17 (emotion, inhibitory control), 16 (working
memory, autobiographical memory, movie watching), O
15 (lingua) participants.
fMRI Tasks
To enable comparison of findings across tasks, we aimed
to fix as many task design parameters as possible. Aside
from the movie watching and resting-state scans, IL
five other scans included block design tasks in which five
40-sec experimental blocks alternated with five 40-sec
control blocks. All tasks were visual in nature and utilized
pictures rather than words/letters. For all tasks except the
autobiographical memory task, blocks were preceded by
a 2-sec cue indicating task instructions for that block and
a 1-sec blank screen. For those four tasks, stimuli were dis-
played in color in the center of a black screen for 600 msec
and followed by a fixed 1000 msec ISI, resulting in 25 trials
per block. Così, stimuli were presented in a rapid, contin-
uous manner, allowing just enough time between stimuli
for participants to make a button press response. On the
other hand, because of the nature of the autobiographical
memory task, each 40-sec block contained a single stimu-
lus and was not preceded by a cue or ISI. Finalmente, all tasks
included three 20-sec rest blocks presented at the begin-
ning, middle, and end of the scan during which a fixation
cross was displayed, producing a total scan duration of
approximately 8 min for each task.
Data Acquisition and Preprocessing
Participants performed a battery of tasks from six different
domini (working memory, inhibitory control, emotion,
lingua, autobiographical memory, and movie watch-
ing) while undergoing fMRI. Functional images were col-
Autobiographical Memory
Each participant provided five photographs they had
taken on different occasions and at different places to
be displayed during the scanning session. Control stimuli
2370
Journal of Cognitive Neuroscience
Volume 27, Numero 12
D
o
w
N
l
o
UN
D
e
D
F
R
o
M
l
l
/
/
/
/
j
T
T
F
/
io
T
.
:
/
/
H
T
T
P
:
/
D
/
o
M
w
io
N
T
o
P
UN
R
D
C
e
.
D
S
F
io
R
o
l
M
v
e
H
R
C
P
H
UN
D
io
io
R
R
e
.
C
C
T
.
o
M
M
/
j
e
o
D
tu
C
N
o
/
C
UN
N
R
UN
T
R
io
T
io
C
C
l
e
e
–
P
–
D
P
D
2
F
7
/
1
2
2
7
/
2
1
3
2
6
/
9
2
1
3
9
6
5
9
0
/
0
1
4
8
7
8
o
3
C
7
N
8
_
5
UN
/
_
j
0
o
0
C
8
N
5
9
_
UN
P
_
D
0
0
B
8
sì
5
G
9
tu
.
e
P
S
T
D
o
F
N
B
0
sì
7
S
M
e
IO
P
T
e
M
l
io
B
B
e
R
R
UN
2
R
0
2
io
3
e
S
/
j
/
T
.
F
tu
S
e
R
o
N
1
7
M
UN
sì
2
0
2
1
consisted of five unfamiliar photographs obtained from
the Internet and loosely matched for content (people,
scenes, edifici, eccetera.). Each photograph was displayed
for 40 sec. During blocks in which the participant’s
own photographs were displayed, the instructions were
to think about and remember in as much detail as possi-
ble the occasion depicted in the picture. Allo stesso modo, partic-
ipants were instructed to imagine the event depicted in
the unfamiliar photographs from the perspective of the
hypothetical person who took the picture, creating hypo-
thetical details related to that picture. At the completion
of the scanning session, participants rated their ability to
remember details related to each of their own pictures
(memory strength) on a scale of 1–5, Dove 5 indicates
a vivid memory with many details recalled. Additionally,
participants reported on 5-point scales how recently the
photographs were taken (cioè., 1 month, 2–12 months, 1–
5 years, 5–10 years, 10+ years) and the emotional quality
of the memory (very negative to very positive). All pho-
tographs for participants included in this study were
rated 3 (neutro ), 4 (somewhat positive), O 5 (very
positive).
Emotion
An emotion judgment task required participants to deter-
mine the valence (“positive” or “negative”) of emotional
facial expressions. Task stimuli were male and female faces
obtained from the NimStim stimulus set (www.macbrain.
org/resources.htm). Participants responded by pressing
one button if the emotion was positive or another button
if the emotion was negative. As a control task, partici-
pants judged the sex of the individual in the image (por-
traying a neutral facial expression) and responded by
pressing one button for male and another for female.
An equal number of each alternative stimulus was pre-
sented in a randomized order. Accuracy and RTs (for
correct responses) were recorded for each trial.
Inhibitory Control
A go/no-go task was used to assess inhibitory control.
Computer-rendered pictures of neutral objects down-
loaded from the Object Databank (Stimulus images cour-
tesy of Michael J. Tarr, Center for the Neural Basis of
Cognition and Department of Psychology, Carnegie
Mellon University, www.tarrlab.org/) served as the task
stimuli. Participants were instructed to respond by press-
ing a button as quickly as possible whenever a picture ap-
peared on the screen but to withhold their response at
the presentation of the no-go stimulus (a “chain”), Quale
appeared in 24% (6/25) of trials. Go/no-go blocks alter-
nated with control blocks in which participants were in-
formed by the preceding cue that the no-go stimulus
would not appear. Accuracy was based on inhibition tri-
COME: A nonresponse for no-go trials was considered cor-
rect, and errors of commission were incorrect. RT was
measured for all responses on go trials.
Working Memory
Working memory was assessed with an n-back task, con-
sisting of a 2-back task and a 0-back control task. Stimuli
for the n-back task were the same as for the go/no-go
task (above) with the exception of the “chain.” During
the 2-back portion of the task, participants were to press
a button if the picture on the screen matched the one
presented two trials before it. The 0-back control task
participants were instructed to respond to the appear-
ance of the target picture (a kite). Button presses were
required in 24% (6/25) of trials for each block. Correct
responses, errors (omission or commission), and RTs
for correct responses were recorded.
Language
For the language domain, we chose a covert (silent)
object-naming task. Experimental stimuli consisted of
125 neutral object images obtained for the Object Data-
bank. During the experimental blocks, participants were
instructed to silently name the object depicted on the
screen. Object naming blocks alternated with control
blocks in which nonsense images (distorted versions of
original images) were displayed and participants were in-
structed to not name the images. Because of the nature
of the task, no behavioral measures were acquired during
this task.
Movie
The movie watching scan consisted of an excerpt from
IL 1925 silent film, Seven Chances, edited to a length
of approximately 7.5 min. The film largely depicts a series
of social interactions that drive various comedic sce-
narios. All intertitles (cioè., dialogue text) were removed
to avoid eliciting brain activity related to reading or a dis-
ruption in the visual flow of the film.
Task instructions were thoroughly explained to partic-
ipants before the scan, and for the tasks requiring button
presses (emotion judgment, n-back, go/no-go), partici-
pants were provided as much time as needed to practice
the tasks before entering the scanner. The resting-state
scan, during which participants were instructed to look
at a white fixation cross on a black screen for 5 min, pre-
ceding all other scans, but otherwise the tasks were coun-
terbalanced across participants using three different
counterbalance orders.
Statistical Analyses
Following data preprocessing, general linear modeling of
the task design, in which the experimental and control
blocks were modeled as boxcar functions and the task
Elton and Gao
2371
D
o
w
N
l
o
UN
D
e
D
F
R
o
M
l
l
/
/
/
/
j
F
/
T
T
io
T
.
:
/
/
H
T
T
P
:
/
D
/
o
M
w
io
N
T
o
P
UN
R
D
C
e
.
D
S
F
io
R
o
l
M
v
e
H
R
C
P
H
UN
D
io
io
R
R
e
.
C
C
T
.
o
M
M
/
j
e
o
D
tu
C
N
o
/
C
UN
N
R
UN
T
R
io
T
io
C
C
l
e
e
–
P
–
D
P
D
2
F
7
/
1
2
2
7
/
2
1
3
2
6
/
9
2
1
3
9
6
5
9
0
/
0
1
4
8
7
8
o
3
C
7
N
8
_
5
UN
/
_
j
0
o
0
C
8
N
5
9
_
UN
P
_
D
0
0
B
8
sì
5
G
9
tu
.
e
P
S
T
D
o
F
N
B
0
sì
7
S
M
e
IO
P
T
e
M
l
io
B
B
e
R
R
UN
2
R
0
2
io
3
e
S
/
j
F
/
.
T
tu
S
e
R
o
N
1
7
M
UN
sì
2
0
2
1
cues modeled as stick functions (where applicable), era
used to detect task-related activity. Activation estimates
from experimental blocks were contrasted with control
blocks to isolate task-related activity in keeping with con-
ventional fMRI task analytical procedures (Friston et al.,
1994). Second-level group analyses of this contrast were
calculated in one-sample mixed-effect meta-analyses
using 3dMEMA in AFNI. Results were corrected for mul-
tiple comparisons (α = 0.05) using a cluster level correc-
zione ( P < .05, minimum cluster size = 128 voxels) based
on Monte Carlo simulations conducted in 3dClustSim in
AFNI.
For seed-based functional connectivity analyses, four
different 8-mm radius spheres were placed in key nodes
of the default node network using previously established
coordinates ( Vincent, Kahn, Snyder, Raichle, & Buckner,
2008) for the posterior cingulate cortex (PCC; MNI coor-
dinates: x = 1, y = −55, z = 17), the ventromedial pFC
(MNI coordinates: x = 0, y = 51, z = −7), the left pos-
terior inferior parietal lobule (MNI coordinates: x = −47,
y = −71, z = 29), and the right posterior inferior parietal
lobule (MNI coordinates: x = 50, y = −64, z = 27). The
mean time series of voxels within each sphere was calcu-
lated for each participant and concatenated across the ex-
perimental task blocks (or the entire scan in the case of
the movie viewing scan) to serve as reference time series
for seed-based functional connectivity analyses. Thus
four seed-based functional connectivity maps were de-
rived for each task. Task-based functional connectivity
maps were compared with similarly derived connectivity
maps for the resting-state scan for each of the four DMN
seeds using a paired t test, consistent with other studies
of task-related functional connectivity changes (Elton &
Gao, 2014; Krienen, Yeo, & Buckner, 2014). Results were
corrected for multiple comparisons ( p < .05, minimum
cluster size = 128). A composite map of significant voxels
from the task-minus-rest contrast for all four seeds was
calculated for each task.
To examine the similarities of DMN connectivity pat-
terns across tasks, we examined the voxel-wise functional
connectivity changes obtained from the seed-based con-
nectivity analyses. Group mean functional connectivity
differences between each task and rest for all voxels in
the brain was vectorized for each ROI. Next, the voxel-
wise correlation across the six tasks was computed and,
following a Fisher z-transform, was then averaged across
the four ROIs.
A secondary analysis, specifically focused on between-
network connections derived from ICA, was conducted
to provide an additional, network level approach to un-
derstand the results obtained from seed-based analyses.
ICA is a data reduction method, which, when applied to
fMRI data, is capable of providing independent compo-
nents that closely correspond with brain networks iden-
tified by other functional connectivity methods (Calhoun,
Adalı, & Pekar, 2004). For the current study, the ICA was
conducted using the Infomax algorithm in the GIFT
group ICA toolbox (v3.0a; Calhoun, Adali, Pearlson, &
Pekar, 2001) implemented in Matlab R2011a. Each fMRI
data set from all participants and all scans was included
in a single group ICA to establish correspondence be-
tween identified components across both individuals and
tasks. On the basis of estimates from 20 ICASSO iterations
to test for the stability of the result, we solved for 15 inde-
pendent components. The 15 component spatial maps
were visually inspected to identify those representing ob-
vious artifacts, resulting in the removal of one motion-
related component in which peak values were around the
outside of the brain. The remaining component spatial
maps corresponded with identifiable neural networks.
For each of the group component spatial maps, GIFT
calculates time courses corresponding with each compo-
nent for each scan entered into the analysis (i.e., for each
participant and each task). Calculation of functional con-
nectivity entailed the pairwise Pearson correlation of the
14 time courses, providing a 14-by-14 correlation matrix,
for each participant and each task. The resulting correla-
tion matrices were normalized with a Fisher z-transform.
Task-related functional connectivity changes were calcu-
lated from paired t tests of each task and rest.
To calculate the canonical correlations between the
seed-based functional connectivity changes and task per-
formance, first, for each task, voxels demonstrating a con-
vergence of significant task-dependent changes in
connectivity across all four ROIs at the group level were
identified for positive and negative regions. Then, for
each task, each participant, and each ROI, the mean dif-
ference in connectivity between the task and resting-state
scans within the intersecting voxels was calculated, pro-
ducing a set of two functional connectivity variables (i.e.,
positive and negative changes) for each task and ROI.
The second set of variables were behavioral measures,
which varied by task: Mean postscan ratings of memory
strength, distance, and emotion for the five photographs
were calculated for the autobiographical memory task;
mean RT, standard deviation of RT, and accuracy were
calculated for the emotion, inhibitory control, and work-
ing memory tasks. The language task and movie watching
did not provide behavioral output and were therefore not
included in behavioral analyses. For this analysis, we con-
sider behavior during the experimental task block only.
For each task with behavioral measures, the two sets of
variables (2 connectivity variables, 3 behavioral variables)
were entered into a partial canonical correlation analysis
controlling for ROI in SAS 9.3 software to examine the
relationship between functional connectivity changes
and task performance. Significance was defined as p <
.05 after FDR correction (Benjamini & Yekutieli, 2001).
RESULTS
The detected activations for each task were highly consis-
tent with previous studies of autobiographical memory
(Spreng, Mar, & Kim, 2009; Summerfield, Hassabis, &
2372
Journal of Cognitive Neuroscience
Volume 27, Number 12
D
o
w
n
l
o
a
d
e
d
f
r
o
m
l
l
/
/
/
/
j
t
t
f
/
i
t
.
:
/
/
h
t
t
p
:
/
D
/
o
m
w
i
n
t
o
p
a
r
d
c
e
.
d
s
f
i
r
o
l
m
v
e
h
r
c
p
h
a
d
i
i
r
r
e
.
c
c
t
.
o
m
m
/
j
e
o
d
u
c
n
o
/
c
a
n
r
a
t
r
i
t
i
c
c
l
e
e
-
p
-
d
p
d
2
f
7
/
1
2
2
7
/
2
1
3
2
6
/
9
2
1
3
9
6
5
9
0
/
0
1
4
8
7
8
o
3
c
7
n
8
_
5
a
/
_
j
0
o
0
c
8
n
5
9
_
a
p
_
d
0
0
b
8
y
5
g
9
u
.
e
p
s
t
d
o
f
n
b
0
y
7
S
M
e
I
p
T
e
m
L
i
b
b
e
r
r
a
2
r
0
2
i
3
e
s
/
j
t
f
/
.
u
s
e
r
o
n
1
7
M
a
y
2
0
2
1
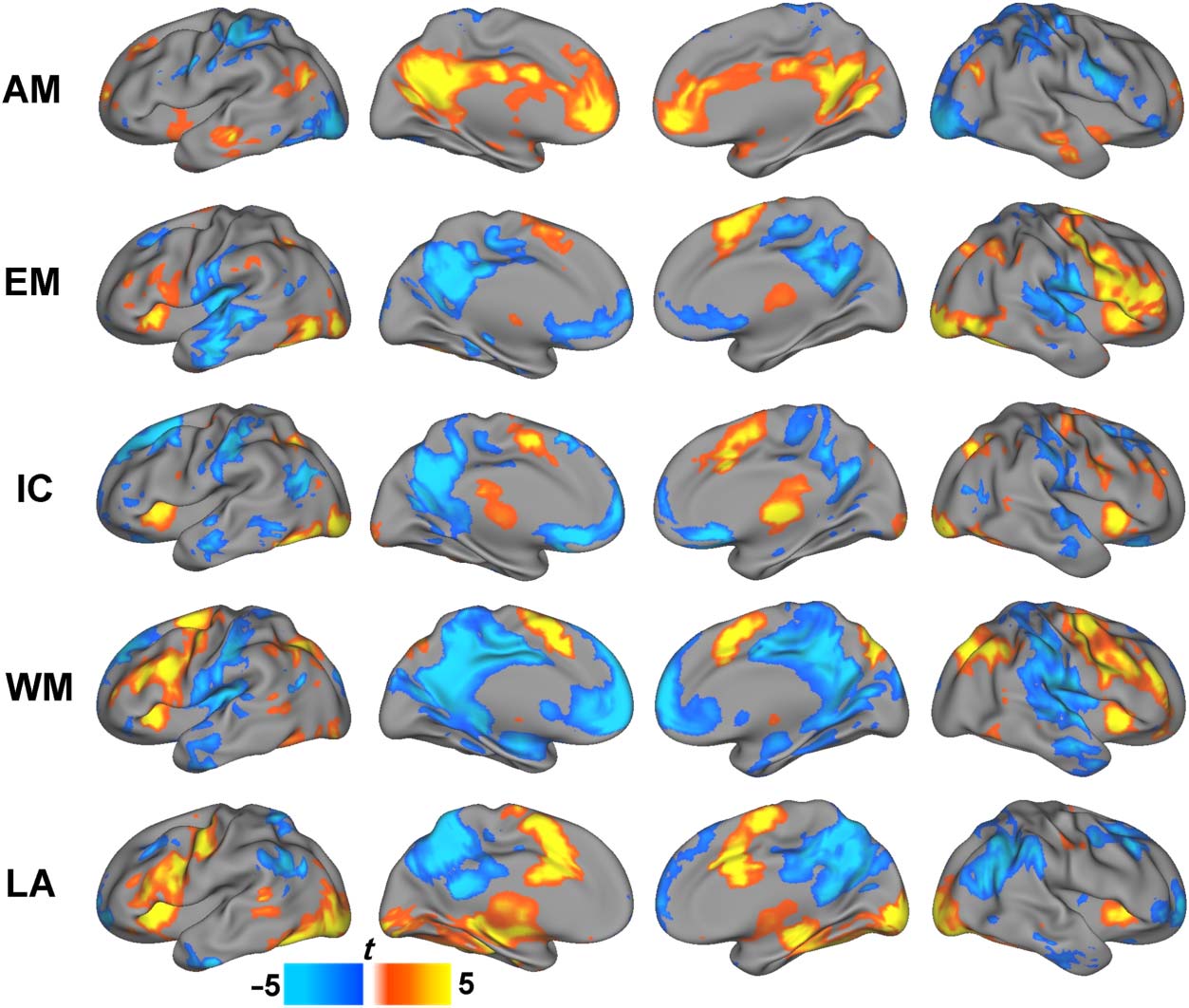
Figure 1. Activity changes
associated with five block
design tasks. Both task-related
activations (warm colors) and
deactivations (cool colors) are
shown. Significance was defined
as cluster-size level corrected
p < .05. AM = autobiographical
memory; EM = emotion;
IC = inhibitory control;
WM = working memory;
LA = language.
D
o
w
n
l
o
a
d
e
d
f
r
o
m
l
l
/
/
/
/
j
t
t
f
/
i
t
.
:
/
/
h
t
t
p
:
/
D
/
o
m
w
i
n
t
o
p
a
r
d
c
e
.
d
s
f
i
r
o
l
m
v
e
h
r
c
p
h
a
d
i
i
r
r
e
.
c
c
t
.
o
m
m
/
j
e
o
d
u
c
n
o
/
c
a
n
r
a
t
r
i
t
i
c
c
l
e
e
-
p
-
d
p
d
2
f
7
/
1
2
2
7
/
2
1
3
2
6
/
9
2
1
3
9
6
5
9
0
/
0
1
4
8
7
8
o
3
c
7
n
8
_
5
a
/
_
j
0
o
0
c
8
n
5
9
_
a
p
_
d
0
0
b
8
y
5
g
9
u
.
e
p
s
t
d
o
f
n
b
0
y
7
S
M
e
I
p
T
e
m
L
i
b
b
e
r
r
a
2
r
0
2
i
3
e
s
/
j
f
.
/
t
u
s
e
r
o
n
1
7
M
a
y
2
0
2
1
Maguire, 2009), emotion judgment (Phan, Wager, Taylor,
& Liberzon, 2002), inhibitory control (Wager et al., 2005;
Rubia et al., 2001), working memory (Owen, McMillan,
Laird, & Bullmore, 2005), and language (Burgund, Lugar,
Miezin, & Petersen, 2003; Figure 1). In particular, the
emotion judgment, inhibitory control, working memory,
and language tasks each exhibited decreased task-related
activity in regions consistent with the DMN (i.e., PCC,
precuneus, medial pFC, middle temporal gyrus). Con-
versely, the autobiographical memory task was character-
ized by task-related increases in activity in the same DMN
regions. The continuous nature of the movie watching
scan precluded an analysis of task-related activity.
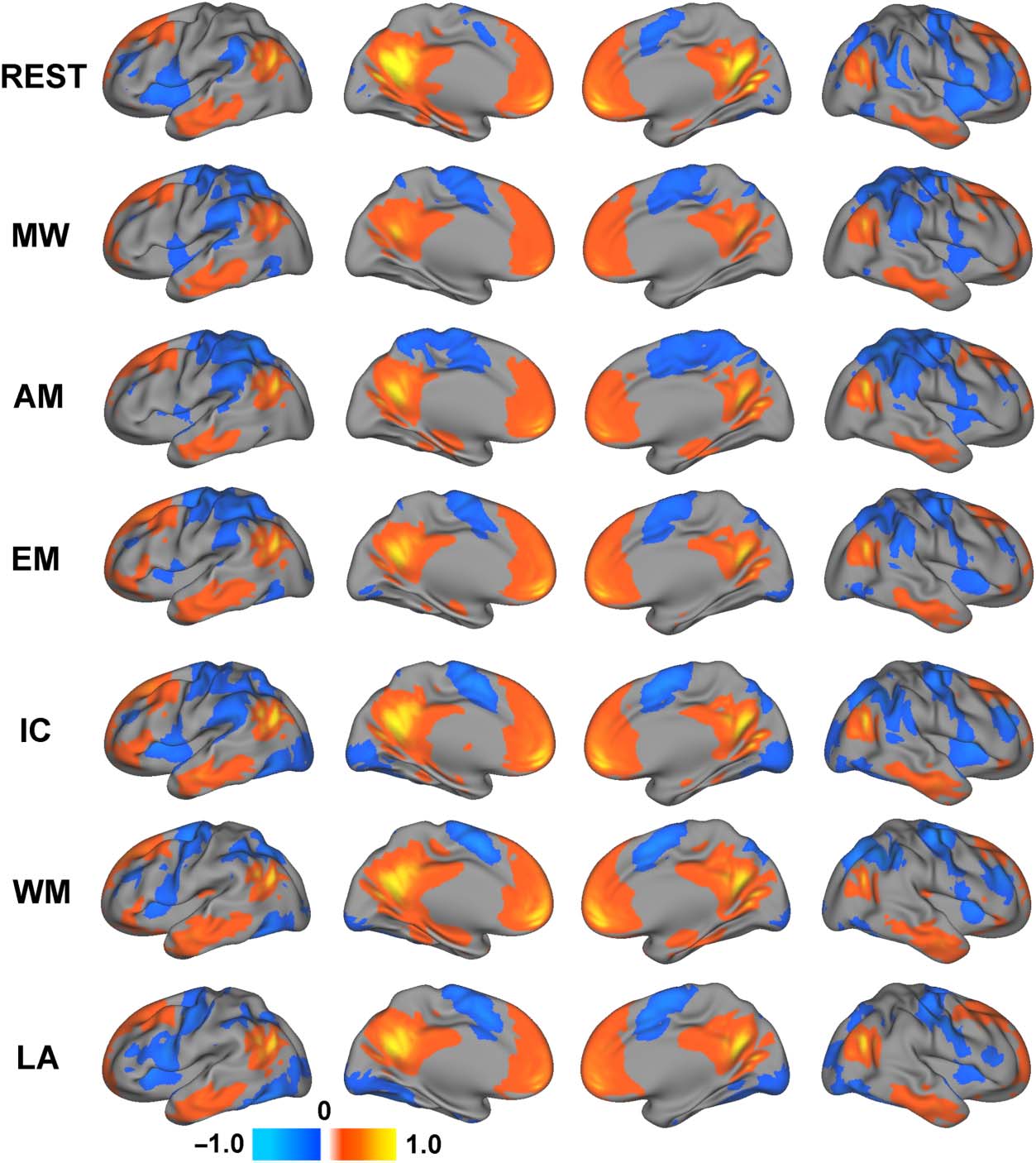
Maps of the average functional connectivity of the four
seeds for each task are presented in Figure 2. The net-
work level composite map of the connectivity dynamics
associated with the four DMN ROIs is presented in Fig-
ure 3. Despite similarities in functional connectivity
across these seven brain states, consistent with prior
work (Krienen et al., 2014), the task-related changes in
functional connectivity relative to the resting state were
extensive. Consistent with our hypothesis, differences
in functional connectivity patterns during task blocks
compared with the resting-state scan were observed for
all defined DMN ROIs. Although each task demonstrated
its own unique functional connectivity pattern, increased
DMN connectivity was generally observed across the
tasks within the middle and inferior frontal cortex and an-
terior insula, which are typically attributed to executive
control functions and traditionally regarded as having
positive contributions to task performance (Spreng,
2012). Furthermore, a pattern of increased anterior insula
connectivity and decreased posterior cingulate connec-
tivity observed for each of these tasks is mirrored in
the task-related activation maps for the four external
tasks, further supporting the task-positive nature of
these connectivity changes.
Certain functional connectivity patterns appeared to be
consistent across all task domains, suggesting some com-
monalities across the tasks relative to rest. To examine
these commonalities, Figure 3B presents regions show-
ing task-related connectivity changes across at least three
ROIs for all six tasks. Relative to the resting-state, DMN
ROIs engaged a cluster centered in the right inferior
frontal gyrus (MNI coordinates: x = 48, y = 35, z = 0;
97 voxels) during all six tasks. Similarly, there was overlap
across all tasks for functional connectivity decreases in the
PCC and medial visual cortex (MNI coordinates: x = −1,
y = −52, z = 19; 356 voxels), brainstem (MNI coor-
dinates: x = −8, y = −24, z = −7; 67 voxels), and cere-
bellum (MNI coordinates: x = 4, y = −51, z = 019;
47 voxels).
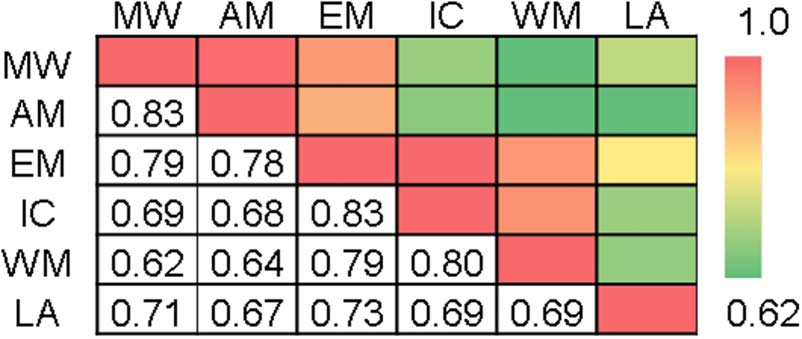
The correlations of the dynamic DMN functional con-
nectivity patterns across the six tasks suggest certain sim-
ilarities exist across the task domains (Figure 4). In
particular, the calculated correlations support the clas-
sification of these tasks into at least two major categories,
which seem to correspond with whether the task re-
quires internally and externally focused cognitions. Spe-
cifically, the two internal tasks—movie watching and
autobiographical memory—demonstrated highly similar
patterns of connectivity (r = .83, p < .001). Similarly,
high correlations were also detected among the emotion,
inhibitory control, and working memory tasks, which
ranged between r = .79 and r = .83. Although the
emotion task exhibited a high degree of similarity to the
Elton and Gao
2373
other tasks requiring external cognitions, there were also
relatively high correlations between the emotion task and
the two internal tasks (movie watching: r = .79, p < .001;
autobiographical memory tasks: r = .83, p < .001), sug-
gesting that this task may involve both internal and exter-
nal cognitions. Functional connectivity changes during
the language task were most closely associated with the
emotion task (r = .73, p < .001).
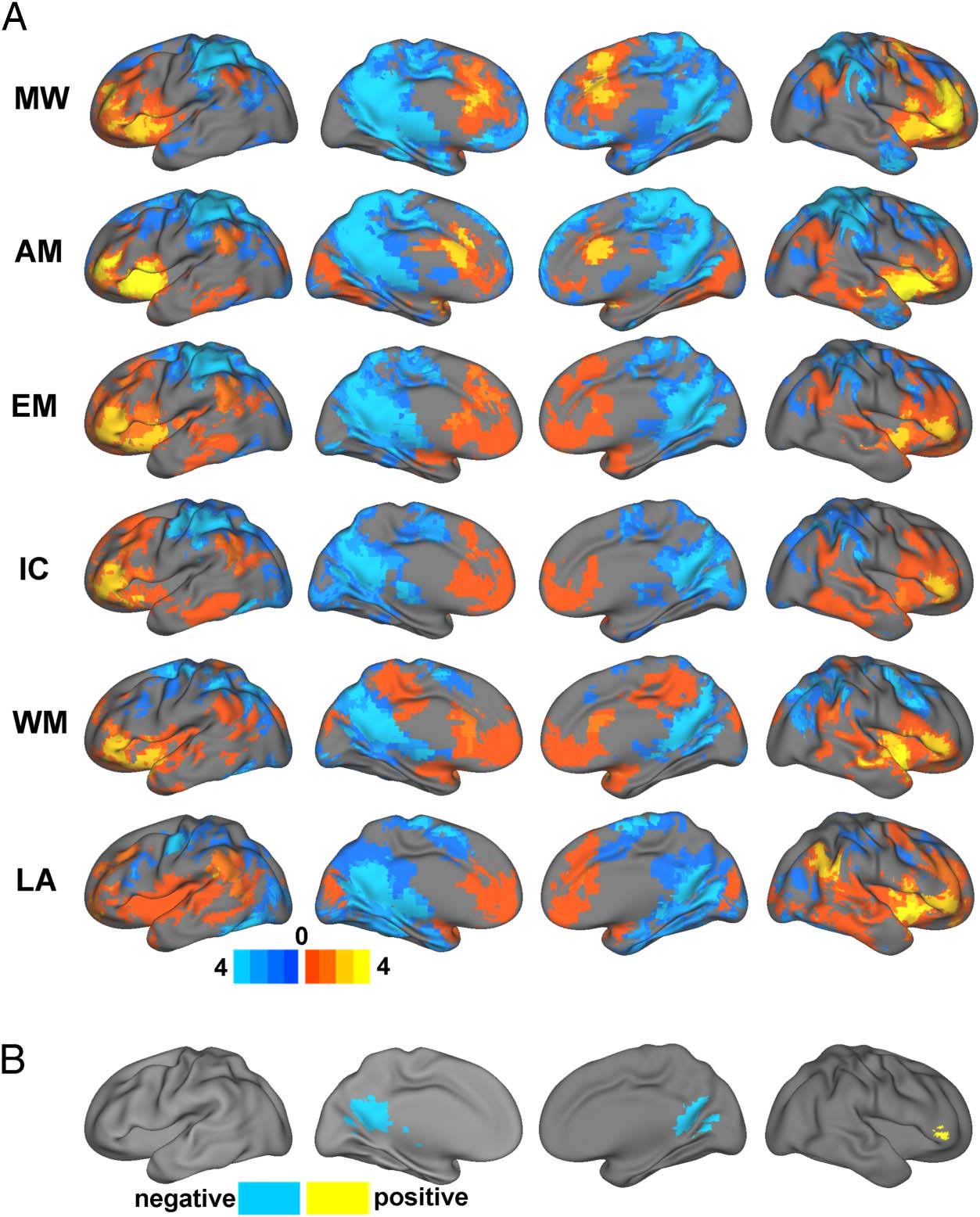
For the ICA analysis, the DMN was represented by two
independent components, corresponding with the poste-
rior (pDM) and anterior (aDM) portions of the network con-
sistent with prior reports (Di & Biswal, 2014; Uddin, Clare
Kelly, Biswal, Castellanos, & Milham, 2009; Damoiseaux
et al., 2006). For descriptive and visualization purposes,
functional connectivity of the two DMN components
was averaged to provide a single network level measure
of DMN functional connectivity. However, changes in
DMN connectivity were considered significant if either
one of the two components demonstrated a significant
effect. Within-DMN functional connectivity was repre-
sented by the connectivity between the two DMN com-
ponents (i.e., pDM and aDM). Four components were
identified as representing higher-order task-positive net-
works belonging to the so-called “control system” as re-
ported in previous studies (Cole, Repovs, & Anticevic,
2014; Di & Biswal, 2014). These networks included the
dorsal attention network, salience network, right fronto-
parietal network, and left frontoparietal network. These
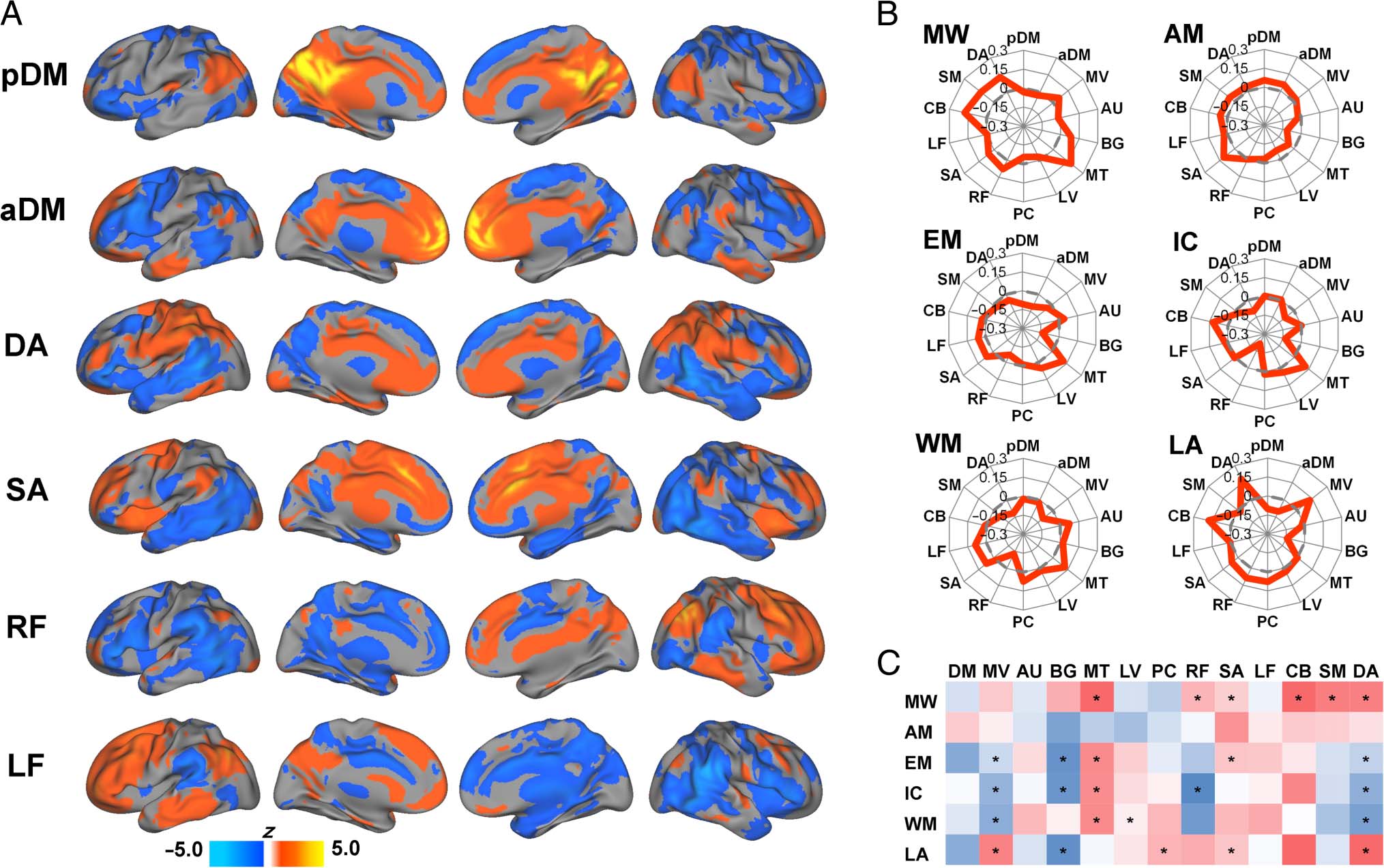
selected networks are displayed in Figure 5A. Networks
were identified and labeled based on visual inspection of
their resemblance to resting-state networks reported in
previous studies (Di & Biswal, 2014; Smith et al., 2009).
To illustrate the task-related dynamic patterns of DMN
connectivity in relation to large-scale networks derived
from ICA, spider plots of the functional connectivity
changes of the DMN components with each of the other
networks are displayed in Figure 5B with significant
changes noted in Figure 5C. The patterns of functional
connectivity changes in this network level analysis dem-
onstrated both commonalities across tasks as well as task-
specific features. Similar to the seed-based results, findings
based on ICA time courses indicated decreased within-
DMN functional connectivity (nonsignificant) for each
of the tasks relative to rest except for the autobiograph-
ical memory task. Regarding functional connectivity with
the designated task-positive networks, one notable com-
monality was that the DMN increased functional connec-
tivity with the salience network for all six tasks compared
with rest, which was statistically significant for pDM dur-
ing the movie watching, emotion, and language tasks.
The left frontoparietal network also demonstrated non-
significant increases with the DMN during five of the
Figure 2. Average functional
connectivity of the four DMN
ROIs during each of the six
tasks as well as the resting
state. REST = resting state;
MW = movie watching;
AM = autobiographical
memory; EM = emotion;
IC = inhibitory control;
WM = working memory;
LA = language.
D
o
w
n
l
o
a
d
e
d
f
r
o
m
l
l
/
/
/
/
j
f
/
t
t
i
t
.
:
/
/
h
t
t
p
:
/
D
/
o
m
w
i
n
t
o
p
a
r
d
c
e
.
d
s
f
i
r
o
l
m
v
e
h
r
c
p
h
a
d
i
i
r
r
e
.
c
c
t
.
o
m
m
/
j
e
o
d
u
c
n
o
/
c
a
n
r
a
t
r
i
t
i
c
c
l
e
e
-
p
-
d
p
d
2
f
7
/
1
2
2
7
/
2
1
3
2
6
/
9
2
1
3
9
6
5
9
0
/
0
1
4
8
7
8
o
3
c
7
n
8
_
5
a
/
_
j
0
o
0
c
8
n
5
9
_
a
p
_
d
0
0
b
8
y
5
g
9
u
.
e
p
s
t
d
o
f
n
b
0
y
7
S
M
e
I
p
T
e
m
L
i
b
b
e
r
r
a
2
r
0
2
i
3
e
s
/
j
.
t
f
/
u
s
e
r
o
n
1
7
M
a
y
2
0
2
1
2374
Journal of Cognitive Neuroscience
Volume 27, Number 12
Figure 3. Functional
connectivity of the DMN during
six tasks contrasted with rest.
(A) Overlap of DMN task-related
connectivity changes versus rest
across four ROIs. Brain maps
represent the number of ROIs
exhibiting significant positive
(warm colors) or negative (cool
colors) task-related functional
connectivity changes at each
voxel. (B) Overlap of DMN task-
related connectivity changes
versus rest across all six tasks.
MW = movie watching;
AM = autobiographical memory;
EM = emotion; IC = inhibitory
control; WM = working
memory; LA = language.
tasks compared with rest with the exception of movie
watching, which conversely demonstrated a significant
increase with the right frontoparietal network (t = 3.34,
p = .002). DMN connectivity changes with the dorsal at-
tention network were also highly task dependent, dem-
onstrating significant increases for the aDM during the
movie watching (t = 3.40, p = .002) and language tasks
(t = 4.10, p < .001), but significant decreases during the
Figure 4. Correlation matrix representing the extent to which
patterns of DMN connectivity (relative to rest) are similar across the
six tasks. MW = movie watching; AM = autobiographical memory;
EM = emotion; IC = inhibitory control; WM = working memory;
LA = language.
working memory (t = −3.03, p = .005), inhibitory con-
trol (t = −2.59, p = .014), and emotion (t = −2.65, p =
.012) tasks. Thus, in support of findings from the seed-
based analyses, these secondary analyses identified a
number of “task-positive” increases in DMN functional
connectivity during performance of various tasks relative
to rest. Furthermore, the unique patterns of functional
connectivity changes support the hypothesis that the
DMN undergoes functional reorganization in a task-specific
manner.
Finally, to determine the behavioral relevance of task-
dependent changes in connectivity, we investigated the
association of DMN connectivity changes with task be-
havioral measures. Because both increases and decreases
in functional connectivity could potentially contribute to
enhanced task performance (Gao et al., 2013), we adopted
a multivariate approach to test the association of the
observed positive and negative functional connectivity
changes with task performance variables using canonical
correlation analysis, which finds the linear combination
of two sets of variables that maximizes their correlation.
Canonical correlations, as well as variable loadings and
cross loadings for significant canonical variate pairs, are
Elton and Gao
2375
D
o
w
n
l
o
a
d
e
d
f
r
o
m
l
l
/
/
/
/
j
t
t
f
/
i
t
.
:
/
/
h
t
t
p
:
/
D
/
o
m
w
i
n
t
o
p
a
r
d
c
e
.
d
s
f
i
r
o
l
m
v
e
h
r
c
p
h
a
d
i
i
r
r
e
.
c
c
t
.
o
m
m
/
j
e
o
d
u
c
n
o
/
c
a
n
r
a
t
r
i
t
i
c
c
l
e
e
-
p
-
d
p
d
2
f
7
/
1
2
2
7
/
2
1
3
2
6
/
9
2
1
3
9
6
5
9
0
/
0
1
4
8
7
8
o
3
c
7
n
8
_
5
a
/
_
j
0
o
0
c
8
n
5
9
_
a
p
_
d
0
0
b
8
y
5
g
9
u
.
e
p
s
t
d
o
f
n
b
0
y
7
S
M
e
I
p
T
e
m
L
i
b
b
e
r
r
a
2
r
0
2
i
3
e
s
/
j
t
.
f
/
u
s
e
r
o
n
1
7
M
a
y
2
0
2
1
D
o
w
n
l
o
a
d
e
d
f
r
o
m
l
l
/
/
/
/
j
t
t
f
/
i
t
.
:
/
/
h
t
t
p
:
/
D
/
o
m
w
i
n
t
o
p
a
r
d
c
e
.
d
s
f
i
r
o
l
m
v
e
h
r
c
p
h
a
d
i
i
r
r
e
.
c
c
t
.
o
m
m
/
j
e
o
d
u
c
n
o
/
c
a
n
r
a
t
r
i
t
i
c
c
l
e
e
-
p
-
d
p
d
2
f
7
/
1
2
2
7
/
2
1
3
2
6
/
9
2
1
3
9
6
5
9
0
/
0
1
4
8
7
8
o
3
c
7
n
8
_
5
a
/
_
j
0
o
0
c
8
n
5
9
_
a
p
_
d
0
0
b
8
y
5
g
9
u
.
e
p
s
t
d
o
f
n
b
0
y
7
S
M
e
I
p
T
e
m
L
i
b
b
e
r
r
a
2
r
0
2
i
3
e
s
/
j
.
f
t
/
u
s
e
r
o
n
1
7
M
a
y
2
0
2
1
Figure 5. Results based on independent component analysis. (A) Mean values (z scores) for selected independent components. (B) Spider web plots
depicting changes in DMN functional connectivity (between each task and rest) across all independent components. (C) Color chart depicting
changes in DMN functional connectivity (between each task and rest) across all independent components. Red indicates degrees of positive change
in connectivity, and blue indicates degrees of negative change in connectivity. Asterisks denote significant functional connectivity changes. MV =
medial visual network; AU = auditory/language network; BG = basal ganglia network; MT = medial-temporal network; LV = lateral visual network;
PC = precuneus network; RF = right frontoparietal network; SA = salience network; LF = left frontoparietal network; CB = cerebellar network;
SM = sensorimotor network; DA = dorsal attention network; MW = movie watching; AM = autobiographical memory; EM = emotion; IC =
inhibitory control; WM = working memory; LA = language.
reported in Table 1. Significant relationships between
functional connectivity changes and task behavioral mea-
sures were detected for each task, demonstrating the be-
havioral relevance of the observed DMN dynamics. For the
autobiographical memory task, the significant canonical
variate pair indicated a particularly strong relationship of
more distant memories with stronger task-dependent in-
creases and decreases in connectivity (Wilks’ Lambda =
0.79, F(6, 112) = 2.39, p = .033). For the emotion task,
the significant canonical variate pair ( Wilks’ Lambda =
0.79, F(6, 88) = 2.25, p = .046) indicated that lesser
task-related functional connectivity increases and de-
creases were associated with longer RTs. The significant
canonical variate pair for the n-back working memory task
(Wilks’ Lambda = 0.78, F(6, 112) = 2.48, p = .027) sug-
gested that lesser task-related connectivity decreases in
negative regions were associated with both longer RTs
and greater RT variability. Finally, the significant canonical
variate pair for the go/no-go inhibitory control task (Wilks’
Lambda = 0.69, F(6, 88) = 2.95, p = .011) demonstrated a
rather complex relationship that does not lend itself to a
clear interpretation.
DISCUSSION
The current study provides strong evidence that, inde-
pendent of the direction of activity changes, the DMN
undergoes task-positive changes in functional connectiv-
ity, which was demonstrated by both task-dependent
reorganization of functional connectivity as well as signif-
icant connectivity–behavior relationships. In fact, the
study findings indicated that the DMN increases func-
tional connectivity with certain regions/networks that
are implicated in the executive control of task perfor-
mance, casting further doubt on the notion that the
DMN is solely associated with task-irrelevant thoughts
or behaviors during external tasks. Thus, the discrepancy
between activity changes and connectivity changes asso-
ciated with the DMN emphasizes the need for future re-
search to consider both channels of information to
2376
Journal of Cognitive Neuroscience
Volume 27, Number 12
provide a more thorough understanding of the functional
roles of this and other networks.
The “tonically active” nature of DMN regions during
unconstrained rest has led to the postulation that the
DMN is involved in broadband monitoring of both the in-
ternal and external world to maintain self-consciousness
and vigilance during this state (Gilbert et al., 2007;
Raichle et al., 2001). This broadband monitoring function
would likely require extensive interactions between the
DMN and various specialized brain networks. Indeed, dif-
ferent studies have consistently documented the “hub”
role of core DMN regions within the brain, based on both
function and structure (van den Heuvel & Sporns, 2013;
Buckner et al., 2009). Therefore, we would argue that just
as the DMN may utilize multimodal information integra-
tion to support broadband monitoring of both the inter-
nal and external world during unconstrained rest, it may
also utilize its vast functional connections to flexibly facil-
itate performance of diverse goals during various task
states. The highly flexible and task-specific reorganization
of DMN connectivity suggest that the DMN may act as a
crossroad for multimodal integration across domain-
specific regions to support the current task. Indeed, we ob-
served positive coupling between the DMN and external
task-activated regions and uncoupling of external task-
deactivated regions with the DMN ROIs (Figures 1 and 2),
which was evident for both external and internal tasks.
The region that showed the most consistent positive cou-
pling with the DMN across all tasks was the right inferior
frontal gyrus, a key region belonging to the salience net-
work (Seeley et al., 2007), so-called for its role in detect-
ing salient stimuli. Indeed, a secondary analysis based on
ICA time series also suggested a task nonspecific role of
functional connectivity between a component identified
as the salience network and the DMN indicating a general
role in goal-directed cognitions. We have previously de-
scribed DMN increases with this network during a global/
local task (Elton & Gao, 2014; Gao et al., 2013), which we
have described as a mechanism to support the monitor-
ing of the internal and external environment. It is likely
that such monitoring is an important functional compo-
nent of each of the tasks included in this study, particu-
larly to identify the appearance of salient stimuli and/or to
monitor performance. Furthermore, recent work sug-
gests that the salience network may modulate DMN inter-
actions with the other “task-positive” networks (Di &
Biswal, 2014), highlighting the potential relevance of
such interactions for task performance.
Table 1. Canonical Correlations and Variable Correlations and Cross-correlations with Canonical Variates for Functional Connectivity
(FC) and Behavior
Task
Canonical Correlation
Autobiographical memory
0.44
p
.033
Variable
FC positive
FC negative
Strength of memory
Distance of memory
Emotional quality
Emotion
0.44
.046
FC positive
FC negative
Accuracy
RT
RT variability
Inhibitory control
0.52
.011
FC positive
FC negative
Accuracy
RT
RT variability
Working memory
0.44
.027
FC positive
FC negative
Accuracy
RT
RT variability
FC
0.64
−0.50
−0.13
0.39
−0.04
−0.36
0.76
0.18
0.35
0.20
0.44
1.00
0.43
0.19
0.22
−0.03
0.95
−0.02
0.32
0.32
Behavior
0.28
−0.22
−0.28
0.89
−0.09
−0.16
0.34
0.39
0.79
0.45
0.22
0.52
0.83
0.36
0.42
−0.02
0.42
−0.04
0.72
0.72
Elton and Gao
2377
D
o
w
n
l
o
a
d
e
d
f
r
o
m
l
l
/
/
/
/
j
t
t
f
/
i
t
.
:
/
/
h
t
t
p
:
/
D
/
o
m
w
i
n
t
o
p
a
r
d
c
e
.
d
s
f
i
r
o
l
m
v
e
h
r
c
p
h
a
d
i
i
r
r
e
.
c
c
t
.
o
m
m
/
j
e
o
d
u
c
n
o
/
c
a
n
r
a
t
r
i
t
i
c
c
l
e
e
-
p
-
d
p
d
2
f
7
/
1
2
2
7
/
2
1
3
2
6
/
9
2
1
3
9
6
5
9
0
/
0
1
4
8
7
8
o
3
c
7
n
8
_
5
a
/
_
j
0
o
0
c
8
n
5
9
_
a
p
_
d
0
0
b
8
y
5
g
9
u
.
e
p
s
t
d
o
f
n
b
0
y
7
S
M
e
I
p
T
e
m
L
i
b
b
e
r
r
a
2
r
0
2
i
3
e
s
/
j
t
f
/
.
u
s
e
r
o
n
1
7
M
a
y
2
0
2
1
The patterns of functional connectivity also showed
variations depending on the task (Figure 4). For example,
the two internal tasks (i.e., autobiographical memory and
movie watching) demonstrated highly similar patterns
but less similarity with the external tasks. The emotion
task showed high similarity to both the internal and ex-
ternal task categories in terms of its dynamic DMN con-
nectivity patterns. Thus, the changes in DMN functional
connectivity patterns appear to be task dependent, yet
also demonstrate a high degree of consistency across
tasks employing similar processes. The importance of
this task-specific reorganization is further underscored
by the association of these DMN connectivity changes
with behavioral variables associated with each of the
tasks (Table 1). As an example, during the emotion task,
both the speed and accuracy for detecting emotional va-
lence were positively related to DMN coupling with task-
positive regions including the anterior insula and inferior
frontal gyrus regions of the salience network. It is likely
that such functional coupling fosters the rapid and accu-
rate detection of the task-relevant feature (i.e., valence)
of the task stimulus (i.e., face). Such functional interac-
tions likely serve to positively influence goal-directed be-
haviors more generally, as the ability to identify salient
stimuli and/or to monitor performance is an important func-
tional component of goal-directed tasks. Overall, these find-
ings suggest that the ability of the DMN to flexibly interact
with extensive and distributed brain regions/networks may
exist not only as a means for broadband monitoring during
the resting state but also as an important functional mech-
anism to facilitate various behavioral goals during different
explicit task states. Therefore, we argue that, although
studies of task-evoked activity changes may have enabled
mapping of specialized functions across the brain, the
study of task-evoked connectivity reorganization may pro-
vide information regarding large-scale functional interac-
tions related to multimodal integration and facilitation, as
observed here for the DMN.
However, how do we reconcile the current findings
with the fact that the DMN typically shows decreased ac-
tivity during most externally oriented tasks? We might
find an explanation from the dual nature of DMN func-
tions. First, numerous studies have found that DMN re-
gions show task-evoked activity increases during a
range of self-referential cognitions including autobio-
graphical memory retrieval, self-related planning, and
mentalizing, indicating a specialized role of the DMN in
self-related thinking processes (Andrews-Hanna, 2012;
Spreng et al., 2009; Buckner & Carroll, 2007). During
the unconstrained resting state, the DMN may support
both self-related thinking (a specialized, domain-specific
function) as well as broadband monitoring (generalized in-
formation integration across domains). However, after
transitioning to external, goal-directed task performance,
the information integration function may continue to sup-
port broadband monitoring, potentially shifting to support
more task-specific multimodal integration/control, which
would likely entail a similar level of information process-
ing and thus a similar level of activity expenditure. On the
other hand, self-referential mental processes are likely
suspended during such external tasks (Christoff, Gordon,
Smallwood, Smith, & Schooler, 2009; Mason et al., 2007),
which would reduce the local processing needs that sup-
port such functions within the DMN. Therefore, the rel-
atively stable cost of large-scale multimodal integration
combined with decreased local processing within the
DMN could explain the net decrease in activity within
the DMN during external tasks. On the other hand, when
the local processing needs of the DMN exceeds that
of the resting state (i.e., during a dedicated internally
focused task such as autobiographical memory), we ob-
serve a corresponding increase of DMN activity (Figure 1).
Taken together, our findings seem to imply that brain
functions during both resting and task states may be sup-
ported by two large processing domains: (1) large-scale
network level information integration and coordination
supported by the connectivity dynamics of different func-
tional networks, including the DMN and likely certain
“control” networks that require a high but relatively con-
stant level of activity regardless of whether the brain is at
rest or involved in a specific task and (2) local and spe-
cialized task-specific processes supported by a discrete
set of regions, likely underlying the subtle activity changes
that are captured by conventional task fMRI activation
analyses. This dual-layer hypothesis provides a simple ex-
planation for the brain’s high level of flexibility to adapt to
different task states while exhibiting minimal (yet statisti-
cally detectable) activity changes.
Regardless of the underlying mechanisms, the ob-
served task-positive connectivity changes of the DMN
across a range of tasks and their association with behav-
ior reinforce the perspective (Spreng, 2012; Hampson,
Driesen, Skudlarski, Gore, & Constable, 2006) that this
network may contribute a wide range of functional defi-
cits spanning diverse domains. For instance, the view that
the DMN is suppressed during most tasks could hardly
reconcile the innumerable findings linking this network
to diverse task-related functional abnormalities spanning
both internal and external domains, including motor con-
trol deficits (e.g., Parkinson disease; Tessitore et al., 2012;
Van Dijk et al., 2010), attention deficits (e.g., ADHD;
Liddle et al., 2011; Uddin et al., 2008), social skill deficits
(e.g., autism; Lynch et al., 2013; Washington et al., 2013;
Murdaugh et al., 2012; Spencer et al., 2012), dysregulated
mood (e.g., bipolar disorder, depression; Sambataro,
Wolf, Pennuto, Vasic, & Wolf, 2013; Marchetti, Koster,
Sonuga-Barke, & De Raedt, 2012; Ongur et al., 2010;
Sheline et al., 2009), psychosis (e.g., schizophrenia;
Guo et al., 2014; Jang et al., 2011; Ongur et al., 2010;
Pomarol-Clotet et al., 2008), and memory loss/cognitive
disabilities (e.g., Alzheimer’s disease and dementia; Qi
et al., 2010; Greicius, Srivastava, Reiss, & Menon, 2004).
Rather, in addition to the typically postulated role of the
DMN in task-independent thoughts and processes, in
2378
Journal of Cognitive Neuroscience
Volume 27, Number 12
D
o
w
n
l
o
a
d
e
d
f
r
o
m
l
l
/
/
/
/
j
t
t
f
/
i
t
.
:
/
/
h
t
t
p
:
/
D
/
o
m
w
i
n
t
o
p
a
r
d
c
e
.
d
s
f
i
r
o
l
m
v
e
h
r
c
p
h
a
d
i
i
r
r
e
.
c
c
t
.
o
m
m
/
j
e
o
d
u
c
n
o
/
c
a
n
r
a
t
r
i
t
i
c
c
l
e
e
-
p
-
d
p
d
2
f
7
/
1
2
2
7
/
2
1
3
2
6
/
9
2
1
3
9
6
5
9
0
/
0
1
4
8
7
8
o
3
c
7
n
8
_
5
a
/
_
j
0
o
0
c
8
n
5
9
_
a
p
_
d
0
0
b
8
y
5
g
9
u
.
e
p
s
t
d
o
f
n
b
0
y
7
S
M
e
I
p
T
e
m
L
i
b
b
e
r
r
a
2
r
0
2
i
3
e
s
/
j
/
f
.
t
u
s
e
r
o
n
1
7
M
a
y
2
0
2
1
which impairments result from a failure to deactivate this
network (Sonuga-Barke & Castellanos, 2007), our findings
suggest that the DMN may also contribute to the neuropa-
thology of different brain disorders through dysconnectiv-
ity with task-relevant regions that support active brain
operations. For example, a previous study investigated al-
tered task-dependent connectivity of the PCC and medial
pFC in individuals with post-traumatic stress disorder
(PTSD) versus healthy controls during a blocked working
memory task (Daniels et al., 2010). The DMN seeds demon-
strated greater task-related connectivity with task-positive
regions in the control group versus the PTSD group,
whereas greater task-dependent connectivity for the PTSD
group was observed within DMN regions. The hypercon-
nectivity of the PTSD group within the DMN and reduced
integration with other task-positive networks during the
working memory task indicate a deficit in the appropriate
engagement of task-related brain networks, rather than sim-
ply an inability to suppress DMN activity. Other evidence of
deficits in multimodal integration of DMN structures con-
tributing to disease comes from reports indicating that
patients with schizophrenia exhibit decreased hub-like or-
ganization of cortical networks (Rubinov et al., 2009), par-
ticularly in the posterior cingulate/precuneus region (Lynall
et al., 2010), suggesting reduced communication between
this DMN region and other large-scale networks. Therefore,
although prevailing theories typically lead to the interpreta-
tion that altered DMN connectivity contributes to deficits in
self-related processes, emerging evidence also suggests that
such abnormalities could impact numerous other func-
tional domains through altered connectivity with relevant
brain regions. Given the role of the DMN as a hub network
in addition to the immense diversity of disorders in which
the DMN has been implicated (Broyd et al., 2009), it is pos-
sible that certain brain disorders could be associated with
multimodal integration deficits related to altered DMN
dynamics. Indeed, the relationship of DMN connectivity
dynamics to behavior demonstrated by the current study
represents a promising model for understanding symp-
toms of mental health disorders.
Several study limitations deserve discussion. First, a seed-
based approach was adopted to delineate the dynamic con-
nectivity of the DMN network, limiting our conclusions to
the specific seeds selected. However, our results showed a
high degree of overlap across all four of the selected DMN
seeds (Figure 3) supporting our network level conclusions
regarding the DMN. Furthermore, our conclusions were
supported by an ICA-based approach. However, a limita-
tion of ICA is the optimization requirement of this ap-
proach, which can produce small variations in results
when solving for a different number of components. An-
other limitation of the current study is that our sample size
is moderate (n = 15–17 participants per task); thus, the
brain–behavioral correlation analyses would be strength-
ened by future independent validation.
Overall, the current findings from six distinct task do-
mains provide compelling evidence that the DMN does
not always operate under a “default” mode and may ac-
tively participate in both internal and external goal-directed
tasks through dynamic connectivity. The functional rele-
vance of such task-related connectivity changes was
demonstrated by their association with behavioral mea-
sures for each task. The current investigation represents
the first systematic investigation focusing on the task-
dependent connectivity of this network and demonstrat-
ing its positive contributions to a range of explicit task
states. A dual-layer functional mechanism may reconcile
the seemingly discrepant activity and connectivity changes
under different task states. The novel findings in this study
provide a new perspective from which to understand
DMN function and its contribution to various brain
disorders.
Acknowledgments
This work was supported by a University of North Carolina at
Chapel Hill start-up to W. G.
Reprint requests should be sent to Wei Gao, Biomedical Imag-
ing Research Institute (BIRI), Department of Biomedical Sciences
and Academic Imaging, Cedars-Sinai Medical Center, PACT 800
7G, 116 N. Robertson Blvd., Los Angeles, CA 90048, or via e-mail:
gaow@cshs.org.
REFERENCES
Andrews-Hanna, J. R. (2012). The brain’s default network and
its adaptive role in internal mentation. Neuroscientist, 18,
251–270.
Benjamini, Y., & Yekutieli, D. (2001). The control of the false
discovery rate in multiple testing under dependency. Annals
of Statistics, 29, 1165–1188.
Broyd, S. J., Demanuele, C., Debener, S., Helps, S. K., James,
C. J., & Sonuga-Barke, E. J. S. (2009). Default-mode brain
dysfunction in mental disorders: A systematic review.
Neuroscience & Biobehavioral Reviews, 33, 279–296.
Buckner, R. L., & Carroll, D. C. (2007). Self-projection and the
brain. Trends in Cognitive Sciences, 11, 49–57.
Buckner, R. L., Sepulcre, J., Talukdar, T., Krienen, F. M., Liu, H.,
Hedden, T., et al. (2009). Cortical hubs revealed by intrinsic
functional connectivity: Mapping, assessment of stability,
and relation to Alzheimer’s disease. Journal of Neuroscience,
29, 1860–1873.
Burgund, E. D., Lugar, H. M., Miezin, F. M., & Petersen, S. E.
(2003). Sustained and transient activity during an object-
naming task: A mixed blocked and event-related fMRI study.
Neuroimage, 19, 29–41.
Calhoun, V. D., Adali, T., Pearlson, G. D., & Pekar, J. J. (2001).
A method for making group inferences from functional
MRI data using independent component analysis. Human
Brain Mapping, 14, 140–151.
Calhoun, V. D., Adalı, T., & Pekar, J. J. (2004). A method for
comparing group fMRI data using independent component
analysis: Application to visual, motor and visuomotor tasks.
Magnetic Resonance Imaging, 22, 1181–1191.
Christoff, K., Gordon, A. M., Smallwood, J., Smith, R., &
Schooler, J. W. (2009). Experience sampling during fMRI
reveals default network and executive system contributions
to mind wandering. Proceedings of the National Academy
of Sciences, U.S.A., 106, 8719–8724.
Elton and Gao
2379
D
o
w
n
l
o
a
d
e
d
f
r
o
m
l
l
/
/
/
/
j
t
t
f
/
i
t
.
:
/
/
h
t
t
p
:
/
D
/
o
m
w
i
n
t
o
p
a
r
d
c
e
.
d
s
f
i
r
o
l
m
v
e
h
r
c
p
h
a
d
i
i
r
r
e
.
c
c
t
.
o
m
m
/
j
e
o
d
u
c
n
o
/
c
a
n
r
a
t
r
i
t
i
c
c
l
e
e
-
p
-
d
p
d
2
f
7
/
1
2
2
7
/
2
1
3
2
6
/
9
2
1
3
9
6
5
9
0
/
0
1
4
8
7
8
o
3
c
7
n
8
_
5
a
/
_
j
0
o
0
c
8
n
5
9
_
a
p
_
d
0
0
b
8
y
5
g
9
u
.
e
p
s
t
d
o
f
n
b
0
y
7
S
M
e
I
p
T
e
m
L
i
b
b
e
r
r
a
2
r
0
2
i
3
e
s
/
j
/
f
t
.
u
s
e
r
o
n
1
7
M
a
y
2
0
2
1
Cole, M. W., Repovs, G., & Anticevic, A. (2014). The
frontoparietal control system: A central role in mental health.
Neuroscientist, 20, 652–664.
Damoiseaux, J. S., Rombouts, S. A. R. B., Barkhof, F., Scheltens,
P., Stam, C. J., Smith, S. M., et al. (2006). Consistent
resting-state networks across healthy subjects. Proceedings
of the National Academy of Sciences, U.S.A., 103,
13848–13853.
Daniels, J. K., McFarlane, A. C., Bluhm, R. L., Moores, K. A.,
Clark, C. R., Shaw, M. E., et al. (2010). Switching between
executive and default mode networks in posttraumatic stress
disorder: Alterations in functional connectivity. Journal of
Psychiatry & Neuroscience, 35, 258.
Di, X., & Biswal, B. B. (2014). Modulatory interactions between
the default mode network and task positive networks in
resting-state. PeerJ, 2, e367.
Elton, A., & Gao, W. (2014). Divergent task-dependent
functional connectivity of executive control and salience
networks. Cortex, 51, 56–66.
Fox, M. D., Snyder, A. Z., Vincent, J. L., Corbetta, M., Van Essen,
D. C., & Raichle, M. E. (2005). The human brain is intrinsically
organized into dynamic, anticorrelated functional networks.
Proceedings of the National Academy of Sciences, U.S.A.,
102, 9673–9678.
Friston, K. J., Holmes, A. P., Worsley, K. J., Poline, J. P., Frith,
C. D., & Frackowiak, R. S. (1994). Statistical parametric maps
in functional imaging: A general linear approach. Human
Brain Mapping, 2, 189–210.
Gao, W., Gilmore, J. H., Alcauter, S., & Lin, W. (2013). The
dynamic reorganization of the default-mode network during
a visual classification task. Frontiers in Systems Neuroscience,
7, 34.
Gao, W., & Lin, W. (2012). Frontal parietal control network
regulates the anti-correlated default and dorsal attention
networks. Human Brain Mapping, 33, 192–202.
Gilbert, S. J., Dumontheil, I., Simons, J. S., Frith, C. D., &
Burgess, P. W. (2007). Comment on “ Wandering minds: The
default network and stimulus-independent thought ”.
Science, 317, 43; author reply 43.
Gilbert, S. J., Simons, J. S., Frith, C. D., & Burgess, P. W. (2006).
Performance-related activity in medial rostral prefrontal
cortex (area 10) during low-demand tasks. Journal of
Experimental Psychology: Human Perception and
Performance, 32, 45–58.
Greicius, M. D., Srivastava, G., Reiss, A. L., & Menon, V. (2004).
Default-mode network activity distinguishes Alzheimer’s
disease from healthy aging: Evidence from functional MRI.
Proceedings of the National Academy of Sciences, U.S.A.,
101, 4637–4642.
Guo, W., Yao, D., Jiang, J., Su, Q., Zhang, Z., Zhang, J., et al.
(2014). Abnormal default-mode network homogeneity in
first-episode, drug-naive schizophrenia at rest. Progress in
Neuro-Psychopharmacology & Biological Psychiatry, 49,
16–20.
Hagmann, P., Cammoun, L., Gigandet, X., Meuli, R., Honey,
C. J., Wedeen, V. J., et al. (2008). Mapping the structural core
of human cerebral cortex. PLoS Biology, 6, e159.
Hahn, B., Ross, T. J., & Stein, E. A. (2007). Cingulate activation
increases dynamically with response speed under stimulus
unpredictability. Cerebral Cortex, 17, 1664–1671.
Hampson, M., Driesen, N. R., Skudlarski, P., Gore, J. C., &
Constable, R. T. (2006). Brain connectivity related to working
memory performance. Journal of Neuroscience, 26,
13338–13343.
Jang, J. H., Jung, W. H., Choi, J. S., Choi, C. H., Kang, D. H.,
Shin, N. Y., et al. (2011). Reduced prefrontal functional
connectivity in the default mode network is related to
greater psychopathology in subjects with high genetic
loading for schizophrenia. Schizophrenia Research, 127,
58–65.
Kelly, A. M., Uddin, L. Q., Biswal, B. B., Castellanos, F. X., &
Milham, M. P. (2008). Competition between functional brain
networks mediates behavioral variability. Neuroimage, 39,
527–537.
Krienen, F. M., Yeo, B. T. T., & Buckner, R. L. (2014).
Reconfigurable task-dependent functional coupling modes
cluster around a core functional architecture. Philosophical
Transactions of the Royal Society, Series B, Biological
Sciences, 369, 20130526.
Liddle, E. B., Hollis, C., Batty, M. J., Groom, M. J., Totman, J. J.,
Liotti, M., et al. (2011). Task-related default mode network
modulation and inhibitory control in ADHD: Effects of
motivation and methylphenidate. Journal of Child
Psychology and Psychiatry, 52, 761–771.
Lynall, M.-E., Bassett, D. S., Kerwin, R., McKenna, P. J.,
Kitzbichler, M., Muller, U., et al. (2010). Functional
connectivity and brain networks in schizophrenia. The
Journal of Neuroscience, 30, 9477–9487.
Lynch, C. J., Uddin, L. Q., Supekar, K., Khouzam, A., Phillips, J.,
& Menon, V. (2013). Default mode network in childhood
autism: Posteromedial cortex heterogeneity and relationship
with social deficits. Biological Psychiatry, 74, 212–219.
Marchetti, I., Koster, E. H., Sonuga-Barke, E. J., & De Raedt, R.
(2012). The default mode network and recurrent depression:
A neurobiological model of cognitive risk factors.
Neuropsychology Review, 22, 229–251.
Mason, M. F., Norton, M. I., Van Horn, J. D., Wegner, D. M.,
Grafton, S. T., & Macrae, C. N. (2007). Wandering minds: The
default network and stimulus-independent thought. Science,
315, 393–395.
Murdaugh, D. L., Shinkareva, S. V., Deshpande, H. R., Wang, J.,
Pennick, M. R., & Kana, R. K. (2012). Differential deactivation
during mentalizing and classification of autism based on
default mode network connectivity. PLoS One, 7, e50064.
Ongur, D., Lundy, M., Greenhouse, I., Shinn, A. K., Menon, V.,
Cohen, B. M., et al. (2010). Default mode network
abnormalities in bipolar disorder and schizophrenia.
Psychiatry Research, 183, 59–68.
Owen, A. M., McMillan, K. M., Laird, A. R., & Bullmore, E.
(2005). n-back working memory paradigm: A meta-analysis of
normative functional neuroimaging studies. Human Brain
Mapping, 25, 46–59.
Phan, K. L., Wager, T., Taylor, S. F., & Liberzon, I. (2002).
Functional neuroanatomy of emotion: A meta-analysis of
emotion activation studies in PET and fMRI. Neuroimage, 16,
331–348.
Pomarol-Clotet, E., Salvador, R., Sarro, S., Gomar, J., Vila, F.,
Martinez, A., et al. (2008). Failure to deactivate in the
prefrontal cortex in schizophrenia: Dysfunction of the
default mode network? Psychological Medicine, 38,
1185–1193.
Qi, Z., Wu, X., Wang, Z., Zhang, N., Dong, H., Yao, L., et al.
(2010). Impairment and compensation coexist in amnestic
MCI default mode network. Neuroimage, 50, 48–55.
Raichle, M. E., MacLeod, A. M., Snyder, A. Z., Powers, W. J.,
Gusnard, D. A., & Shulman, G. L. (2001). A default mode of
brain function. Proceedings of the National Academy of
Sciences, U.S.A., 98, 676–682.
Rubia, K., Russell, T., Overmeyer, S., Brammer, M. J., Bullmore,
E. T., Sharma, T., et al. (2001). Mapping motor inhibition:
Conjunctive brain activations across different versions of
go/no-go and stop tasks. Neuroimage, 13, 250–261.
Rubinov, M., Knock, S. A., Stam, C. J., Micheloyannis, S., Harris,
A. W. F., Williams, L. M., et al. (2009). Small-world properties
of nonlinear brain activity in schizophrenia. Human Brain
Mapping, 30, 403–416.
2380
Journal of Cognitive Neuroscience
Volume 27, Number 12
D
o
w
n
l
o
a
d
e
d
f
r
o
m
l
l
/
/
/
/
j
t
t
f
/
i
t
.
:
/
/
h
t
t
p
:
/
D
/
o
m
w
i
n
t
o
p
a
r
d
c
e
.
d
s
f
i
r
o
l
m
v
e
h
r
c
p
h
a
d
i
i
r
r
e
.
c
c
t
.
o
m
m
/
j
e
o
d
u
c
n
o
/
c
a
n
r
a
t
r
i
t
i
c
c
l
e
e
-
p
-
d
p
d
2
f
7
/
1
2
2
7
/
2
1
3
2
6
/
9
2
1
3
9
6
5
9
0
/
0
1
4
8
7
8
o
3
c
7
n
8
_
5
a
/
_
j
0
o
0
c
8
n
5
9
_
a
p
_
d
0
0
b
8
y
5
g
9
u
.
e
p
s
t
d
o
f
n
b
0
y
7
S
M
e
I
p
T
e
m
L
i
b
b
e
r
r
a
2
r
0
2
i
3
e
s
/
j
/
t
.
f
u
s
e
r
o
n
1
7
M
a
y
2
0
2
1
Sambataro, F., Wolf, N. D., Pennuto, M., Vasic, N., & Wolf, R. C.
(2013). Revisiting default mode network function in major
depression: Evidence for disrupted subsystem connectivity.
Psychological Medicine, 44, 2041–2051.
Seeley, W. W., Menon, V., Schatzberg, A. F., Keller, J., Glover,
G. H., Kenna, H., et al. (2007). Dissociable intrinsic connectivity
networks for salience processing and executive control.
Journal of Neuroscience, 27, 2349–2356.
Sheline, Y. I., Barch, D. M., Price, J. L., Rundle, M. M., Vaishnavi,
S. N., Snyder, A. Z., et al. (2009). The default mode
network and self-referential processes in depression.
Proceedings of the National Academy of Sciences, U.S.A.,
106, 1942–1947.
Shulman, G., Fiez, J., Corbetta, M., Buckner, R., & Miezin, F.
(1997). Common blood flow changes across visual tasks: II.
Decreases in cerebral cortex. Journal of Cognitive
Neuroscience, 9, 648–663.
Smith, S. M., Fox, P. T., Miller, K. L., Glahn, D. C., Fox, P. M.,
Mackay, C. E., et al. (2009). Correspondence of the brain’s
functional architecture during activation and rest.
Proceedings of the National Academy of Sciences, U.S.A.,
106, 13040–13045.
Sonuga-Barke, E. J. S., & Castellanos, F. X. (2007). Spontaneous
attentional fluctuations in impaired states and pathological
conditions: A neurobiological hypothesis. Neuroscience &
Biobehavioral Reviews, 31, 977–986.
Spencer, M. D., Chura, L. R., Holt, R. J., Suckling, J., Calder, A. J.,
Bullmore, E. T., et al. (2012). Failure to deactivate the default
mode network indicates a possible endophenotype of
autism. Molecular Autism, 3, 15.
Spreng, R. N. (2012). The fallacy of a “task-negative” network.
Frontiers in Psychology, 3, 145.
Spreng, R. N., Mar, R. A., & Kim, A. S. (2009). The common
neural basis of autobiographical memory, prospection,
navigation, theory of mind, and the default mode: A
quantitative meta-analysis. Journal of Cognitive
Neuroscience, 21, 489–510.
Spreng, R. N., Stevens, W. D., Chamberlain, J. P., Gilmore, A. W.,
& Schacter, D. L. (2010). Default network activity, coupled
with the frontoparietal control network, supports goal-
directed cognition. Neuroimage, 53, 303–317.
Summerfield, J. J., Hassabis, D., & Maguire, E. A. (2009).
Cortical midline involvement in autobiographical memory.
Neuroimage, 44, 1188–1200.
Tessitore, A., Esposito, F., Vitale, C., Santangelo, G., Amboni,
M., Russo, A., et al. (2012). Default-mode network
connectivity in cognitively unimpaired patients with
Parkinson disease. Neurology, 79, 2226–2232.
Uddin, L. Q., Clare Kelly, A. M., Biswal, B. B., Castellanos, F. X.,
& Milham, M. P. (2009). Functional connectivity of default
mode network components: Correlation, anticorrelation, and
causality. Human Brain Mapping, 30, 625–637.
Uddin, L. Q., Kelly, A. M., Biswal, B. B., Margulies, D. S.,
Shehzad, Z., Shaw, D., et al. (2008). Network homogeneity
reveals decreased integrity of default-mode network in
ADHD. Journal of Neuroscience Methods, 169, 249–254.
van den Heuvel, M. P., & Sporns, O. (2013). Network hubs
in the human brain. Trends in Cognitive Sciences, 17,
683–696.
Van Dijk, K. R., Hedden, T., Venkataraman, A., Evans, K. C.,
Lazar, S. W., & Buckner, R. L. (2010). Intrinsic functional
connectivity as a tool for human connectomics: Theory,
properties, and optimization. Journal of Neurophysiology,
103, 297–321.
Vincent, J. L., Kahn, I., Snyder, A. Z., Raichle, M. E., &
Buckner, R. L. (2008). Evidence for a frontoparietal
control system revealed by intrinsic functional connectivity.
Journal of Neurophysiology, 100, 3328–3342.
Wager, T. D., Sylvester, C.-Y. C., Lacey, S. C., Nee, D. E.,
Franklin, M., & Jonides, J. (2005). Common and unique
components of response inhibition revealed by fMRI.
Neuroimage, 27, 323–340.
Wagner, A. D., Shannon, B. J., Kahn, I., & Buckner, R. L. (2005).
Parietal lobe contributions to episodic memory retrieval.
Trends in Cognitive Sciences, 9, 445–453.
Washington, S. D., Gordon, E. M., Brar, J., Warburton, S.,
Sawyer, A. T., Wolfe, A., et al. (2013). Dysmaturation
of the default mode network in autism. Human
Brain Mapping, 35, 1284–1296.
Weissman, D. H., Roberts, K. C., Visscher, K. M., & Woldorff,
M. G. (2006). The neural bases of momentary lapses in
attention. Nature Neuroscience, 9, 971–978.
D
o
w
n
l
o
a
d
e
d
f
r
o
m
l
l
/
/
/
/
j
f
/
t
t
i
t
.
:
/
/
h
t
t
p
:
/
D
/
o
m
w
i
n
t
o
p
a
r
d
c
e
.
d
s
f
i
r
o
l
m
v
e
h
r
c
p
h
a
d
i
i
r
r
e
.
c
c
t
.
o
m
m
/
j
e
o
d
u
c
n
o
/
c
a
n
r
a
t
r
i
t
i
c
c
l
e
e
-
p
-
d
p
d
2
f
7
/
1
2
2
7
/
2
1
3
2
6
/
9
2
1
3
9
6
5
9
0
/
0
1
4
8
7
8
o
3
c
7
n
8
_
5
a
/
_
j
0
o
0
c
8
n
5
9
_
a
p
_
d
0
0
b
8
y
5
g
9
u
.
e
p
s
t
d
o
f
n
b
0
y
7
S
M
e
I
p
T
e
m
L
i
b
b
e
r
r
a
2
r
0
2
i
3
e
s
/
j
.
/
t
f
u
s
e
r
o
n
1
7
M
a
y
2
0
2
1
Elton and Gao
2381