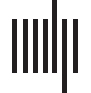ARTICLE DE RECHERCHE
Behavioral and Neural Correlates of Speech Motor
Sequence Learning in Stuttering and Neurotypical
Speakers: An fMRI Investigation
un accès ouvert
journal
Alfonso Nieto-Castañón1, Matthias Heyne1, Saul A. Frankford1
Matthew Masapollo1,2, Jennifer A. Segawa1,3
, Deryk S. Beal1,4
, Jason A. Tourville1
, and Frank H. Guenther1,3,5,6
,
1Department of Speech, Language, and Hearing Sciences, Université de Boston, Boston, MA, Etats-Unis
2Department of Speech, Language, and Hearing Sciences, University of Florida, Gainesville, FL, Etats-Unis
3Departments of Neuroscience and Biology, Stonehill College, Easton, MA, Etats-Unis
4Department of Speech-Language Pathology, University of Toronto, Toronto, Canada
5Department of Biomedical Engineering, Université de Boston, Boston, MA, Etats-Unis
6Picower Institute for Learning and Memory, Massachusetts Institute of Technology, Cambridge, MA, Etats-Unis
Mots clés: speech motor control, motor sequence learning, stuttering, GODIVA model, IRMf
ABSTRAIT
Stuttering is a neurodevelopmental disorder characterized by impaired production of coordinated
articulatory movements needed for fluent speech. It is currently unknown whether these abnormal
production characteristics reflect disruptions to brain mechanisms underlying the acquisition
and/or execution of speech motor sequences. To dissociate learning and control processes, nous
used a motor sequence learning paradigm to examine the behavioral and neural correlates of
learning to produce novel phoneme sequences in adults who stutter (AWS) and neurotypical
controls. Participants intensively practiced producing pseudowords containing non-native
consonant clusters (par exemple., “GVAZF”) over two days. The behavioral results indicated that although the
two experimental groups showed comparable learning trajectories, AWS performed significantly
worse on the task prior to and after speech motor practice. Using functional magnetic resonance
imaging (IRMf), the authors compared brain activity during articulation of the practiced words
and a set of novel pseudowords (matched in phonetic complexity). FMRI analyses revealed no
differences between AWS and controls in cortical or subcortical regions; both groups showed
comparable increases in activation in left-lateralized brain areas implicated in phonological
working memory and speech motor planning during production of the novel sequences compared
to the practiced sequences. De plus, activation in left-lateralized basal ganglia sites was negatively
correlated with in-scanner mean disfluency in AWS. Collectively, these findings demonstrate
that AWS exhibit no deficit in constructing new speech motor sequences but do show impaired
execution of these sequences before and after they have been acquired and consolidated.
INTRODUCTION
Stuttering is a neurodevelopmental disorder affecting children into adulthood with devastating
social effects that impede speech communication. The presenting clinical signs of the disorder
include involuntary repetitions and prolongations of phonemes, syllables, or words, ainsi que
involuntary silent pauses (Bloodstein & Ratner, 2008). Despite considerable research, chercheurs
still have a relatively poor understanding of the neural deficits underlying the disorder (see Craig-
McQuaide et al., 2014, for a review). Neurobiological accounts of stuttering, such as Alm (2004),
Citation: Masapollo, M., Segawa, J.. UN., Beal,
D. S., Tourville, J.. UN., Nieto-Castañón, UN.,
Heyne, M., Frankford, S. UN., & Guenther, F. H.
(2021). Behavioral and neural correlates
of speech motor sequence learning in
stuttering and neurotypical speakers:
An fMRI investigation. Neurobiology of
Language, 2(1), 106–137. https://est ce que je
.org/10.1162/nol_a_00027
EST CE QUE JE:
https://doi.org/10.1162/nol_a_00027
Informations complémentaires:
https://doi.org/10.1162/nol_a_00027
Reçu: 5 Octobre 2020
Accepté: 11 Novembre 2020
Intérêts concurrents: Les auteurs ont
a déclaré qu'aucun intérêt concurrent
exister.
Auteur correspondant:
Matthew Masapollo
mmasapollo@phhp.ufl.edu
Éditeur de manipulation:
Kate Watkins
droits d'auteur: © 2021 Massachusetts
Institut de technologie. Published
under a Creative Commons Attribution
4.0 International (CC PAR 4.0) Licence.
La presse du MIT
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
e
d
toi
n
o
/
je
/
je
un
r
t
je
c
e
–
p
d
F
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
un
_
0
0
0
2
7
p
d
.
/
je
F
b
oui
g
toi
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
Max et al. (2004), and recently Connally et al. (2018) and Chang and Guenther (2020), proposed
that stuttering disfluencies arise from an underlying defect in the (left-lateralized) cortico-basal-
ganglia-thalamocortical (cortico-BG) loops (Alexander et al., 1986), which are hypothesized to
be responsible for selecting and triggering articulatory motor programs, while suppressing others,
at the appropriate time during speech sequencing.
There is an extensive body of evidence (recently reviewed by Chang & Guenther, 2020) dans
favor of the idea that stuttering involves an inability to initiate, sustain, and/or terminate speech
motor programs due to anomalous basal ganglia function. Par exemple, numerous acoustic
investigations have provided evidence that stuttering involves an impaired ability to transition from
the articulatory gestures affiliated with an initial phoneme of a syllable to subsequent gestures, comme
indicated by protracted voice onset times and formant transition rates (par exemple., Boutsen, 1993; Chang
et coll., 2002; Robb & Blomgren, 1997; Robb et al., 1998; Yaruss & Conture, 1993). As for the
underlying neural circuitry, abnormal functional activity in the basal ganglia has frequently been
implicated in stuttering severity (Connally et al., 2018; Giraud et al., 2008; Ingham et al., 2012;
Toyomura et al., 2015). En outre, stuttering-like disfluencies have been reported to emerge in
neurodegenerative diseases that impair the function of the basal ganglia, such as Parkinson’s
maladie (Niethammer et al., 2012), and deep brain stimulation applied to either the subthalamic
nucleus or the globus pallidus pars interna of the basal ganglia has been shown to relieve or
exacerbate stuttering disfluencies (see Sapir, 2014, and Skodda, 2012, for reviews).
De plus, and of particular relevance to the current research, some existing theoretical
models attribute a role in speech motor sequence learning to the cortico-BG loops (par exemple.,
Guenther, 2016, Chapters 8 & 10; Kotz & Schwartze, 2010; see also Fee & Goldberg, 2011,
for a similar account in songbird vocal learning). Par exemple, in the Gradient Order Directions
Into Velocities of the Articulators (GODIVA) model of speech sequencing and initiation (Bohland
et coll., 2010; Civier et al., 2013; Guenther, 2016, Chapter 8; Segawa, Masapollo, et coll., 2019), it
is proposed that continual speech motor practice leads to the formation of optimized motor
programs for frequently occurring kinematic sequences, such as the successive gestures making
up a word or syllable (see also, Cholin et al. 2006). These learned movement “chunks” or
“templates” are then encoded and sequentially activated (or “read out”) by the cortico-BG loops
during production. The fundamental idea is that the cortico-BG loops perform two distinct, mais
related, cognitive operations during the speech production process, namely, forming regularized
motor chunks and initiating/activating motor programs affiliated with those chunks.
Results from a functional magnetic resonance imaging (IRMf) study of speech motor sequence
learning with neurotypical adults (Segawa, Tourville, et coll., 2015) provided empirical support for
this view. In the initial training phase of the study, participants performed a cued motor sequence
learning task (outside the scanner) in which they intensively practiced producing pseudowords
(monosyllabic CCVCC[C], where C = consonant, V = vowel) consisting of consonant clusters that
were either phonotactically legal in their native language of English (par exemple., “BLERK”) or illegal in
English but legal in other natural human languages (par exemple., “GVAZF”). The results indicated that,
behaviorally, motor practice led to measurable performance gains (c'est à dire., reduced error rates and
shorter utterance durations), presumably because the sequences of independent speech move-
ments had become concatenated into larger cohesive chunks (see Segawa, Masapollo, et coll.,
2019, for supporting evidence).
In the subsequent test phase (inside the scanner), the authors measured the blood oxygen-level
dependent (AUDACIEUX) response during production of the practiced native and practiced non-native
words as well as a novel set of non-native words (matched in phonetic complexity) using sparse
sampling fMRI (voir, par exemple., Gracco et al., 2005). In one set of imaging analyses, BOLD activity
Basal ganglia:
Set of nuclei found underneath the
cortex in the medial position of
the brain.
Motor sequence learning:
Learning how to plan and execute an
appropriate order of discrete actions.
Motor chunks:
Cohesive action units composed of
frequently occurring subsequences
of movements.
Functional magnetic resonance
imaging (IRMf):
Imaging that measures changes in
blood oxygenation levels that occur
in response to neural firing, allowing
precise localization of brain activity.
Consonant clusters:
Consonant sequences at the start or
end of a syllable.
Neurobiology of Language
107
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
e
d
toi
n
o
/
je
/
je
un
r
t
je
c
e
–
p
d
F
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
un
_
0
0
0
2
7
p
d
.
/
je
F
b
oui
g
toi
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
Anterior insula (aINS):
Brain region that plays a role in speech
motor sequence learning.
associated with production of the novel non-native words was contrasted with that of the practiced
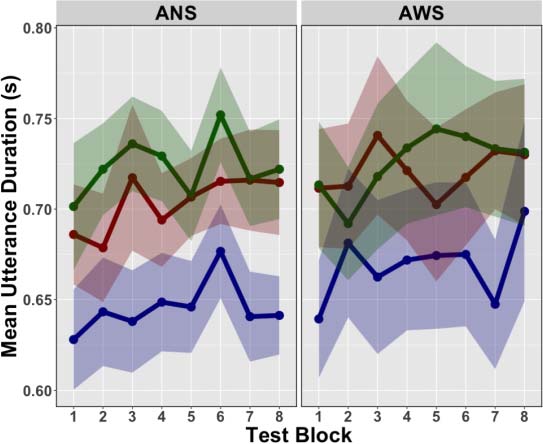
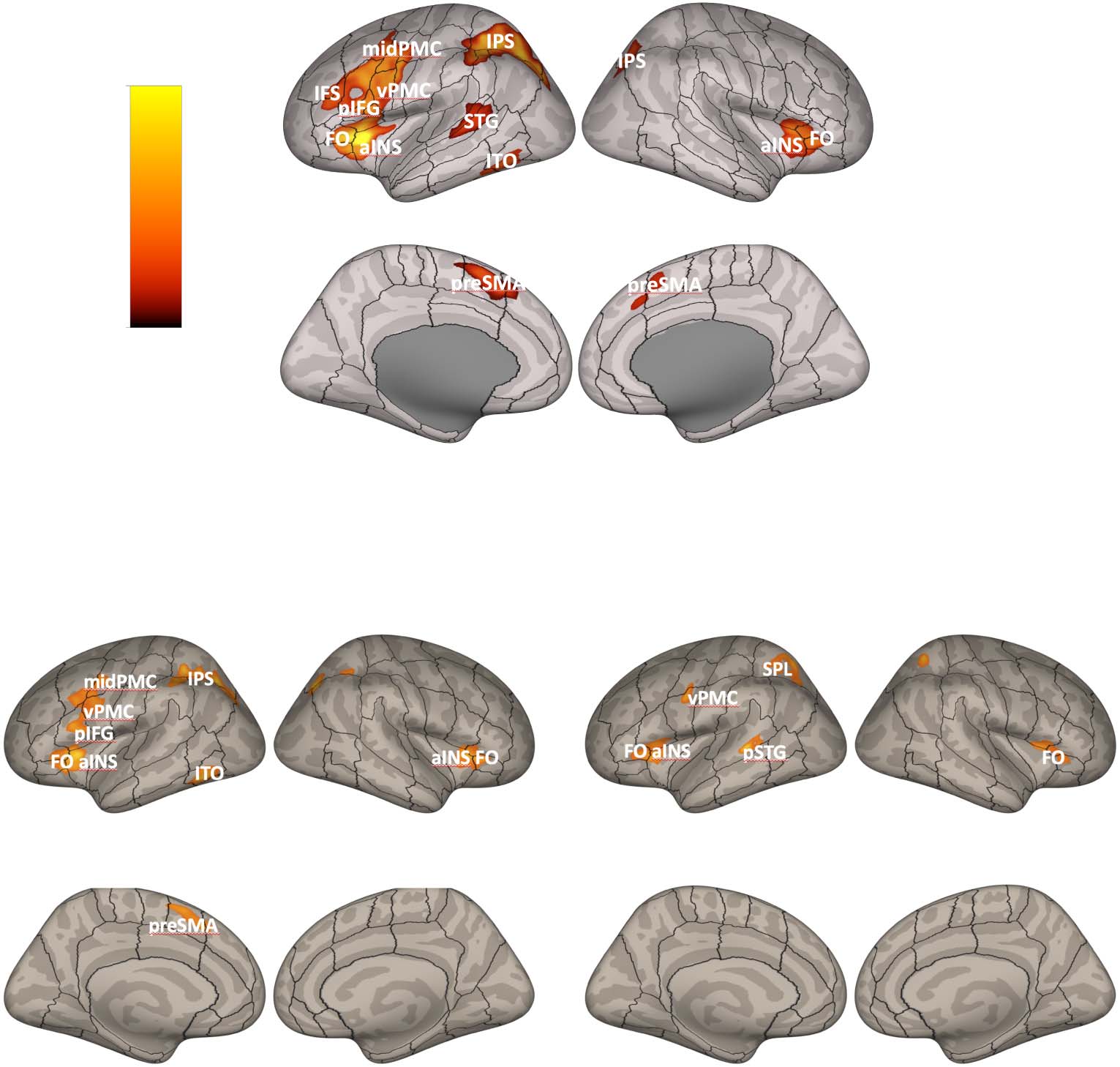
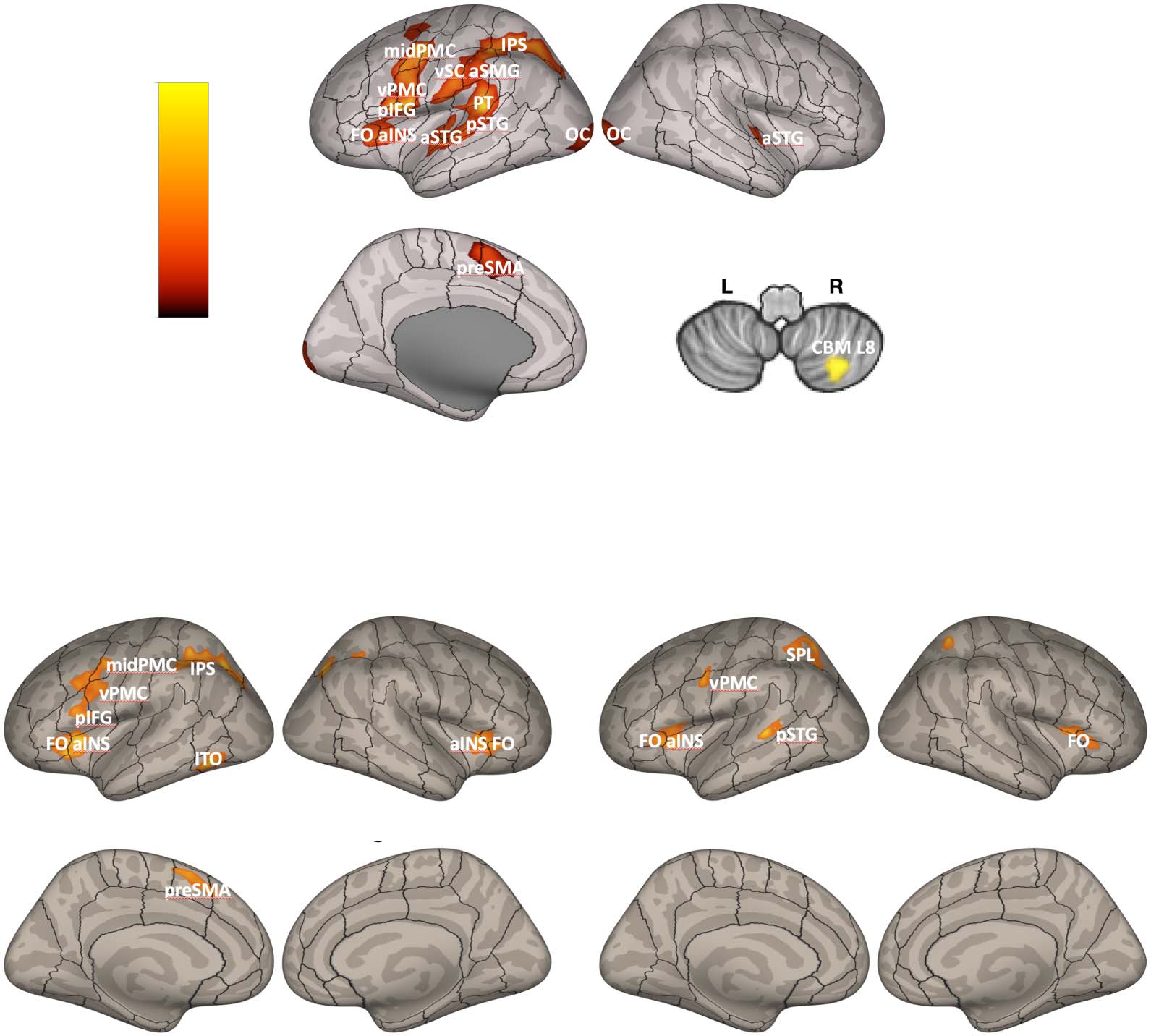
non-native words. The results demonstrated increased activation in (left-lateralized) cortical (c'est à dire.,
pre-supplementary motor area [preSMA], anterior insula [aINS], inferior frontal sulcus [IFS], inferior
parietal sulcus [IPS], ventral premotor cortex [vPMC]), and subcortical basal ganglia (c'est à dire., globus
pallidus [GP]) regions involved in the cortico-BG loops (Alexander et al., 1986). It was further dem-
onstrated that the degree of motor performance improvements between training and test was cor-
related positively with activity in the left aINS. The cortical regions that were found to be recruited in
this task overlap with activation foci that have been identified in meta-analyses of working memory
neuroimaging studies (voir, par exemple., Rottschy et al., 2012). Ainsi, these findings are in keeping with the
hypothesis that the cortico-BG loops play an important role in learning and sequencing speech
movement chunks since a higher number of chunks will have to be concatenated in a working
memory repository (or phonological output store) and then subsequently read out during the pro-
duction of novel sequences compared to practiced ones.
In another set of analyses contrasting BOLD activity associated with production of the prac-
ticed non-native and practiced native words, the authors found increased activation in a similar
network of brain areas, although notably no increased activation was found in the left GP for this
contraste. They did, cependant, report increased activation in the right cerebellum (lobule VI) for this
contraste, suggesting that other subcortical structures that extend beyond the cortico-BG may also
play a role in speech motor sequencing learning and motor control (a point we will return to in
the general discussion).
Nevertheless, if the cortico-BG loops are involved in successively encoding and activating
speech motor chunks during sequence learning and production, then that raises the possibility that
the neural deficits hypothesized to underlie stuttering are related not only to problems with motor
execution, but also to impaired motor learning mechanisms. Consistent with this view are experi-
mental findings, in the behavioral literature, that adults who stutter (AWS) and adults with normal
speech (ANS) often show differences in their ability to learn a variety of novel speech and non-
speech (c'est à dire., finger tapping) movement sequences (par exemple., Ludlow et al., 1997; Namasivayam &
van Lieshout, 2008; Smits-Bandstra et al., 2006). Qualitatively similar findings have also been
found in patients with Parkinson’s disease (Ferraro et al., 1993; Helmuth et al., 2000; Jackson
et coll., 1995; Mollaei et al., 2013; Smits-Bandstra & Gracco, 2013; Vakil et al., 2000).
The Current Study
In the current research, we performed a functional brain-imaging study to distinguish between
motor execution and motor learning impairments in AWS at both the behavioral and neural
levels, by replicating and extending Segawa, Tourville, et autres. (2015) with a cohort of adult
speakers with persistent developmental stuttering. Toward this end, we trained AWS and ANS
to produce pseudowords containing non-native consonant clusters. Participants were first trained
over several days (outside the scanner) to produce two sets of novel CCVCC pseudowords: (1)
syllables that involved native consonant clusters ( practiced native) et (2) syllables consisting of
non-native consonant clusters that are phonotactically illegal in English ( practiced non-native).
Based on previous results (Segawa, Tourville, et coll., 2015; see also Segawa, Masapollo, et coll.,
2019), we expected to observe significantly larger performance gains throughout the course of
training for the practiced non-native words than for the practiced native words (for which perfor-
mance is already expected to be near ceiling at the beginning of training). During a subsequent
testing phase (inside the scanner), we then contrasted BOLD activity associated with the produc-
tion of the practiced non-native and practiced native words, and the practiced non-native and
practiced native sequences.
Neurobiology of Language
108
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
e
d
toi
n
o
/
je
/
je
un
r
t
je
c
e
–
p
d
F
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
un
_
0
0
0
2
7
p
d
/
.
je
F
b
oui
g
toi
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
We hypothesized that, if motor sequence learning mechanisms per se are impaired in stuttering,
alors: (1) At the behavioral level, AWS should show a slower rate of learning over the course of
training and/or poorer learning outcomes compared to ANS; et (2) At the neural level, contrasting
the BOLD signal for the novel non-native–practiced non-native and/or practiced non-native–
practiced native conditions should yield less activity in the regions of the cortico-BG circuit pre-
viously identified in Segawa, Tourville, et autres. (2015) in AWS, especially the left aINS, since they
will not have fully formed cohesive motor chunks for the trained speech sequences.
If, cependant, the only core deficit underlying stuttering is related to motor implementation
rather than to motor learning mechanisms, alors: (1) At the behavioral level, AWS should show
comparable performance gains over time but will display poorer (and/or slower) overall motor
performance compared to ANS; et (2) At the neural level, AWS should show a similar reduction
in activations in the aforementioned regions of the cortico-BG circuit implicated in motor
sequences learning and working memory (the left aINS, preSMA, and IFS) as ANS, but should still
show differences in primary motor (and possibly premotor) cortex; et (3) The size of the BOLD
activity difference in (at least some of ) these brain areas will covary with stuttering severity (and/or
in-scanner disfluency) since mechanisms involved in selecting and initiating successive speech
motor programs will be more impaired in AWS with more severe stuttering.
MATERIALS AND METHODS
Participants
Sixteen AWS (14 male, age range 18–42, median age 26) and fifteen ANS (13 male, age range
18–40, median age 25) were paid for participating in three testing sessions on three separate days.
Participants reported normal (or corrected-to-normal) vision and no history of hearing, speech,
langue, or neurological deficits (apart from stuttering in the AWS group). All were right-handed
(Oldfield, 1971; mean score = 80.46) and native speakers of American English with no previous
experience with any of the languages used in stimulus creation (see following text). Individuals
were excluded from taking part in the study if they were currently on medications that may
have substantial effects on neural activity, or if they had claustrophobia preventing them from
completing the MRI protocol. All participants underwent a magnetic resonance safety
screening.
An experimenter interviewed all participants to confirm the diagnosis of persistent develop-
mental stuttering in AWS and to confirm normal speech production in ANS. None of the AWS
were enrolled in a fluency-shaping program at the time of participation. The stuttering severity of
each AWS was assessed using the Stuttering Severity Instrument, Edition 4 (SSI-4; Riley, 2009). Comme
part of this assessment, each AWS was video recorded while reading aloud, conversing with an
experimenter, and speaking on the telephone. A certified speech-language pathologist then
rated the frequency and duration of the stuttering events and the presence of physical concomitants
that accompanied the moments of disfluency (par exemple., eye-blinking). Stuttering severity in the AWS
group ranged from 13 à 48, with a median of 27, and an interquartile range of 17 à 34. Five
of the 16 AWS were categorized as “very mild” (SSI-4 score <17), 1 as “mild” (18–24), 5 as
“moderate” (25–31), 3 as “severe” (32–36), and 2 as “very severe” (37–46).
Stimuli
The speech stimuli consisted of several sets of monosyllabic pseudoword sequences (15 words
per set). As shown in Table 1, all items contained either native or non-native syllable-initial (onset)
and syllable-final (coda) consonant clusters. In the native sequences (e.g., “BLERK,” “THRIMF,”
“TRALP”), the onset and coda clusters are phonotactically legal in English; in the non-native
Neurobiology of Language
109
l
D
o
w
n
o
a
d
e
d
f
r
o
m
h
t
t
p
:
/
/
d
i
r
e
c
t
.
m
i
t
.
e
d
u
n
o
/
l
/
l
a
r
t
i
c
e
-
p
d
f
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
a
_
0
0
0
2
7
p
d
/
.
l
f
b
y
g
u
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
Table 1.
stimuli used to elicit the native (left) and non-native (right) target onset and coda clusters
International phonetic alphabet (IPA) transcription and orthography for experimental
IPA
blɚɹk
bɹalk
dɹalf
flIsk
fɹʌmp
glæntʃ
gɹalv
klεlθ
kɹeInθ
plaɹθ
pɹʌndʒ
ʃ ɹidθ
tɹælp
θɹImf
dwIlm
kwanst
gwεfθ
twɚɹv
θwIlb
splɚɹst
spɹIdθ
swarf
skεln
stIsp
Native
Orthography
BLERK
BRALK
DRALF
FLISK
FREMP
GLANCH
GRALVE
KLELTH
KRENGTH
PLARTH
PRENGE
SHRIDTH
TRALP
THRIMF
DWILM
KWANST
GWEFTH
TWERVE
THWILB
SPLERST
SPRIDTH
SWARF
SKELN
STISP
Phonotactics
Non-native
IPA
fsεfk
fʃ Ikp
fθæmtʃ
fzItʃ b
vsεpʃ
vðæʃ p
zvεktʃ
fpεstʃ
ftεbstʃ
ʃ kεvt
ʃ tæzg
vbImk
vgæmʃ
zbæpk
zdεbg
bvImpf
bzInstʃ
gvæzf
kvætʃ k
tfIpʃ tʃ
tvItp
bdeŋt
dkεdv
gbεsb
kpεʃ tʃ
ptætʃst
tbæstf
tgItk
tpIpf
zgεkf
Orthography
FSEFK
FSHIKP
FTHAMCH
FZICHB
VSEPSH
VTHASHP
ZVEKCH
FPESCH
FTEBSCH
SHKEVT
SHTAZG
VBIMK
VGAMSH
ZBAPK
ZDEBG
BVIMPF
BZINSCH
GVAZF
KVACHK
TFIPSHCH
TVITP
BDANGT
DKEDV
GBESB
KPESHCH
PTACHST
TBASTF
TGITK
TPIPF
ZGEKF
110
Neurobiology of Language
l
D
o
w
n
o
a
d
e
d
f
r
o
m
h
t
t
p
:
/
/
d
i
r
e
c
t
.
m
i
t
.
e
d
u
n
o
/
l
/
l
a
r
t
i
c
e
-
p
d
f
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
a
_
0
0
0
2
7
p
d
/
.
l
f
b
y
g
u
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
sequences (e.g., “FPESCH,” “GVAZF,” “TPIPF”), the clusters are phonotactically illegal in
English, but do occur in some other natural human language. The non-native clusters were taken
from a variety of languages and language families including Hebrew, Leti, Taba, Romani, Polish,
Lithuanian, Romanian, Georgian, Tepehua, Hungarian, and Pima; participants reported no prior
experience with any languages in which these consonant clusters readily occur. None of the
stimuli were an orthographic or a phonological word according to the MRC Psycholinguistic
Database (Coltheart, 1981). All items containing non-native clusters had a neighborhood size
of 0, and no real English words could be created by adding, deleting, or substituting a single
phoneme in any subsequence. All of the clusters—both native and non-native alike—were
bi- or triconsonantal and used in either onset position or coda position, but not both. Each
cluster was used in only one word (i.e., no two words contained the same consonant cluster).
The number of phonemes per word was counterbalanced across experimental conditions (see
below).
To create the prompts for the elicited production task, a female native speaker of American
English was recorded producing the words. The model speaker was phonetically trained and had
previously practiced producing the sequences until each stimulus could be executed fluently
(i.e., without vocoid epenthesis or phoneme omissions, swaps, or substitutions). All recordings
took place in a sound-attenuated booth. The speech was recorded directly to a computer using
Audacity® software ( Version 2.0.3, Audacity Team) via a microphone (Samson C01U studio
condenser) connected to a pre-amplifier (44.1-kHz sampling rate, 32-bit quantization). The
speaker recorded multiple randomized repetitions of each token. From these repetitions, one
instance of each token was selected on the basis of clarity and acoustic similarity in voice pitch
(f0) to the other stimuli in the set. Using Praat software (Boersma & Weenink, 2020), all recorded
tokens were digitally edited to be matched for peak intensity and duration (i.e., 480 ms) without
changing f0.
Procedure and Design
Participants completed a cued sequence production task (see, e.g., Segawa, Masapollo, et al.,
2019; Segawa, Tourville, et al., 2015). The experiment consisted of a training phase, during
which participants learned to produce 15 words containing native clusters and 15 words con-
taining non-native clusters, followed by a test phase, during which participants were tested on
their ability to produce all 30 of the previously learned words and 15 novel words containing
non-native consonant clusters. FMRI data were only collected during the test phase (see below).
For both phases, participants were asked to repeat aloud each of the target words individually,
which were presented both auditorily over noise-cancelling headphones and visually using text
stimuli (as shown in Table 1). The training phase consisted of eight blocks of trials over two
consecutive days (four on day one and four on day two). Each training block contained eight
repetitions of each word, for a total of 240 trials per block. The test phase consisted of eight
blocks of trials performed on day three (inside the MRI scanner) after completing the training
phase (outside the scanner on days one and two). Each test block contained five to six repetitions
of each word, for an average of 40 trials per block, from three stimulus categories (conditions):
(1) practiced native words (i.e., words comprising native clusters that were previously encoun-
tered in the training phase), (2) practiced non-native words (i.e., words comprising non-native
clusters that were previously encountered in the training phase), and (3) novel non-native words
(i.e., novel words with novel clusters that were not encountered in the training phase).
Participants were divided into four groups, each of which practiced producing a different
subset of the native and non-native sequences during training. The non-native words that were
not practiced during training were used as novel non-native words during the imaging session.
Neurobiology of Language
111
l
D
o
w
n
o
a
d
e
d
f
r
o
m
h
t
t
p
:
/
/
d
i
r
e
c
t
.
m
i
t
.
e
d
u
n
o
/
l
/
l
a
r
t
i
c
e
-
p
d
f
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
a
_
0
0
0
2
7
p
d
.
/
l
f
b
y
g
u
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
Assignment of non-native words to the practiced non-native and novel non-native categories
was counterbalanced across participants. The training phase occurred one to two days before
the test phase to allow for sleep-mediated memory consolidation of the newly acquired motor
traces (see, e.g., Doyon, Albouy, et al., 2015; Doyon, Bellec, et al., 2009; Fenn et al., 2003;
Vahdat et al., 2017; cf. Brawn et al., 2010; Pan & Rickard, 2015).
During training, participants were seated in a chair in front of a laptop (IBM Lenovo ThinkPad
X61s) computer screen in a sound-treated laboratory room that was dimly lit. The auditory speech
stimuli were presented over headphones (Sennheiser, HD 280 Pro) at a comfortable listening
level, and utterances produced by the participants were recorded with a Samson (Hauppauge,
NY) C01U USB studio condenser microphone connected to the computer via a MOTU micro-
book audio interface. Utterances were recorded using MATLAB (MathWorks Inc., Natick, MA) at
44.1 kHz. During testing, participants laid supine in an MRI scanner. Instructions and visual stim-
uli were projected onto a screen viewed from within the scanner via a mirror attached to the head
coil. The auditory stimuli were played over Sensimetrics model S-14 MRI-compatible earphones.
Participants’ productions were transduced by a Fibersound model FOM1-MR-30m fiber-optic
microphone, sent to a Lenovo ThinkPad X61s, and recorded using MATLAB at 44.1 kHz.
The trial structure was identical during training and testing. First, the orthographic display of a
given syllable was centrally presented in tandem with its corresponding auditory prompt.
Participants heard each prompt only once on each trial. Then, after the offset of the auditory
presentation, a tone was presented for 50 ms. The time between stimulus offset and tone onset
was randomly jittered between 500 and 1,000 ms. This tone served as a “go” signal that prompted
the participant to go ahead and repeat the token as clearly and accurately as possible. For all
phases, participants produced the target syllables in a pseudo-random order. The combination
of the auditory and orthographic presentations was necessary because prior studies have shown
that listeners tend to perceive non-native consonant clusters as epenthesized disyllabic
sequences (e.g., Berent et al., 2007; Davidson & Shaw, 2012; Dupoux, Kakehi, et al., 1999;
Dupoux, Parlato, et al., 2011; Pitt, 1998). Moreover, in another study that directly examined
the effects of stimulus input modality (audio only vs. audio and text) on speakers’ ability to pro-
duce non-native consonant clusters (Davidson, 2010), it was found that the presence of text led to
an improvement in overall task performance.
For both phases, participants were instructed to repeat the target syllable as clearly and accu-
rately as possible as soon as they heard the tone. Several familiarization trials with experimenter
feedback were included at the start of the experiment to confirm that participants understood the
task instructions and were able to perform the task. The sequences used during these initial prac-
tice trials were not used at any point in the rest of the study. Feedback was not provided during
the actual training or test phases.
Behavioral Data Analyses
We conducted several analyses to provide evidence, at the behavioral level, that incremental
motor learning occurred throughout the two days of speech motor practice (outside the MRI
scanner), and that such learning was retained at test (inside the scanner). In the first analysis,
we analyzed three temporally sensitive measures of learning across the eight training blocks
(on days 1 and 2): (1) articulatory sequencing error rate, (2) utterance duration (interval from
utterance onset to offset), and (3) reaction time (RT) (interval from the offset of the go-signal to
utterance onset). These measures are generally believed to quantify the ease or difficulty with
which speakers retrieve and execute speech sequences (Sternberg et al., 1978) and are com-
monly used in both the motor sequence learning and second language learning literatures as
Neurobiology of Language
112
l
D
o
w
n
o
a
d
e
d
f
r
o
m
h
t
t
p
:
/
/
d
i
r
e
c
t
.
m
i
t
.
e
d
u
n
o
/
l
/
l
a
r
t
i
c
e
-
p
d
f
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
a
_
0
0
0
2
7
p
d
/
.
l
f
b
y
g
u
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
measures of learning extent (see, e.g., Brawn et al., 2010; Buchwald et al., 2019; Doyon, Albouy,
et al., 2015; Nakamura et al., 1998; Rauschecker et al., 2008; Segawa, Masapollo, et al., 2019).
Sequencing errors were defined as phoneme additions (including schwa insertions), deletions,
and substitutions, and utterance repetitions, omissions, and restarts. Based on prior findings
reported with neurotypical speakers (Segawa, Masapollo, et al., 2019; Segawa, Tourville,
et al., 2015), we expected to observe greater learning (at least in the ANS group) for the non-native
sequences because those sequences included both novel words and novel onset and coda clusters,
whereas the native sequences included novel words with familiar onset and coda clusters.
In the second analysis, to provide evidence of the retention of learning at test, we examined the
same three performance measures for each experimental group (ANS vs. AWS) and condition
(practiced native vs. practiced non-native vs. novel non-native) during the test phase (on day 3).
For each word production, each measure was calculated following the removal of noise associ-
ated with the scanner bore echo and peripheral equipment using a Wiener filter (Wiener, 1949).
The coder was blind to the condition (practiced vs. novel) of the non-native syllables. We hypoth-
esized that if participants formed regularized motor chunks for the previously trained speech
sequences, then they should perform more accurately (and/or faster) at generating the practiced
native and non-native sequences compared to the novel non-native sequences.
In a final analysis, we examined whether, and if so, how, speech motor practice influenced
fluency in the AWS group. Toward this end, we compared the mean number of trials perceptually
coded as containing at least one stuttering-like disfluency during both the training and test phases
(for the AWS group only) for each stimulus condition ( practiced native vs. practiced non-native
vs. novel non-native). Disfluencies were determined by a certified speech-language pathologist.
For each of these analyses, custom MATLAB software was used to perceptually rate and acous-
tically measure onsets and offsets of syllables by viewing the waveform and spectrogram and
listening to the audio files. Listeners first marked, blinded to experimental condition, whether
the target phoneme sequence was produced fluently. Disfluent productions were further catego-
rized into four possible disfluency types: (1) repetition (i.e., a phoneme or syllable was repeated),
(2) prolongation (i.e., a phoneme was prolonged), (3) audible or silent blocking (involuntary filled
or unfilled pauses in speaking), and (4) clustered (i.e., more than one stutter type was produced for
a given sequence).
Each utterance was then marked for four possible sequencing error subtypes: (1) approxima-
tion of the target (i.e., phoneme deletion/omission, insertion, substitution, vowel epenthesis); (2)
unrecognizable from the target (i.e., an entirely different sequence was produced); (3) unfinished
word (i.e., the sequence produced was not completed before the end of the recording window);
and (4) silence (i.e., no sequence was produced at the time of recording). Since we were con-
cerned with the learning of non-native phonotactics rather than subphonemic allophonic details,
productions from either the model speaker or the participants were not judged on how natural
they sounded in the language from which they were derived.
In addition, it is important to note that it was impossible to reliably distinguish involuntary
“silent” pauses or blocks associated with stuttering from intentional speech onset delays with
the audio-only recordings that we obtained in the scanner (although there were constraints on
what the delay could be since the length of the recording window was fairly short). Thus, we
suspect that trials with stuttering pauses were categorized as sequencing error subtype 4 (i.e.,
no sequence produced). This is a limitation inherent in the current study and all studies of this kind
in stuttering (unless video recordings of articulatory behavior inside the scanner are obtained). In
order to explore the possibility that some trials containing “silent” stuttering blocks were included
in the neuroimaging analyses, we plotted histograms showing the distribution of RT scores for
Neurobiology of Language
113
l
D
o
w
n
o
a
d
e
d
f
r
o
m
h
t
t
p
:
/
/
d
i
r
e
c
t
.
m
i
t
.
e
d
u
n
o
/
l
/
l
a
r
t
i
c
e
-
p
d
f
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
a
_
0
0
0
2
7
p
d
/
.
l
f
b
y
g
u
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
each stuttering speaker in the test phase (inside the scanner). Critically, these plots did not reveal
a bimodal distribution with one peak having frequency values clustered to the right. This finding
suggests that any potential blocks or pauses were likely categorized as silent errors (sequencing
subtype 4 as described above) and were excluded from the imaging analyses, in which case, the
BOLD responses would not have been contaminated by potential delays in RT. See the
Supplementary Materials in the online supporting information located at https://www
.mitpressjournals.org/doi/suppl/10.1162/nol_a_00027 for further details.
Mean disfluency rates for each subject were calculated as the percentage of trials that
contained one or more disfluency error subtypes, and mean error rates for each subject were
calculated as the percentage of trials that contained one or more error subtypes. For each produc-
tion containing no sequencing or disfluency errors, utterance onset and offset were automatically
labeled based on sound pressure level thresholds, then hand-checked. Note that, in the neuro-
imaging component of the study (described below), only trials in which participants produced
the target sequence accurately and fluently were analyzed.
FMRI Paradigm
In addition to the three speaking conditions ( practiced native vs. practiced non-native vs. novel
non-native), a silent baseline condition was intermixed during imaging in which participants
viewed a series of asterisks on the screen instead of the orthographic stimulus and rested quietly
instead of uttering a word. FMRI data were acquired using a sparse sampling protocol (see Belin
et al., 1999; Gracco et al., 2005; Perrachione & Ghosh, 2013) that allowed participants to pro-
duce the target syllables during silent intervals between volume acquisitions. A single volume was
acquired approximately 4 s after speech onset on each trial which aligns with the 4–6 s delay in
peak BOLD response onset (Belin et al., 1999). By scanning between speech productions, this
protocol avoids the influence of scanner noise on speaker performance and brain activity
responses and image artifacts resulting from speech-induced motion of the head.
The cued sequence paradigm was identical to that used during training (outside of the scanner)
except with an additional pause after the production of each syllable to temporally align the
image acquisition to the expected peak of the hemodynamic response. As previously described,
the test phase consisted of eight blocks of trials which corresponded to eight functional runs. A
single volume was recorded on each trial and the delay between volumes was approximately
10 s. Each functional run contained five or six productions of each stimulus item, for a total of
40 trials per run (each run lasted approximately 6–7 min). This resulted in a total of 320 test trials
(80 trials per condition, including baseline). Conditions were shuffled and pseudorandomly
distributed across the eight functional runs with at least eight instances of each condition
appearing in each run.
Image Acquisition
All neuro-imaging data were acquired using a 3-Tesla Siemens TIM Trio scanner, equipped with
a 32-channel head coil. T2*-weighted gradient echo-planar fMRI images were collected
to assess BOLD responses during the test phase. Forty-one horizontal slices were collected in
each functional volume (in-plane resolution = 3.1 mm2, slice thickness = 3 mm, gap = 25%,
acquisition time = 2.5 s, echo time [TE] = 20 ms); volumes were automatically aligned to the
anterior commissure–posterior commissure line. Prior to collecting functional data, a gradient-
echo field map sequence was collected; the resulting magnitude and phase images enabled
offline correction of magnetic field distortions in functional images during data preprocessing
(see below; Jezzard & Balaban, 1995). Structural images were collected using a T1-weighted
Neurobiology of Language
114
l
D
o
w
n
o
a
d
e
d
f
r
o
m
h
t
t
p
:
/
/
d
i
r
e
c
t
.
m
i
t
.
e
d
u
n
o
/
l
/
l
a
r
t
i
c
e
-
p
d
f
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
a
_
0
0
0
2
7
p
d
.
/
l
f
b
y
g
u
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
multi-echo MPRAGE pulse sequence (MEMPRAGE, voxel size = 1 mm3, 176 axial slices, 256 ×
256 field of view, repetition time [TR] = 2,530 ms, TE = 3.44 ms, flip angle = 7°).
FMRI Data Analysis
Image preprocessing
Functional data were processed using tools from the following software packages that were inte-
grated into a processing stream within SPM12 (Statistical Parametric Mapping, v12; www.fil.ion
.ucl.ac.uk/spm/): FreeSurfer (Dale et al., 1999; Fischl, Salat, et al., 2002; Fischl, Sereno, et al.,
1999; www.freesurfer.net), Artifact Detection Tools (ART; www.nitrc.org/projects/artifact
_detect/), and the CONN toolbox (Whitfield-Gabrieli & Nieto-Castañón, 2012). Freesurfer was
used to remove non-brain components of the T1 structural volumes; segment the brain into gray
matter, white matter, and cerebral spinal fluid components; generate a reconstruction of the
cortical surfaces of each hemisphere; and identify cortical and subcortical regions of interest
(ROIs) (see below). Functional data were preprocessed through two pipelines: a surface/vertex-
based pipeline for analysis of cortical responses and a volume/voxel-based pipeline for analysis
of subcortical basal ganglia and cerebellar responses. Prior to pre-processing, the first volume
of each functional series was removed because it served only as a trigger for the initial
experimental trial.
A surface/vertex-based analysis pipeline was used to assess BOLD response differences for
each experimental group and for each contrast in the cerebral cortex. Functional images from
each subject were simultaneously realigned to the mean subject image and unwarped (motion-
by-inhomogeneity interactions) using the SPM12 realign and unwarp procedure (Andersson
et al., 2001). Outlier scans were detected using ART based on framewise displacement (scan-
to-scan motion threshold of 0.9 mm) and mean signal change (scan-to-scan signal change
threshold of 5 standard deviations above the mean (see Nieto-Castañón, 2020, for details).
Framewise displacement was computed at each timepoint by considering a 140 × 180 × 115 mm
bounding box around the brain and estimating the largest displacement among six control points
placed at the center of the bounding-box faces. Global BOLD signal change was computed at
each timepoint as the change in average BOLD signal within SPM’s global-mean mask scaled to
standard deviation units. Functional volumes from each subject were then coregistered with their
high-resolution T1 structural images and resliced using SPM12’s inter-modal registration proce-
dure with a normalized mutual information objective function. The functional data were then
resampled at the location of the FreeSurfer fsaverage level-8 tessellation (163,842 vertices and
327,680 faces) projected on each subject-specific cortical surface, averaged across 10 intervals
along the normal between the white matter and pial surfaces, and smoothed using iterative
diffusion smoothing with a series of 40 discrete steps, approximately equivalent to a 8 mm full-
width half-maximum (FWHM) two-dimensional Gaussian smoothing kernel (Hagler et al., 2006;
Nieto-Castañón, 2020).
A volume/voxel-based analysis pipeline was used to identify differences in subcortical BOLD
responses. Following the realignment, unwarping steps described above, functional volumes,
and the original T1 structural volumes, were simultaneously segmented and normalized directly
to Montreal Neurological Institute (MNI) space using SPM12’s combined normalization and
segmentation procedure (Ashburner & Friston, 2005). Prior to MNI-normalization, both the func-
tional and anatomical volume origins were centered to coordinates [0, 0, 0] in order to improve
the quality of the iterative procedure initial starting estimates. Functional volumes were then
spatially smoothed using an 8 mm FWHM Gaussian kernel in order to increase BOLD signal-
to-noise ratio and reduce the influence of residual variability in functional and gyral anatomy
across subjects (Nieto-Castañón, 2020).
Neurobiology of Language
115
l
D
o
w
n
o
a
d
e
d
f
r
o
m
h
t
t
p
:
/
/
d
i
r
e
c
t
.
m
i
t
.
e
d
u
n
o
/
l
/
l
a
r
t
i
c
e
-
p
d
f
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
a
_
0
0
0
2
7
p
d
.
/
l
f
b
y
g
u
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
Subject-level BOLD contrast analyses
After preprocessing, BOLD responses were estimated using a general linear model in SPM12.
Because functional volumes were acquired in a sparse sequence, the hemodynamic response
function for each stimulus event was modeled as a finite impulse response. The model included
four condition-specific variables (practiced native, practiced non-native, novel non-native, and
baseline). Trials with productions that were perceptually rated as containing either a sequencing
error (e.g., phoneme deletions, insertions or substitutions) or a stuttering-like disfluency (e.g., pho-
neme/syllable repetitions, prolongations, or blocks), or were found to be outliers by ART were
modeled as separate conditions (one individual regressor per trial) thereby removing variability
resulting from these trials from the effects- and contrast-of-interest estimates. For each individual
run, regressors were added to the model to remove linear effects of time (e.g., signal drift, ad-
aptation) and the six motion covariates (taken from the realignment step) and a constant term.
The model was estimated at each vertex (surface-based) analysis or voxel (volume-based) anal-
ysis for each participant, resulting in two surface maps (one for each hemisphere) and a volume
map of the model regressor coefficients for each condition. These condition estimates were then
contrasted to yield effect-size maps of the following contrasts of interest: differences in the
response during novel non-native and practiced non-native conditions (novel non-native−
practiced non-native), and differences in the response during the practiced non-native and
practiced native conditions ( practiced non-native−practiced native).
Group-level analyses
Group-level t statistics were calculated separately for each contrast map. Cluster-level inferences
were based on Threshold Free Cluster Enhancement (TFCE; Smith & Nichols, 2009) with default
parameters extent = 0.5 and height = 2. This method assigns TFCE scores to each voxel
characterizing the amount of cluster-like local spatial support at each location. These scores
are then compared to their expected distribution under the null hypothesis, estimated using
1,000 randomization/permutation iterations, and the results are thresholded at family-wise
error (FWE) false positive probability ( pFWE) < 0.025. This threshold was used as a conservative
means to ensure a cluster-level pFWE < 0.05 across the cortical surface and subcortical volume
analyses.
Region-of-Interest-Based Analyses
To increase statistical power and sensitivity, we supplemented the surface/vertex-based and
volume/voxel-based analyses with ROI analyses (Nieto-Castañón et al., 2003) based on a priori
hypotheses derived from the results of Segawa, Tourville, et al. (2015). Specifically, we used
anatomically defined ROIs in the speech production network that overlapped areas of significant
BOLD activity in Segawa, Tourville, et al. (2015) for each contrast of interest. The predefined
cortical and subcortical (basal ganglia and cerebellar) ROIs are listed in Table 2; a detailed
description of the anatomical landmarks used for cortical parcellation are provided in
Tourville and Guenther (2003). For the novel non-native−practiced non-native contrast, we
tested whether brain activity was significantly different in globus pallidus internal (GPi) and
external (GPe) segment ROIs; these ROIs were derived from the probabilistic atlas of the basal
ganglia described by Keuken et al. (2014). For the practiced non-native−practiced native con-
trast, we tested whether brain activity was significantly different in right cerebellum lobule VI,
which was derived from the SUIT probabilistic atlas of the cerebellum (Diedrichsen et al.,
2009). Affirmation of differences in these ROIs would replicate the Segawa, Tourville, et al.
(2015) findings with neurotypical speakers.
Neurobiology of Language
116
l
D
o
w
n
o
a
d
e
d
f
r
o
m
h
t
t
p
:
/
/
d
i
r
e
c
t
.
m
i
t
.
e
d
u
n
o
/
l
/
l
a
r
t
i
c
e
-
p
d
f
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
a
_
0
0
0
2
7
p
d
.
/
l
f
b
y
g
u
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
Table 2.
in the current analyses
List of the predefined cortical, subcortical, and cerebellar regions-of-interest (ROIs) used
Novel non-native−practiced non-native contrast
Subcortical ROIs
L GPi
L GPe
Practiced non-native−practiced native contrast
Subcortical ROIs
R cerebellum lobule VI
Brain-behavior motor learning correlation analyses
Cortical ROIs
L aINS
R aINS
L PT
R preSMA
L vPMC
L pSTS
L midPMC
L aFO
R aFO
L pIFS
L STG
L STS
Subcortical ROIs
L GPi
L GPe
Brain-behavior stuttering severity correlation analyses (AWS only)
Cortical ROIs
L vPMC + midPMC + vMC + midMC
L preSMA + SMA
Neurobiology of Language
117
l
D
o
w
n
o
a
d
e
d
f
r
o
m
h
t
t
p
:
/
/
d
i
r
e
c
t
.
m
i
t
.
e
d
u
n
o
/
l
/
l
a
r
t
i
c
e
-
p
d
f
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
a
_
0
0
0
2
7
p
d
.
/
l
f
b
y
g
u
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
Table 2.
(continued )
Subcortical ROIs
L GPi
L GPe
L putamen
L caudate
L VA
L VL
Brain-Behavior Correlation Analyses
Two types of hypothesis-driven analyses were conducted to identify potential relationships
between behavioral measures and brain activity. In the first type of analysis, we conducted
multiple correlation tests to identify relationships between behavioral measures of motor
sequence learning success and BOLD activity. Specifically, we tested for correlations between
(i) the mean BOLD response in the cortical and subcortical ROIs that showed task-activated
clusters in Segawa, Tourville, et al. (2015; Table 2) in both the novel non-native−practice
non-native and the practiced non-native−practiced native contrasts and (ii) each of the three
motor learning indices (error rate, utterance duration, and RT). No correction was applied for
the number of ROIs in this ROI list. FreeSurfer was used to define the ROIs on each individual
cortical surface using the labeling system described in Cai et al. (2014). These ROIs included
the left-lateralized aINS, vPMC, anterior frontal operculum (aFO), preSMA, IFS, posterior
superior temporal sulcus (pSTS), and planum temporale (PT), as well as the left GPi and
GPe. For each participant, we computed each motor learning index by subtracting the mean
performance difference between the novel non-native and practiced non-native productions.
We normalized for differences between participants by dividing these differences by the prac-
ticed non-native syllable measure, on a per-participant basis. For example, the utterance dura-
tion learning measure was the mean duration difference between the novel non-native and
practiced non-native productions divided by mean duration of the practiced non-native pro-
ductions. Each motor learning index was then correlated with the mean beta coefficient within
each significant cluster from each contrast.
In the second type of analysis, SSI measures (Riley, 2009) of stuttering frequency and overall
severity (described above) were used, in addition to the number of disfluencies produced
during the test phase (inside the scanner), to identify potential relations between stuttering-
related behaviors and brain activity. Specifically, we tested for correlations in the AWS group
only with the mean composite SSI scores and mean disfluency rates during test (inside the
scanner) and the mean BOLD responses in eight hypothesis-based ROIs within the cortico-
BG loops (listed in Table 2) in the novel non-native−practice non-native contrast (based on
fluent trials only). These ROIs included left premotor and primary motor areas (ventral and
mid premotor cortices [vPMC, midPMC] + ventral and mid motor cortices [vMC, midMC]), left
medial prefrontal areas (left SMA + preSMA), as well as several left-lateralized subcortical
basal ganglia sites (GPi, GPe, caudate, putamen, ventral anterior nucleus [VA], ventral lateral
nucleus [VL]).
Ventral premotor cortex (vPMC):
Brain region that is critically important
in programming speech movements.
Neurobiology of Language
118
l
D
o
w
n
o
a
d
e
d
f
r
o
m
h
t
t
p
:
/
/
d
i
r
e
c
t
.
m
i
t
.
e
d
u
n
o
/
l
/
l
a
r
t
i
c
e
-
p
d
f
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
a
_
0
0
0
2
7
p
d
.
/
l
f
b
y
g
u
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
RESULTS
Behavioral Measures of Speech Motor Sequence Learning
Training phase
Table 3 shows a summary of the behavioral results for each phase of the experiment and for each
group (ANS vs. AWS). Our first set of analyses examined, at the behavioral level, whether partic-
ipants showed evidence of incremental speech motor sequence learning over the course of the
training phase by examining the time course of improvement in each of the three performance
measures—mean error rates, utterance durations, and RTs—over the two days of speech motor
practice. Each performance measure was averaged within each group, training block, condition,
and participant. Again, duration and RT measures were only analyzed for utterances that were
perceptually coded as having been properly executed (see above) on each day. To visualize the
running estimate of the learning trajectories across participants, Figure 1 plots the time course of
improvement of each performance measure as a function of group, training block, and condition.
Separate analyses of variance (ANOVAs) were performed on the mean sequencing error rates,
durations, and RTs with training block (1–8) and condition (native vs. non-native) as within-
subjects factors and group (ANS vs. AWS) as a between-subjects factor. In these and all subse-
quent ANOVAs, Greenhouse-Geisser corrections were applied when appropriate and partial
eta-squared effect sizes were calculated for all main effects and interactions. Post-hoc pairwise
comparisons were reported as significant at the 0.05 level.
2 = 0.878] and block [F(7, 203) = 15.985, p < 0.001, (cid:1)
p
The ANOVA performed on mean error rates (shown in Figure 1A) revealed a significant main
effect of group [F(1, 29) = 5.036, p = 0.033, (cid:1)
2 = 0.148], such that AWS [mean (M ) = 32.7, stan-
p
dard deviation (SD) = 13.6] produced more sequencing errors than ANS [M = 23.0, SD = 10.3] in
general. There were also highly significant main effects of condition [F(1, 29) = 208.822, p <
0.001, (cid:1)
2 = 0.355]. There was one sig-
p
nificant interaction, the stimulus condition × block interaction [F(7, 203) = 8.863, p < 0.001, (cid:1)
2 =
p
0.234]. All other interactions were not significant [p > 0.05, in all cases]. Simple effects tests on
the condition × block interaction revealed that there was a significant effect of block on both the
native sequences [F(7, 210) = 2.958, p = 0.045, (cid:1)
2 = 0.090] and the non-native sequences [F(7,
p
210) = 20.495, p < 0.001, (cid:1)
2 = 0.406]. However, the effect size was considerably larger in the
p
non-native condition, indicating that sequence learning was greater for novel words with unfa-
miliar consonant clusters than novel words with familiar consonant clusters during training. This
is in keeping with the behavioral results reported by Segawa and colleagues (Segawa, Tourville,
et al., 2015; Segawa, Masapollo, et al., 2019) which found that relatively less learning occurred
for native compared to non-native sequences.
The ANOVA performed on mean utterance duration (shown in Figure 1B) revealed a main
effect of group [F(1, 29) = 8.963, p = 0.006, (cid:1)p
2 = 0.236], such that ANS produced shorter utter-
ances [M = 0.59, SD = 0.05] than AWS [M = 0.69, SD = 0.10]. There was also a significant effect
of stimulus condition [F(1, 7) = 34.318, p < 0.001, (cid:1)
2 = 0.542], such that the native sequences
p
[M = 0.61, SD = 0.09] were uttered faster than the non-native sequences [M = 0.67, SD = 0.10],
as well as a condition × block interaction [F(7, 203) = 2.066, p = 0.049, (cid:1)
2 = 0.066]. All other
p
interactions were not significant [p > 0.05, in all cases]. Consistent with our expectations based
on our prior studies, simple effects tests on the condition × block interaction revealed that there
was a significant effect of block on the non-native sequences [F(7, 210) = 3.340, p = 0.002, (cid:1)
2 =
p
0.100] but not on the native sequences [F(7, 210) = 1.038, p = 0.382, (cid:1)
2 = 0.033]. Ainsi,
p
participants only got reliably faster at executing novel words during training if they contained
unfamiliar consonant clusters, likely because they were near ceiling performance for novel
words using native clusters.
Neurobiology of Language
119
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
e
d
toi
n
o
/
je
/
je
un
r
t
je
c
e
–
p
d
F
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
un
_
0
0
0
2
7
p
d
.
/
je
F
b
oui
g
toi
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
Dependent measure: Error rates
Training phase
Tableau 3.
ANOVA results for behavioral analyses
Test phase
Effect
Block
Condition
Groupe
Block × Condition
Block × Group
Condition × Group
Block × Condition × Group
F
df
p
η
2
p
Effect
F
df
p
η
2
p
15.985
208.822
5.036
8.863
1.554
1.137
1.312
7
1
1
7
7
1
7
<0.001 0.355 Block <0.001 0.878 Condition 0.033 0.148 Group 5.291 91.164 1.371 7 2 1 <0.001 0.159 <0.001 0.765 0.251 0.047 <0.001 0.234 Block × Condition 1.504 14 0.106 0.051 0.183 0.051 Block × Group 0.295 0.038 Condition × Group 1.164 0.838 7 2 0.325 0.04 0.438 0.029 0.268 0.043 Block × Condition × Group 0.647 14 0.825 0.023 Dependent measure: Utterance duration Effect Block Condition Group Block × Condition Block × Group Condition × Group Block × Condition × Group F df p η 2 p Effect F df p η 2 p 2.278 34.318 8.963 2.066 0.132 0.506 1.358 7 1 1 7 7 1 7 0.09 0.073 Block <0.001 0.542 Condition 0.006 0.236 Group 2.638 40.733 0.112 7 2 1 0.033 0.083 <0.001 0.584 0.74 0.004 0.049 0.066 Block × Condition 1.118 14 0.354 0.037 0.931 0.005 Block × Group 0.483 0.017 Condition × Group 0.517 0.949 7 2 0.738 0.018 0.357 0.032 0.25 0.045 Block × Condition × Group 1.059 14 0.388 0.035 Dependent measure: Reaction time Effect Block Condition Group Block × Condition Block × Group Condition × Group Block × Condition × Group F df p ηp 2 Effect F df p ηp 2 16.742 0.487 1.934 1.938 0.36 3.732 1.099 7 1 1 7 7 1 7 <0.001 0.366 Block 0.491 0.017 Condition 0.175 0.063 Group 3.114 1.688 5.926 7 2 1 0.039 0.097 0.2 0.054 0.021 0.17 0.094 0.063 Block × Condition 1.012 14 0.376 0.034 0.807 0.012 Block × Group 0.063 0.114 Condition × Group 0.851 4.973 7 2 0.455 0.029 0.012 0.146 0.363 0.037 Block × Condition × Group 0.805 14 0.664 0.027 Neurobiology of Language 120 l D o w n o a d e d f r o m h t t p : / / d i r e c t . m i t . e d u n o / l / l a r t i c e - p d f / / / / 2 1 1 0 6 1 8 9 9 6 7 7 n o _ a _ 0 0 0 2 7 p d / . l f b y g u e s t t o n 0 8 S e p e m b e r 2 0 2 3 Neural correlates of speech motor sequence learning Table 3. (continued ) Dependent measure: Disfluency rates (AWS only) Training phase Test phase Effect Block Condition F 2.618 1.379 df 7 2 p η 2 p Effect 0.084 0.158 Block 0.26 0.09 Condition F 1.095 9.959 df 7 2 p η 2 p 0.372 0.068 0.001 0.399 Block × Condition 2.038 14 0.108 0.127 Block × Condition 0.923 14 0.482 0.058 Note. Shown are the F value, the degrees of freedom, p value, and partial-eta-squared value for each effect. The ANOVA performed on mean RT scores (shown in Figure 1C) revealed a highly significant effect for block [F(7, 203) = 16.742, p < 0.001, (cid:1) 2 = 0.366], such that participants got faster at p initiating their utterances during the training sessions. The effects of group, condition, and all other interactions were not significant [p > 0.05 in all cases].
In an additional analysis, we examined the mean disfluency rates for the AWS group only.
These are shown in Figure 2 averaged across each sequence type (native vs. non-native) et
block (1–8). An ANOVA on these scores—sequence type × block—showed no significant main
effects or interaction [p > 0.05 in all cases], indicating that stuttering speakers were equally
disfluent for both sequence types throughout training.
Test phase
Our second set of analyses examined the same set of behavioral measures of sequence generation
while participants were inside the MRI scanner during the test phase. Each performance measure
was averaged within each group, testing block, condition, and participant. Duration and RT
measures were again only analyzed for utterances coded as properly sequenced productions
(see above) on each day. Tableau 4 shows the mean error rates during the test phase as a function
Chiffre 1. Time course of improvement of behavioral performance measures during training (outside the scanner) for the practiced native and
practiced non-native sequences as a function of training block (1–8) and group (ANS vs. AWS). (UN) Mean percentage of errors of each se-
quence type; (B) Mean durations of the properly executed utterances of each sequence type; (C) Mean reaction times of the properly executed
utterances of each sequence type. The shaded area represents the standard error of the mean.
Neurobiology of Language
121
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
e
d
toi
n
o
/
je
/
je
un
r
t
je
c
e
–
p
d
F
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
un
_
0
0
0
2
7
p
d
.
/
je
F
b
oui
g
toi
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
Chiffre 2. Mean percentage of disfluency errors for the practiced native and practiced non-native
sequences during training (outside the scanner) as a function of training block (1–8) for AWS only.
The shaded area represents the standard error of the mean.
of error subtype and sequence type. Chiffre 3 shows the mean error rates, utterance duration, et
RT scores as a function of group, test block, and condition. Separate ANOVAs were performed on
the mean error rates, utterance durations, and RTs with group (AWS vs. ANS) as a between-
subjects factor, and test block (1–8) and experimental condition ( practiced native vs. practiced
non-native vs. novel non-native) as within-subjects factors.
ANOVA performed on mean error rates (shown in Figure 3A) revealed significant main effects
of block [F(7, 196) = 5.291, p < 0.001, (cid:1)2 = 0.159] and condition [F(2, 56) = 91.164, p < 0.001,
(cid:1)2 = 0.765]. There was no significant main effect of group [F(1, 28) = 1.371, p = 0.251, (cid:1)2 = 0.092]
or interaction effects [p > 0.05 in all cases]. Post-hoc t-test comparisons performed on the main
effect of condition indicated that, regardless of group, the mean error rates for the practiced native
syllables [M = 4.0. SD = 7.2] were significantly lower than the practiced non-native syllables [M =
34.8, SD = 20.2, t(30) = −9.357, p < 0.001, Cohen’s d = 2.03] and novel non-native syllables [M =
44.7, SD = 21.0, t(30) = −10.978, p < 0.001, d = 2.59], and the mean error rates for the practiced
Table 4. Mean error rates in the test blocks (inside the scanner) by error subtype and sequence type
Sequencing errors
Approximation
of target
Unrecognizable
from target
Unfinished utterance
Silence
Practiced
native
14.6
0.0
0.0
0.0
Neurobiology of Language
ANS
Sequence type
Practiced
non-native
30.7
Novel
non-native
39.4
Practiced
native
32.5
AWS
Sequence type
Practiced
non-native
33.3
0.0
0.0
0.0
0.0
0.0
0.0
0.3
0.2
0.6
0.1
2.8
1.8
Novel
non-native
37.6
0.3
2.3
2.7
122
l
D
o
w
n
o
a
d
e
d
f
r
o
m
h
t
t
p
:
/
/
d
i
r
e
c
t
.
m
i
t
.
e
d
u
n
o
/
l
/
l
a
r
t
i
c
e
-
p
d
f
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
a
_
0
0
0
2
7
p
d
.
/
l
f
b
y
g
u
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
Figure 3. Behavioral performance measures during test (inside the scanner) for the practiced native, practiced non-native, and novel non-
native sequences as a function of test block (1–8) and group (ANS vs. AWS). (A) Mean percentage of errors of each sequence type; (B) Mean
durations of the properly executed utterances of each sequence type; (C) Mean reaction times of the properly executed utterances of each
sequence type. The shaded area represents the standard error of the mean.
non-native syllables were lower than the novel non-native syllables [t(30) = −4.653, p < 0.001,
d = 0.48].
Measures of mean utterance duration (shown in Figure 3B) patterned similarly. ANOVA
performed on mean utterance durations indicated that there were also significant main effects
of block [F(7, 203) = 2.638, p = 0.033, (cid:1)2 = 0.083] and condition [F(2, 58) = 40.733, p <
0.001, (cid:1)2 = 0.584]. Again, there was no significant main effect of group [F(1, 29) = 0.112,
p = 0.740, (cid:1)2 = 0.004] or interaction effects [p > 0.05 in all cases]. Post-hoc t-test comparisons
performed on the main effect of condition indicated that, regardless of group, participants were
faster at executing the practiced native syllables [M = 0.67, SD = 0.15] than the practiced
non-native syllables [M = 0.74, SD = 0.16, t(30) = −6.797, p < 0.001, d = 0.45] and novel
non-native syllables [M = 0.77, SD = 0.19, t(30) = −7.454, p < 0.001, d = 0.58]. In addition,
participants were faster at executing the practiced non-native syllables than the novel non-
native syllables [t(30) = −3.044, p = 0.005, d = 0.17].
ANOVA performed on mean RT scores (shown in Figure 3C) showed no main effect of con-
dition [F(2, 58) = 1.688, p = 0.200, (cid:1)
2 = 0.054]. There were, however, significant main effects of
p
block [F(7, 203) = 3.114, p = 0.039, (cid:1)
2 = 0.097] and group [F(1, 29) = 5.926, p = 0.021, (cid:1)
2 =
p
p
0.170], such that AWS took longer to initiate their utterances [M = 0.71, SD = 0.36] than ANS
[M = 0.48, SD = 0.16]. The condition × group interaction effect also reached statistical signif-
icance [F(2, 58) = 4.973, p = 0.012, (cid:1)
2 = 0.146]. Simple effects tests on the condition × group
p
interaction indicated that the effect of condition approached significance in AWS [F(2, 30) =
3.555, p = 0.053, (cid:1)
2 = 0.037]. All
p
other interactions were not significant [p > 0.05 in all cases].
2 = 0.369], but not in ANS [F(2, 28) = 0.540, p = 0.589, (cid:1)
p
Enfin, we again examined mean disfluency rates for the AWS group while in the scanner.
These scores are shown in Figure 4. ANOVA performed on these scores—sequence type ×
block—revealed a highly significant main effect of condition [F(2, 30) = 9.959, p = <
0.001, (cid:1)p
2 = 0.399]. There was no significant main effect of block or interaction effect [p >
0.05 in both cases]. Post-hoc t-test comparisons performed on the main effect of condition
indicated that AWS were less disfluent when executing the practiced native syllables [M =
4.6, SD = 11.8] compared to the practiced non-native syllables [M = 7.5, SD = 15.6, t(15) =
−2.442, p = 0.027, d = 0.32] and novel non-native syllables [M = 9.8, SD = 19.4, t(15) =
−3.476, p = 0.003, d = 0.49]. As well, AWS were less disfluent when producing the practiced
non-native than the novel non-native syllables [t(15) = −2.307, p = 0.036, d = 0.19].
Neurobiology of Language
123
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
e
d
toi
n
o
/
je
/
je
un
r
t
je
c
e
–
p
d
F
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
un
_
0
0
0
2
7
p
d
/
.
je
F
b
oui
g
toi
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
Chiffre 4. Mean percentage of disfluency errors (inside the scanner) for the practiced native, prac-
ticed non-native, and novel non-native sequences during test (inside the scanner) as a function of
test block (1−8) for AWS only. The shaded area represents the standard error of the mean.
En résumé, we obtained similar results to those in our prior studies on speech motor sequence
learning with neurotypical speakers (Segawa, Tourville, et coll., 2015; Segawa, Masapollo, et coll.,
2019): Participants—ANS and AWS alike—showed incremental improvements in performance
speed and accuracy, especially for the non-native sequences, with repetition and practice during
training and these gains were maintained overnight between experimental sessions. De plus,
the data indicated that, after two days of extended speech motor practice, speakers from both
groups showed further performance gains in accuracy (reduced error rate) for the practiced
non-native sequences throughout the test phase (Figure 3A). Although both groups showed com-
parable gains, the AWS produced the sequences slower and/or less accurately prior to and after
entraînement. Dans l'ensemble, these findings provide evidence at the behavioral level that AWS do not show
deficits in the acquisition or retention of new speech motor sequences. Enfin, RT scores were
notably higher during test (inside the scanner) than during training (outside of the scanner), surtout-
cially for the AWS group. Given that the experimental paradigm during the test phase was iden-
tical to that used during the training phase, this difference in initiation speed likely reflects
differences in performance anxiety outside versus inside the scanner.
Neural Correlates of Speech Motor Sequence Learning
FMRI analysis
Before performing direct group comparisons, we first assessed differences between the condition-
specific brain activations using the pooled results (ANS and AWS combined). For each of the two
speech contrast conditions, we report the results from one-sided (positive only) tests. Figure 5A and
Tableau 5 show the brain regions that were significantly more active during the execution of novel
non-native than practiced non-native syllables (TFCE cluster-level pFWE < 0.05) aggregated across
both experimental groups. The surface/vertex-based analyses revealed that the production of novel
non-native syllables resulted in greater BOLD response in preSMA, aINS, aFO, pFO, and IPS bilat-
erally. In the left hemisphere, additional cortical clusters were noted with peaks in IFS, vPMC, pos-
terior inferior frontal gyrus (pIFG), midPMC, vMC, midMC, superior temporal gyrus (STG), and
inferior temporal occipital (ITO) region. The volume/voxel-based analysis found no statistically
Neurobiology of Language
124
l
D
o
w
n
o
a
d
e
d
f
r
o
m
h
t
t
p
:
/
/
d
i
r
e
c
t
.
m
i
t
.
e
d
u
n
o
/
l
/
l
a
r
t
i
c
e
-
p
d
f
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
a
_
0
0
0
2
7
p
d
/
.
l
f
b
y
g
u
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
l
D
o
w
n
o
a
d
e
d
f
r
o
m
h
t
t
p
:
/
/
d
i
r
e
c
t
.
m
i
t
.
e
d
u
n
o
/
l
/
l
a
r
t
i
c
e
-
p
d
f
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
a
_
0
0
0
2
7
p
d
/
.
l
f
b
y
g
u
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Figure 5. Brain areas showing greater BOLD activation for novel non-native than practiced non-native trials. (A) Averaged across all 15 ANS
and 16 AWS; (B) 15 ANS (left) and 16 AWS (right). Activation is displayed on a canonical inflated cortical surface. Colors indicate the relative
significance level at each voxel/vertex for the comparison of novel non-native and practiced non-native trials.
significant differences in subcortical activity. We supplemented this voxel-based analysis with a
hypothesis-based subcortical ROI analysis based on the results reported in Segawa, Tourville,
et al. (2015) for this contrast. Specifically, we tested for differences in activation in the left GPi
and GPe. Results from this analysis demonstrated that the left GPi was more active during produc-
tion of the novel non-native than the practiced non-native words [t(30) = 2.22, punc = 0.034, r =
0.375], in effect replicating the finding of Segawa, Tourville, et al. (2015) for the same contrast. No
significant groups effects (ANS vs. AWS) were found for any cortical or subcortical region in either
hemisphere for the novel non-native–practiced non-native contrast; nevertheless, we present the
results (albeit non-significant) for the two groups separately in Figure 5B.
Figure 6A and Table 6 show the brain regions that were significantly more active during the
execution of practiced non-native than practiced native syllables (TFCE cluster-level pFWE <
0.05). The surface/vertex-based analyses revealed that the production of practiced non-native
words resulted in greater BOLD response in pSTG and occipital cortex (OC) bilaterally. The in-
creased activity in OC likely reflects greater attention and/or depth of processing while reading
Neurobiology of Language
125
Neural correlates of speech motor sequence learning
Table 5.
Summary of significant cortical and subcortical activation peaks for the novel non-native−practiced non-native contrast
Novel non-native–practiced non-native contrast*
Anatomical region**
Left hemisphere
L pIFS, L aINS, L vPMC, L pFO, L midPMC,
L aFO, L pMFG, L aIFS, L midMC, L dIFO,
L vIFO, L IFR, L vMC, L aMFG, L FOC
No.
peaks
MNI coordinates of peak vertex
y
z
x
Size
TFCE
pFWE
33
−29
26
7
10170
5292.04
<0.0001
L SPL, L AG, L pSMG, L aSMG, L OC, L midSC
27
L ITO, L pITG, L TOF
L preSMA, L SFG, L SMA
L pdSTS, L pSTG, L PT, L pvSTS
L pITG
Right hemisphere
R aINS, R pFO, R aFO, R IFR, R FOC
R SPL, R AG, R OC
R SFG, R preSMA
R preSMA
7
3
3
5
9
1
3
1
−27
−47
−7
−63
−47
36
29
9
7
−65
−59
12
−35
−43
30
−62
21
16
37
−9
55
4
−21
−1
35
38
55
7665
4210.05
<0.0001
713
2009.43
0.001
1150
1663.39
0.002
1662
1262.41
0.005
23
936.82
0.024
2202
2562.18
<0.0001
1066
1923.23
0.002
180
267
1003.95
0.019
953.0
0.024
Note. From left to right, the columns show the anatomical region name(s), number of peaks, MNI stereotactic coordinates, cluster size, TFCE value, and pFWE value.
* For the one-sided (positive-only) contrast.
** Cluster regions are listed in descending order of number of significant vertices.
l
D
o
w
n
o
a
d
e
d
f
r
o
m
h
t
t
p
:
/
/
d
i
r
e
c
t
.
m
i
t
.
e
d
u
n
o
/
l
/
l
a
r
t
i
c
e
-
p
d
f
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
a
_
0
0
0
2
7
p
d
/
.
l
f
b
y
g
u
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
the non-native text stimuli, rather than differences in speech production. In the left hemisphere,
additional cortical clusters were noted with peaks in preSMA, aINS, FO, pIFG, vPMC, midPMC,
aSTG, ventral division of somatosensory cortex (vSC), and IPS. The volume/voxel-based analysis
found increased activation in the right cerebellum (lobule XIII) during the practiced non-native
than the practiced native condition. A hypothesis-based subcortical ROI analysis based on the
results reported in Segawa, Tourville, et al. (2015) for this contrast was then performed.
Specifically, we tested for differences in activation in right cerebellum (lobule VI). Results from
this analysis demonstrated that this region was more active during production of the practiced
non-native than the practiced native words [t(30) = 2.61, pFDR = 0.014, r = 0.430], again repli-
cating findings from Segawa, Tourville, et al. (2015). As in the other contrast, no region in either
hemisphere was found to be significantly more active for this contrast in either group (ANS vs.
AWS) at the cortical or subcortical levels, further bolstering the view that speech motor learning
mechanisms are unimpaired in stuttering; again, we present the results (albeit non-significant) for
the two groups separately in Figures 6B.
Although no significant groups effects were found, several interesting trends emerged that are
worth noting and speculating about. First, brain areas linked to speech premotor planning (vPMC,
midPMC, FO, aINS) showed a non-significant trend toward greater activation in ANS during
novel sequence production, suggesting that neurotypical speakers may show greater learning
Neurobiology of Language
126
Neural correlates of speech motor sequence learning
l
D
o
w
n
o
a
d
e
d
f
r
o
m
h
t
t
p
:
/
/
d
i
r
e
c
t
.
m
i
t
.
e
d
u
n
o
/
l
/
l
a
r
t
i
c
e
-
p
d
f
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
a
_
0
0
0
2
7
p
d
/
.
l
Figure 6. Brain areas showing greater BOLD activation for practiced non-native than practiced native trials. (A) Averaged across all 15 ANS
and 16 AWS; (B) 15 ANS (left) and 16 AWS (right). Activation is displayed on a canonical inflated cortical surface. The significant subcortical
cluster is shown on a slice through the cerebellum at z = −54 in the MNI template (panel A, bottom right); left and right hemispheres are
indicated by L and R, respectively. Colors indicate the relative significance level at each voxel/vertex for the comparison of novel non-native
and practiced non-native trials.
f
b
y
g
u
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
of the motor chunks for the trained sequences than AWS. Second, STG, an auditory processing
area, showed a non-significant trend toward greater activation in AWS during novel sequence
production, suggesting that, following motor practice, stuttering speakers may still be relying
more on auditory feedback to guide them toward the intended production targets compared to
ANS. Critically, however, there were no brain areas activated in AWS that fell outside the network
of areas identified in ANS and in the previous study (Segawa, Tourville, et al., 2015).
Brain-Behavior Correlation Analysis
The correlation tests between behavioral measures of learning success and mean activation in the
12 hypothesis-based ROIs from Segawa, Tourville, et al. (2015) (listed in Table 2) for the novel
non-native−practiced non-native contrast revealed no group differences at either the cortical or
subcortical levels. In the analyses performed on the pooled data (ANS and AWS combined), we
found that learning success, as measured by the participant-normalized difference in mean error
Neurobiology of Language
127
Neural correlates of speech motor sequence learning
Table 6.
Summary of significant cortical and subcortical activation peaks for the practiced non-native−practiced native syllable contrast
Practiced non-native–practiced native contrast*
Anatomical region**
Left hemisphere
L SPL, L PT, L aSMG, L vSC, L PO, L pSTG, L H,
L PP, L pSMG,L AG, L midSC, L pdSTS, L aSTG,
L pCO, L OC, L adSTS, L pINS
L vPMC, L midPMC, L aINS, L pFO, L aFO, L vIFO,
L vMC, L dIFO, L pIFS, L IFR, L aCO, L pMFG
L preSMA, L SMA, L dCMA
L OC
L pMFG, L pdPMC, L mdPMC, L midPMC
Right hemisphere
R PP, R H, R pINS
R OC
Cerebellum lobule VIII
Cerebellum lobule VII b
No.
peaks
47
17
1
1
1
5
1
11
1
MNI coordinates of peak vertex
y
z
x
Size
TFCE
pFWE
−48
−51
−9
−15
−30
46
22
18
32
−38
1
12
−101
−8
−18
−100
−70
−74
13
13952
4603.4
<0.0001
45
50
−7
46
2
6
−46
−56
6766 3863.43 <0.0001
1246 1502.18
0.001
2089 1246.82
0.009
726
977.68
0.019
407
1668.33
0.001
1358 1583.67
0.001
304
846.54
0.009
1
645.83
0.024
Note. From left to right, the columns show the anatomical region name(s), number of peaks, MNI stereotactic coordinates, cluster size, TFCE value, and pFWE value.
* For the one-sided (positive-only) contrast.
** Cluster regions are listed in descending order of number of significant vertices.
rates between the novel non-native and practiced non-native words, was positively correlated
with the mean activation in both the left [t(29) = 2.22, p = 0.034, r = 0.381] and right aINS
[t(29) = 2.38, p = 0.024, r = 0.404], such that processing in these areas increased most with learning
in participants who showed the greatest performance improvements (shown in Figure 7). These
results provide support for the hypothesis that these premotor areas are involved in speech motor
sequence learning. No significant correlations emerged between the participant-normalized differ-
ences in mean utterance duration (between the novel non-native and practiced non-native words)
and the mean activation in any of the ROIs (in all cases, punc > 0.05).
Enfin, within the AWS group, we assessed whether stuttering severity (as indexed by SSI
scores and mean disfluency rates inside the scanner during the test phase) were correlated with
activation in eight hypothesis-based ROIs within the cortico-BG loops (listed in Table 2) for the
novel non-native−practiced non-native contrast. Correlation tests revealed that mean disfluency
rates inside the scanner were negatively correlated with mean activation in the left GPe [t(14) =
−2.88, pFDR = 0.047, r = −0.609] and the left caudate [t(14) = −2.74, pFDR = 0.047, r = −0.591]
(c'est à dire., the more disfluent participants were during the test phase, the less BOLD activity change was
observed in these basal ganglia sites for this contrast; shown in Figure 8). Although this result is
correlational and therefore a causal relationship cannot be firmly established, it is nonetheless
Neurobiology of Language
128
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
e
d
toi
n
o
/
je
/
je
un
r
t
je
c
e
–
p
d
F
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
un
_
0
0
0
2
7
p
d
.
/
je
F
b
oui
g
toi
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
Significant correlation between participant-normalized difference in mean error rates between the novel non-native and practiced
Chiffre 7.
non-native words and the mean activation in the left aINS [r = 0.381, p = 0.034] (left panel) and right aINS [r = 0.404, p = 0.024] (right panel).
compatible with the idea that speakers are more likely to stutter when the left GPe and the left
caudate are more hypoactive. In contrast, no statistically significant correlations were found
between SSI scores and activation in any of the eight ROIs. The latter result is perhaps unsurprising
given that the SSI does not measure disfluency during non-native sequence production and its
affiliated scores were not based on speech uttered inside the scanner.
En résumé, we found that, across both ANS and AWS, greater activity was observed during the
production of novel sequences in brain regions previously associated with learning and maintaining
speech motor programs, including lateral premotor cortex, FO, aINS, posterior superior temporal
cortex, and right cerebellum (Guenther, 2016; Segawa, Tourville, et coll., 2015). Measures of learning
success correlated positively with activity in the left and right aINS, suggesting that this brain area
plays an important role in speech motor learning. In AWS, measures of in-scanner disfluency rates
were negatively correlated with activity in left-lateralized basal ganglia structures. Dans l'ensemble, ces
Chiffre 8. Significant correlation between mean disfluency rates during the test phase (inside the scanner) and mean activation in the left GPe
[r = −0.609, pFDR = 0.047] (left panel) and the left caudate [r = −0.591, pFDR = 0.047] (right panel).
Neurobiology of Language
129
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
e
d
toi
n
o
/
je
/
je
un
r
t
je
c
e
–
p
d
F
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
un
_
0
0
0
2
7
p
d
.
/
je
F
b
oui
g
toi
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
findings provide evidence that the neurobiological correlates of speech motor sequence learning do
not differ across ANS and AWS.
DISCUSSION
The goal of the current study was to investigate the behavioral and neural correlates of speech
motor sequence learning in both stuttering and neurotypical adult speakers. Using a cued
sequence production task, we examined changes in the performance of pseudowords containing
non-native consonant clusters over time as a function of speech motor practice. At the behavioral
level, we found that, although AWS were less accurate and/or slower compared to ANS at
producing words involving non-native consonant clusters regardless of whether the words were
practiced, they showed improvements in these measures with practice that were statistically
comparable to those seen in ANS. At the neural level, we found that, across both groups and both
speech contrasts (novel non-native−practiced non-native and practiced non-native−practiced
native), practice producing words with non-native clusters led to significant decreases in BOLD
activity in brain areas implicated in verbal working memory and speech motor planning (dans-
cluding the left preSMA, FO, aINS, and bilateral IPS), suggesting that processing load and artic-
ulatory effort decrease as the nervous system forms regularized motor programs (or chunks) pour
coordinated sequences of vocal tract gestures. Critique, there were no reliable group differences
in either contrast map in any areas involved in the cortico-BG loops (or otherwise). En outre,
the degree of motor performance gains in accuracy were correlated positively with activity in the
left and right aINS. Collectively, these findings suggest that cortico-BG-mediated mechanisms
involved in learning novel phoneme sequences are not impaired in AWS; instead, the impairment
is primarily in the motor execution of speech sequences, novel and learned alike.
As in our prior study (Segawa, Tourville, et coll., 2015), we also observed pre- to post-training
BOLD activation changes in cortical areas that extend beyond those implicated in verbal working
memory and speech motor planning processes. Spécifiquement, we found greater activity in areas
related to auditory processes (the left PT, aSTG, and pSTG) during production of the novel non-
native sequences relative to the practiced non-native sequences (Chiffre 5). These auditory
regions are thought to be involved in guiding speech movements based on self-generated audi-
tory feedback (Guenther, 2016; Hickok, 2012). During speech production, activity in this area
has been reported to be greater when there is a mismatch between predicted and actual auditory
feedback (Tourville et al., 2008). Several existing theoretical models of speech production (par exemple.,
Guenther, 2016; Hickok, 2012) propose that error signals arising from these regions are used to
fine-tune speech motor programs over the course of repeated production attempts. Ainsi, learning
is thought to rely on the transmission of these auditory error signals to frontal regions involved in
motor planning and execution.
We also noted greater activity in cortical areas related to orthographic processing (the left ITO)
during production of the novel non-native sequences relative to the practiced non-native
sequences (Chiffre 5). Prior neuroimaging studies suggest that the ITO is a higher-level visual
processing area involved in identifying letters and words from lower-level shape images (voir,
par exemple., Pugh, Mencl, et coll., 2001) and is therefore highly likely to be related to viewing and decoding
the pseudowords that our participants were instructed to produce, rather than speech motor
control. In contrast, production of the practiced non-native sequences did not produce signifi-
cantly greater activity compared with the practiced native sequences in the ITO (Chiffre 6).
This difference between the condition-specific brain activations suggests that pseudowords
containing unfamiliar phonotactic sequences are more difficult to process prior to reading and
repetition practice (cf. Pugh, Frost, et coll., 2008).
Neurobiology of Language
130
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
e
d
toi
n
o
/
je
/
je
un
r
t
je
c
e
–
p
d
F
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
un
_
0
0
0
2
7
p
d
/
.
je
F
b
oui
g
toi
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
The finding that the right cerebellum (which is structurally and functionally connected with left
cortical areas) was recruited to a greater extent across both groups during the production of the
practiced non-native than the practiced native sequences (shown in Figure 6) was also observed
by Segawa, Tourville, et autres. (2015) in ANS, and corroborates other reports with neurotypical
participants that the cerebellum plays an important role in motor sequence learning and fine
motor control (par exemple., Ackermann, 2008; Doyon, Song, et coll., 2002; Guenther, 2016, Chapter 2).
Par exemple, Bohland and Guenther (2006) found that different regions of the cerebellum
showed differential sensitivity to syllable complexity (par exemple., “stra” vs. “ta”) and serial complexity
(par exemple., “da-da-da” vs. “da-ru-ti”) in ANS, suggesting involvement in speech sequencing. That said,
it is somewhat surprising that we did not find significant differences in cerebellar activity in the
novel non-native−practiced non-native contrast. Cependant, this comparison possessed less
statistical power because of more removed error trials.
An alternative, but not mutually exclusive, interpretation to the cortical activity differences we
found for both speech contrasts is that these differences result from the fact that the novel non-
native and practiced non-native sequences are more difficult to vocally imitate than the practiced
native sequences. Two sources of evidence support this conjecture. D'abord, prior neuroimaging
études (par exemple., Irwin et al., 2011) have reported that imitation of auditory speech produces addi-
tional significant activations in the left IFG and aINS when compared to a passive listening con-
dition. Deuxième, current computational models, such as the Directions Into Velocities of the
Articulators (DIVA) model (Guenther, 2016), propose that speech motor learning is driven by
initial mismatches between newly acquired sound targets and one’s own production attempts
as represented in the auditory cortex. Auditory error signals are then transformed into corrective
motor commands, and these corrective commands alter the feedforward commands for the next
production attempt. As the feedforward commands improve, fewer error signals are generated
and thus the contribution of the feedback control system gradually diminishes. The DIVA model
thus predicts decreases in BOLD activation in both motor planning and auditory cortical areas as
a consequence of imitation-based learning. In this view, the nervous system begins to form
speech motor programs via an imitation-based learning mechanism, which may account for
the increased activity in motor and auditory areas for the novel non-native compared with the
practiced non-native sequences, and also for the practiced non-native compared with the
practiced native sequences.
A number of other neuroimaging studies, compiled and discussed in a meta-analysis by Belyk
et coll., (2015), have reported that AWS tend to display higher activity in a number of right-
hemisphere regions during fluently produced native speech when compared to ANS. Some
researchers have proposed that this right-hemisphere cortical hyperactivity arises from impaired
left-hemisphere function (voir, par exemple., Belyk et al., 2015; Fox et al., 1996; Guenther, 2016; Neef et al.,
2017; cf. Connally et al., 2018). In view of these results, it is perhaps surprising that no clusters
emerged in the right hemisphere for AWS in the current study. Cependant, it is possible that right-
hemisphere hyperactivity in AWS may not occur in all speaking situations. En particulier, le
current speaking task likely required more attention and articulatory effort than those in prior
études, since the participants were required to produce non-native sequences of segments, lequel
is known to be difficult (par exemple., Davidson, 2006, 2010). En outre, there may be methodological
issues that concern the difference between the experimental procedures we used to test for a
functional activation difference between ANS and AWS and the ones used by other research
groupes, as the existing literature on stuttering encompasses a diverse set of sampling protocols
and motor tasks.
In the current study, it was further demonstrated that, in AWS, the in-scanner mean disfluency
covaried with BOLD responses in left-lateralized basal ganglia sites (the GPe and left caudate),
Neurobiology of Language
131
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
e
d
toi
n
o
/
je
/
je
un
r
t
je
c
e
–
p
d
F
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
un
_
0
0
0
2
7
p
d
/
.
je
F
b
oui
g
toi
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
such that the more disfluent participants were during the test phase, the less BOLD activity change
was observed in these basal ganglia sites for the novel non-native−practiced non-native contrast.
This negative correlation does not establish a causal relationship; rather it establishes an asso-
ciative link. Although this relationship is broadly consistent with the long-standing view that
stuttering reflects a malfunction within the cortico-BG loops (par exemple., Alm, 2004; Chang & Guenther,
2020; Connally et al., 2018; Fox et al., 1996; Giraud et al., 2008; Kell et al., 2009; Lu et al., 2010;
Watkins et al., 2008), it would appear to be at odds with other neuroimaging studies (albeit that
did not use a sparse-sampling method) which have reported positive relationships between
basal ganglia activity and stuttering severity (par exemple., Giraud et al., 2008; see also Metzger et al.,
2018, for a similar finding based on manual motor responses in AWS). Differences between the
findings of the current study and these prior studies suggest that the brain mechanisms underlying
general stuttering traits and transient disfluent states are dissociable (for further discussion, voir
Connally et al., 2018).
At first glance, the lack of a robust learning difference between the ANS and AWS groups may
appear to be at odds with other behavioral studies reporting motor learning deficits in AWS
(Ludlow et al., 1997; Namasivayam & van Lieshout, 2008; Smits-Bandstra et al., 2006). Several
important differences between those studies and ours might account for the divergent results.
D'abord, our study measured sequence and cluster learning using perception-based segmental
transcription and simple acoustic measures. Cependant, this approach required transcribers to make
categorical decisions regarding the segments that speakers produced, and thus did not permit
quantitative analysis of the presence or magnitude of various gestures, or of how speakers
continually updated their implementation of novel gestural scores. In contrast, other motor
learning studies (par exemple., Namasivayam & van Lieshout, 2008) used kinematic measures to com-
pare more subtle aspects of speech articulation between ANS and AWS. Ainsi, subtle differences
in the articulatory correlates of speech motor learning may exist between ANS and AWS.
Ongoing studies based on the current design that use electromagnetic articulography will
determine whether AWS and ANS differ in their ability to learn to execute and coordinate inter-
articulator movements for novel speech sequences.
Deuxième, our study focused on the learning of new phoneme sequences within a single syllable
with which the participants had no prior experience, since they violated English phonotactic
constraints. This task required the learning of new articulatory sequences at a motoric level. Dans
contraste, other studies in the literature on speech motor sequence learning in AWS have involved
production of more extended, multi-syllabic sequences that conform to English phonotactics
(par exemple., Namasivayam & van Lieshout, 2008). Cependant, there is evidence that multi-syllabic
sequences place higher demands on phonological working memory mechanisms than on motor
programming mechanisms. Par exemple, in another fMRI study, McGettigan et al. (2011) trouvé
that activity in the PT, a region implicated in auditory working memory, increased as the number
of syllables in either a non-word repetition or a passive listening task increased, whereas activity
in the left SMA, a region implicated in speech motor programming, increased as the number of
consonant clusters increased. Dans cette lumière, the increased error rates reported in other studies with
AWS might be interpreted instead as reflecting differences in phonological working memory
mechanisms rather than motor learning or implementation mechanisms (see also Yang et al.,
2019). Yet others have reported some differences between ANS and AWS in the degree of motor
adaptation following an experimental perturbation to the online sensory feedback (par exemple., Daliri
et coll., 2018), suggesting that mechanisms involved in updating existing motor programs may still
be impaired in stuttering.
It is also important to note that the present fMRI measures focused on the outcome of speech
motor sequence learning, not the online process of learning. Cependant, previous research spanning
Neurobiology of Language
132
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
e
d
toi
n
o
/
je
/
je
un
r
t
je
c
e
–
p
d
F
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
un
_
0
0
0
2
7
p
d
.
/
je
F
b
oui
g
toi
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
a wide range of learning tasks and paradigms has shown that participants may have similar learning
outcomes despite very different learning trajectories (see Karuza et al., 2014, for a review). Ainsi,
despite showing comparable BOLD activity patterns at test, ANS and AWS may not show evidence
of the same qualitative neural changes during the course of speech motor sequence training. Il
would therefore be of interest to conduct further fMRI studies, based on the current design, que
perform BOLD contrast analyses throughout both the training and the test phases.
Enfin, the current study focused primarily on the mechanisms of speech motor learning and
control, from the phonetic encoding stage down to the motor commands to the speech articula-
tors. Spécifiquement, we examined how ANS and AWS learn to transform discrete phonological
chunks (speech sounds that can be phonemes, syllables, or words) into a set of articulator move-
ments that achieves the intended auditory “target.” We employed monosyllabic pseudoword
stimuli to provide a “purer” measure of the speech motor system, as this would limit the recruit-
ment of higher-level cognitive and linguistic processing strategies to assist task performance (voir
also McGettigan et al., 2011). Cependant, any viable theory of speech production learning will
ultimately have to explicate how these phonological chunks are integrated with prosodic struc-
photos, as well as their relation to syntactic/semantic planning processes. Future studies will aim to
develop predictions from the highly controlled laboratory experiments presented here and else-
où (e.g.,Segawa, Masapollo, et coll., 2019; Segawa, Tourville, et coll., 2015) that may be tested
when speakers are instructed to produce novel speech sequences in a more natural context.
REMERCIEMENTS
We are grateful to Barbara Holland and Diane Constantino for assistance with participant recruit-
ment and/or data collection. We also thank Riccardo Falsini, Farwa Faheem, Abigail Cragin, Ariel
Gordon, Angelise Bulit, and Jessica Smith for help with data analysis and visualization. Enfin,
this work benefited from helpful discussions with, or comments from, Elaine Kearney, Megan
Thompson, Elizabeth Heller Murray, Jason Bohland, Cara Stepp, Kenneth Logan, and Tyler
Perrachionne. Research reported in this publication was supported by the National Institute on
Deafness and other Communication Disorders of the National Institutes of Health under award
number R01DC007683 (F. H. Guenther, PI). The content is solely the responsibility of the authors
and does not necessarily represent the official views of the National Institutes of Health.
INFORMATIONS SUR LE FINANCEMENT
Frank H. Guenther, National Institute on Deafness and Other Communication Disorders (http://
dx.doi.org/10.13039/100000055), Award ID: R01DC007683.
CONTRIBUTIONS DES AUTEURS
Matthew Masapollo: Visualisation; Analyse formelle; Rédaction – ébauche originale. Jennifer Segawa: Données
curation; Analyse formelle. Deryk S. Beal: Conservation des données; Analyse formelle. Jason A. Tourville:
Conceptualisation; Conservation des données; Analyse formelle, Rédaction – révision & édition. Alfonso Nieto-
Castañón: Analyse formelle; Rédaction – révision & édition. Matthias Heyne: Analyse formelle. Saul
UN. Frankford: Conservation des données; Analyse formelle. Frank H. Guenther: Conceptualisation; En écrivant -
revoir & édition.
RÉFÉRENCES
Ackermann, H. (2008). Cerebellar contributions to speech produc-
tion and speech perception: Psycholinguistic and neurobiological
perspectives. Trends in Neurosciences, 31(6), 265–272. EST CE QUE JE:
https://doi.org/10.1016/j.tins.2008.02.011, PMID: 18471906
Alexander, G. E., DeLong, M.. R., & Strick, P.. L. (1986). Parallel orga-
nization of functionally segregated circuits linking basal ganglia and
cortex. Revue annuelle des neurosciences, 9, 357–381. EST CE QUE JE: https://
doi.org/10.1146/annurev.ne.09.030186.002041, PMID: 3085570
Neurobiology of Language
133
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
e
d
toi
n
o
/
je
/
je
un
r
t
je
c
e
–
p
d
F
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
un
_
0
0
0
2
7
p
d
.
/
je
F
b
oui
g
toi
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
Alm, P.. UN. (2004). Stuttering and the basal ganglia circuits: A critical
review of possible relations. Journal of Communication Disorders,
37(4), 325–369. EST CE QUE JE: https://doi.org/10.1016/j.jcomdis.2004
.03.001, PMID: 15159193
Andersson, J.. L., Hutton, C., Ashburner, J., Tourneur, R., & Friston, K.
(2001). Modeling geometric deformations in EPI time series.
NeuroImage, 13(5), 903–919. EST CE QUE JE: https://doi.org/10.1006/nimg
.2001.0746, PMID: 11304086
Ashburner, J., & Friston, K. J.. (2005). Unified segmentation.
NeuroImage, 26(3), 839–851. EST CE QUE JE: https://est ce que je.org/10.1016
/j.neuroimage.2005.02.018, PMID: 15955494
Belin, P., Zatorre, R.. J., Hoge, R., Evans, UN. C., & Pike, B. (1999).
Event-related fMRI of the auditory cortex. NeuroImage, 10(4), 417–429.
EST CE QUE JE: https://doi.org/10.1006/nimg.1999.0480, PMID: 10493900
Belyk, M., Kraft, S. J., & Brun, S. (2015). Stuttering as a trait or
state—an ALE meta-analysis of neuroimaging studies. européen
Journal des neurosciences, 41(2), 275–284. EST CE QUE JE: https://est ce que je.org/10
.1111/ejn.12765, PMID: 25350867
Berent, JE., Steriade, D., Lennertz, T., & Vaknin, V. (2007). Ce que nous
know about what we have never heard: Evidence from perceptual
illusions. Cognition, 104(3), 591–630. EST CE QUE JE: https://est ce que je.org/10
.1016/j.cognition.2006.05.015, PMID: 16934244
Bloodstein, O., & Ratner, N. B. (2008). A handbook on stuttering.
Thomson/Delmar Learning.
Bohland, J.. W., Bullock, D., & Guenther, F. H. (2010). Neural repre-
sentations and mechanisms for the performance of simple speech
sequences. Journal des neurosciences cognitives, 22(7). EST CE QUE JE: https://
doi.org/10.1162/jocn.2009.21306, PMID: 19583476, PMCID:
PMC2937837
Bohland, J.. W., & Guenther, F. H. (2006). An fMRI investigation of
syllable sequence production. NeuroImage, 32, 821–841. EST CE QUE JE:
https://doi.org/10.1016/j.neuroimage.2006.04.173, PMID:
16730195
Boersma, P., & Weenink, D. (2020). Praat: Doing phonetics by
computer ( Version 6.1.09) [Computer program]. Retrieved 26
Janvier 2020 from https://www.praat.org/.
Boutsen, F. (1993). A comparative study of stress timing of stutterers
and nonstutterers. Journal of Fluency Disorders, 20(2), 145–155.
EST CE QUE JE: https://doi.org/10.1016/0094-730X(94)00023-M.
Brawn, T. P., Fenn, K. M., Nusbaum, H. C., & Margoliash, D.
(2010). Consolidating the effects of waking and sleep on motor-
sequence learning. Journal des neurosciences, 30(42), 13977–13982.
EST CE QUE JE: https://doi.org/10.1523/JNEUROSCI.3295-10.2010, PMID:
20962219, PMCID: PMC2978076
Buchwald, UN., Calhoun, H., Rimikis, S., Steinberg Lowe, M.,
Wellner, R., & Edwards, D. (2019). Using tDCS to facilitate motor
learning in speech production: The role of timing. Cortex, 111,
274–285. EST CE QUE JE: https://doi.org/10.1016/j.cortex.2018.11.014,
PMID: 30551048, PMCID: PMC6358507
Cai, S., Tourville, J.. UN., Beal, D. S., Perkell, J.. S., Guenther, F. H., &
Ghosh, S. S. (2014). Diffusion imaging of cerebral white matter in
persons who stutter: Evidence for network-level anomalies.
Frontiers in Human Neuroscience, 8(54). EST CE QUE JE: https://doi.org
/10.3389/fnhum.2014.00054, PMID: 24611042, PMCID:
PMC3920071
Chang, S.-E., & Guenther, F. H. (2020). Involvement of the cortico-
basal-ganglia-thalamocortical loop in developmental stuttering.
Frontiers in Psychology, 10, 1–15. EST CE QUE JE: https://doi.org/10.3389
/fpsyg.2019.03088, PMID: 32047456, PMCID: PMC6997432
Chang, S.-E., Ohde, R.. N., & Conture, E. G. (2002). Coarticulation
and formant transition rate in young children who stutter. Journal
of Speech, Language, and Hearing Research, 45(4), 676–688.
EST CE QUE JE: https://doi.org/10.1044/1092-4388(2002/054)
Cholin, J., Levelt, W. J.. M., & Schiller, N. Ô. (2006). Effects of syllable
frequency in speech production. Cognition, 99(2), 205–235. EST CE QUE JE:
https://doi.org/10.1016/j.cognition.2005.01.009, PMID:
15939415
Civier, O., Bullock, D., Max, L., & Guenther, F. H. (2013).
Computational modeling of stuttering caused by impairments
in a basal ganglia thalamocortical circuit involved in syllable
selection and initiation. Brain and Language, 126(3), 263–278.
EST CE QUE JE: https://doi.org/10.1016/j.bandl.2013.05.016, PMID:
23872286, PMCID: PMC3775364
Connally, E. L., Ward, D., Pliatsikas, C., Finnegan, S., Jenkinson,
M., Boyles, R., & Watkins, K. E. (2018). Separation of trait and
state in stuttering. Human Brain Mapping, 39(8), 3109–3126.
EST CE QUE JE: https://doi.org/10.1002/ hbm.24063, PMID: 29624772,
PMCID: PMC6055715
Coltheart, M.. (1981). The MRC Psycholinguistic Database. Le
Quarterly Journal of Experimental Psychology Section A, 33(4),
497–505. EST CE QUE JE: https://doi.org/10.1080/14640748108400805
Craig-McQuaide, UN., Akram, H., Zrinzo, L., & Tripoliti, E. (2014). UN
review of brain circuitries involved in stuttering. Frontiers in
Human Neuroscience. EST CE QUE JE: https://doi.org/10.3389/fnhum
.2014.00884, PMID: 25452719, PMCID: PMC4233907
Dale, UN. M., Fischl, B., & Séréno, M.. je. (1999). Cortical surface-based
analysis—I. Segmentation and surface reconstruction. NeuroImage,
9(2), 179–194. EST CE QUE JE: https://doi.org/10.1006/nimg.1998.0395,
PMID: 9931268
Daliri, UN., Wieland, E. UN., Cai, S., Guenther, F. H., & Chang, S. E.
(2018). Auditory-motor adaptation is reduced in adults who stutter
but not in children who stutter. Developmental Science, 21(2),
e12521. EST CE QUE JE: https://doi.org/10.1111/desc.12521, PMID:
28256029, PMCID: PMC5581739
Davidson, L. (2006). Phonology, phonetics, or frequency:
Influences on the production of non-native sequences. Journal
of Phonetics, 34, 104–137. EST CE QUE JE: https://doi.org/10.1016/j.wocn
.2005.03.004
Davidson, L. (2010). Phonetic bases of similarities in cross-language
production: Evidence from English and Catalan. Journal de
Phonetics, 38(2), 272–288. EST CE QUE JE: https://doi.org/10.1016/j.wocn
.2010.01.001
Davidson, L., & Shaw, J.. (2012). Sources of illusion in consonant
cluster perception. Journal of Phonetics, 40(2), 234–248. EST CE QUE JE:
https://doi.org/10.1016/j.wocn.2011.11.005
Diedrichsen, J., Balsters, J.. H., Flavell, J., Cussans, E., & Ramnani, N.
(2009). A probabilistic MR atlas of the human cerebellum.
NeuroImage, 15(1), 39–46. EST CE QUE JE: https://doi.org/10.1016/j.neuroimage
.2009.01.045, PMID: 19457380
Doyon, J., Albouy, G., Vahdat, S., & King, B. (2015). Neural correlates
of motor skill acquisition and consolidation. In A. Toga (Ed.), Cerveau
mapping: An encyclopedic reference. Elsevier. EST CE QUE JE: https://est ce que je
.org/10.1016/B978-0-12-397025-1.00275-X
Doyon, J., Bellec, P., Amsel, R., Penhune, V., Monchi, O., Carrier,
J., Lehericy, S., & Benali, H. (2009). Contributions of the basal
ganglia and functionally related brain structures to motor learning.
Behavioral Brain Research, 199(1), 61–75. EST CE QUE JE: https://est ce que je.org/10
.1016/j.bbr.2008.11.012, PMID: 19061920
Doyon, J., Song, UN. W., Karni, UN., Lalonde, F., Adams, M.. M., &
Ungerleider, L. G. (2002). Experience-dependent changes in
cerebellar contributions to motor sequence learning. Procédure
of the National Academy of Sciences, 99(2), 1017–1022. EST CE QUE JE:
https://doi.org/10.1073/pnas.022615199, PMID: 11805340,
PMCID: PMC117423
Dupoux, E., Kakehi, K., Pallier, Y., Hirose, C., & Mehler, J.. (1999).
Epenthetic vowels in Japanese: A perceptual illusion? Journal de
Neurobiology of Language
134
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
e
d
toi
n
o
/
je
/
je
un
r
t
je
c
e
–
p
d
F
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
un
_
0
0
0
2
7
p
d
/
.
je
F
b
oui
g
toi
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
Experimental Psychology: Perception humaine et performance,
25(6), 1568–1578. EST CE QUE JE: https://doi.org/10.1037/0096-1523.25
.6.1568
Dupoux, E., Parlato, E., Frota, S., Hirose, Y., & Peperkamp, S. (2011).
Where do illusory vowels come from? Journal of Memory and
Language, 64(3), 199–210. EST CE QUE JE: https://doi.org/10.1016/j.jml
.2010.12.004
Fee, M.. S., & Goldberg, J.. H. (2011). A hypothesis for basal ganglia
dependent reinforcement learning in the songbird. Neurosciences,
198, 152–170. EST CE QUE JE: https://doi.org/10.1016/j.neuroscience
.2011.09.069, PMID: 22015923, PMCID: PMC3221789
Fenn, K. M., Nusbaum, H. C., & Margoliash, D. (2003).
Consolidation during sleep of perceptual learning of spoken
langue. Nature, 425, 614–616. EST CE QUE JE: https://est ce que je.org/10.1038
/nature01951, PMID: 14534586
Ferraro, F. R., Balota, D. UN., & Connor, L. T. (1993). Implicit memory
and the formation of new associations in nondemented Parkinson’s
disease individuals and individuals with senile dementia of the
Alzheimer type: A serial reaction time (SRT) enquête. Cerveau
et cognitif, 21(2), 163–180. EST CE QUE JE: https://doi.org/10.1006/brcg
.1993.1013, PMID: 8442933
Fischl, B., Salat, D. H., Busa, E., Albert, M., Dieterich, M.,
Haselgrove, C., van der Kouwe, UN., Killiany, R., Kennedy, D.,
Klaveness, S., Montillo, UN., Makris, N., Rosen, B., & Dale, UN. M..
(2002). Whole brain segmentation: Automated labeling of neuro-
anatomical structures in the human brain. Neurone, 33(3), 341–355.
EST CE QUE JE: https://doi.org/10.1016/S0896-6273(02)00569-X
Fischl, B., Séréno, M.. JE., & Dale, UN. M.. (1999). Cortical surface-based
analysis—II: Inflation, flattening, and a surface-based coordinate
système. NeuroImage, 9(2), 195–207. EST CE QUE JE: https://est ce que je.org/10
.1006/nimg.1998.0396, PMID: 9931269
Fox, P.. T., Ingham, R.. J., Ingham, J.. C., Hirsch, T. B., Downs, J.. H.,
Martine, C., Jerabek, P., Verre, T., & Lancaster, J.. L. (1996). A PET study
of the neural systems of stuttering. Nature, 382(6587), 158–161.
EST CE QUE JE: https://doi.org/10.1038/382158a0, PMID: 8700204
Giraud, A.-L., Neumann, K., Bachoud-Levi, A.-C., von Gudenberg,
UN. W., Euler, H. UN., Lanfermann, H., & Preibisch, C. (2008).
Severity of dysfluency correlates with basal ganglia activity in
persistent developmental stuttering. Brain and Language, 104(2),
190–199. EST CE QUE JE: https://doi.org/10.1016/j.bandl.2007.04.005,
PMID: 17531310
Gracco, V. L., Tremblay, P., & Pike, G. B. (2005). Imaging speech
production using fMRI. NeuroImage, 26, 294–301. EST CE QUE JE: https://
doi.org/10.1016/j.neuroimage.2005.01.033, PMID: 15862230
Guenther, F. H. (2016). Neural control of speech. AVEC Presse. EST CE QUE JE:
https://doi.org/10.7551/mitpress/10471.001.0001
Hagler, D. J., Jr., Saygin, UN. P., & Séréno, M.. je. (2006). Smoothing
and cluster thresholding for cortical surface-based group analysis
of fMRI data. NeuroImage, 33(4), 1093–1103. EST CE QUE JE: https://doi.org
/10.1016/j.neuroimage.2006.07.036, PMID: 17011792, PMCID:
PMC1785301
Helmuth, L., Mayr, U., & Daum, je. (2000). Sequence learning in
Parkinson’s disease: A comparison of spatial-attention and number-
response sequences. Neuropsychologie, 38, 1443–1451. EST CE QUE JE:
https://doi.org/10.1016/S0028-3932(00)00059-2
Hickok, G. (2012). Computational neuroanatomy of speech pro-
duction. Nature Revues Neurosciences, 13(2), 135–145. EST CE QUE JE:
https://doi.org/10.1038/nrn3158, PMID: 22218206, PMCID:
PMC5367153
Ingham, R.. J., Grafton, S. T., Bothe, UN. K., & Ingham, J.. C. (2012).
Brain activity in adults who stutter: Similarities across speaking
tasks and correlations with stuttering frequency and speaking
rate. Brain and Language, 122(1), 11–24. EST CE QUE JE: https://doi.org
/10.1016/j.bandl.2012.04.002, PMID: 22564749, PMCID:
PMC3372660
Irwin, J.. R., Frost, S. J., Mencl, E. W., Chen, H., & Fowler, C. UN.
(2011). Functional activation for imitation of seen and heard
speech. Journal of Neurolinguistics, 24(6), 611–618. EST CE QUE JE: https://
doi.org/10.1016/j.jneuroling.2011.05.001, PMID: 21966094,
PMCID: PMC3182484
Jackson, G. M., Jackson, S. R., Harrison, J., Henderson, L., & Kennard,
C. (1995). Serial reaction time learning in Parkinson’s disease:
Evidence for a procedural learning deficit. Neuropsychologie, 33(5),
577–593. EST CE QUE JE: https://doi.org/10.1016/0028-3932(95)00010-Z
Jezzard, P., & Balaban, R.. S. (1995). Correction for geometric distor-
tion in echo planar images from B0 field variations. Magnetic
Resonance in Medicine, 34(1), 65–73. EST CE QUE JE: https://est ce que je.org/10
.1002/mrm.1910340111, PMID: 7674900
Karuza, E. UN., Emberson, L. L., & Aslin, R.. N. (2014). Combining
fMRI and behavioral measures to examine the process of human
learning. Neurobiology of Learning and Memory, 109, 193–206.
EST CE QUE JE: https://doi.org/10.1016/j.nlm.2013.09.012, PMID: 24076012,
PMCID: PMC3963805
Kell, C. UN., Neumann, K., von Kriegstein, K., Posenenske, C., von
Gudenberg, UN. W., Euler, H., & Giraud, A.-L. L. (2009). How the
brain repairs stuttering. Cerveau, 132(10), 2747–2760. EST CE QUE JE: https://
doi.org/10.1093/brain/awp185, PMID: 19710179
Keuken, M.. C., Bazin, P.-L., Crown, L., Hootsmans, J., Laufer, UN.,
Müller-Axt, C., Sier, R., van der Putten, E. J., Schafer, UN., Tourneur, R.,
& Forstmann, B. U. (2014). Quantifying inter-individual anatomical
variability in the subcortex using 7T structural MRI. NeuroImage,
94, 40–46. EST CE QUE JE: https://doi.org/10.1016/j.neuroimage.2014
.03.032, PMID: 24650599
Kotz, S. UN., & Schwartze, M.. (2010). Cortical speech processing
unplugged: A timely subcortico-cortical framework. Trends in
Sciences cognitives, 14(9), 392–399. EST CE QUE JE: https://est ce que je.org/10.1016
/j.tics.2010.06.005, PMID: 20655802
Lu, C., Chen, C., Ning, N., Ding, G., Guo, T., Peng, D., Lequel, Y., Li,
K., & Lin, C. (2010). The neural substrates for atypical planning
and execution of word production in stuttering. Experimental
Neurologie, 221(1), 146–156. EST CE QUE JE: https://est ce que je.org/10.1016/j
.expneurol.2009.10.016, PMID: 19879262
Ludlow, C. L., Siren, K., & Zikria, M.. (1997). Speech production
learning in adults with chronic developmental stuttering. In W.
Hujstijn, H. F. M.. Peters, & P.. H. H. M.. van Lieshout (Éd.),
Speech production: Motor control, brain research and fluency
disorders (pp. 212–229). Elsevier Science Publishers.
Max, L., Guenther, F. H., Gracco, V. L., Ghosh, S. S., & Wallace,
M.. E. (2004). Unstable or insufficiently activated internal models
and feedback-biased motor control as sources of dysfluency: UN
theoretical model of stuttering. Contemporary Issues in
Communication Science and Disorders, 31, 105–122. EST CE QUE JE:
https://doi.org/10.1044/cicsd_31_S_105
McGettigan, C., Warren, J.. E., Eisner, F., Maréchal, C. R.,
Shanmugalingam, P., & Scott, S. K. (2011). Neural correlates of
sublexical processing in phonological working memory. Journal de
Neurosciences cognitives, 23(4), 961–977. EST CE QUE JE: https://doi.org
/10.1162/jocn.2010.21491, PMID: 20350182, PMCID: PMC3376447
Metzger, F. L., Auer, T., Helms, G., Paulus, W., Frahm, J., Sommer,
M., & Neef, N. E. (2018). Shifted dynamic interactions between
subcortical nuclei and inferior frontal gyri during response prep-
aration in persistent developmental stuttering. Brain Structure
and Function, 223, 165–182. EST CE QUE JE: https://doi.org/10.1007
/s00429-017-1476-1, PMID: 28741037, PMCID: PMC5772149
Mollaei, F., Shiller, D. M., & Gracco, V. L. (2013). Sensorimotor adap-
tation of speech in Parkinson’s disease. Journal of Movement
Neurobiology of Language
135
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
e
d
toi
n
o
/
je
/
je
un
r
t
je
c
e
–
p
d
F
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
un
_
0
0
0
2
7
p
d
/
.
je
F
b
oui
g
toi
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
Disorders, 28(12), 1668–1674. EST CE QUE JE: https://doi.org/10.1002/mds
.25588, PMID: 23861349, PMCID: PMC3812368
Nakamura, K., Sakai, K., & Hikosaka, Ô. (1998). Neuronal activity
in medial frontal cortex during learning of sequential procedures.
Journal de neurophysiologie, 80(5), 2671–2687. EST CE QUE JE: https://est ce que je
.org/10.1152/jn.1998.80.5.2671, PMID: 9819272
Namasivayam, UN. K., & Van Lieshout, P.. H. H. M.. (2008). Bite-
block perturbation in people who stutter: Immediate compensatory
and delayed adaptive processes. Journal of Communication
Disorders, 41(4), 372–294. EST CE QUE JE: https://doi.org/10.1016/j.jcomdis
.2008.02.004, PMID: 18405914
Neef, N. E., Anwander, UN., Butfering, C., Schmidt-Samoa, C.,
Friederici, UN. D., Paulus, W., & Sommer, M.. (2017). Structural
connectivity of right frontal hyperactive areas scales with stuttering
severity. Cerveau, 141(1), 191–204. EST CE QUE JE: https://est ce que je.org/10.1093
/brain/awx316, PMID: 29228195, PMCID: PMC5837552
Niethammer, M., Feigin, UN., & Eidelber, D. (2012). Functional neuro-
imaging in Parkinson’s disease. Cold Spring Harbor Perspectives
in Medicine, 2(5), a009274. EST CE QUE JE: https://doi.org/10.1101
/cshperspect.a009274, PMID: 22553499, PMCID: PMC3331691
Nieto-Castañón, UN. (2020). Handbook of functional connectivity
magnetic resonance imaging methods in CONN. Hilbert Press.
Nieto-Castañón, UN., Ghosh, S. S., Tourville, J.. UN., & Guenther, F. H.
(2003). Region of interest based analysis of functional imaging
data. Neuroimage, 19(4), 1303–1316. EST CE QUE JE: https://est ce que je.org/10
.1016/S1053-8119(03)00188-5
Oldfield, R.. C. (1971). The assessment and analysis of handedness:
The Edinburgh inventory. Neuropsychologie, 9(1), 97–113. EST CE QUE JE:
https://doi.org/10.1016/0028-3932(71)90067-4
Pan, S. C., & Rickard, T. C. (2015). Sleep and motor learning: Is
there room for consolidation? Psychological Bulletin, 141(4),
812–834. EST CE QUE JE: https://doi.org/10.1037/ bul0000009, PMID:
25822130
Perrachione, T. K., & Ghosh, S. S. (2013). Optimized design and anal-
ysis of sparse-sampling fMRI experiments. Frontiers in Neuroscience,
18(7). EST CE QUE JE: https://doi.org/10.3389/fnins.2013.00055, PMID:
23616742, PMCID: PMC3629333
Pitt, M.. UN. (1998). Phonological processes and the perception of pho-
notactically illegal consonant clusters. Perception & Psychophysics,
60(6), 941–951. EST CE QUE JE: https://doi.org/10.3758/BF03211930, PMID:
9718954
Pugh, K. R., Frost, S. F., Sandak, R., Landi, N., Rueckl, J.. G.,
Constable, R.. T., Seidenberg, M., Fulbright, R., Katz, L., &
Mencl, W. E. (2008). Effects of stimulus difficulty and repetition
on printed word identification: An fMRI comparison of non-
impaired and reading disabled adolescent cohorts. Journal de
Neurosciences cognitives, 20(7), 1146–1160. EST CE QUE JE: https://doi.org
/10.1162/jocn.2008.20079, PMID: 18284344, PMCID:
PMC3152957
Pugh, K. R., Mencl, W. E., Jenner, UN. R., Lee, J.. R., Katz, L., Frost, S. J.,
Shaywitz, S. E., & Shaywitz, B. UN. (2001). Neurobiological studies
of reading and reading disability. Journal of Communicable
Diseases, 34(6), 479–492. EST CE QUE JE: https://doi.org/10.1016/S0021
-9924(01)00060-0
Rauschecker, UN. M., Pringle, UN., & Watkins, K. E. (2008). Changes
in neural activity associated with learning to articulate novel au-
ditory pseudowords by covert repetition. Human Brain Mapping,
29(11), 1231–1242. EST CE QUE JE: https://doi.org/10.1002/ hbm.20460,
PMID: 17948887, PMCID: PMC6870739
Riley, G. D. (2009). Stuttering severity instrument for children and
adultes (SSI-4) (4th ed.). Pro-Ed.
Robb, M., & Blomgren, M.. (1997). Analysis of F2 transitions in the
speech of stutterers and nonstutterers. Journal of Fluency
Disorders, 22, 1–16. EST CE QUE JE: https://doi.org/10.1016/S0094-730X
(96)00016-2
Robb, M., Blomgren, M., & Chen, Oui. (1998). Formant frequency
fluctuation in stuttering and nonstuttering adults. Journal de
Fluency Disorders, 23, 73–84. EST CE QUE JE: https://est ce que je.org/10.1016
/S0094-730X(97)00029-6
Rottschy, C., Langner, R., Dogan, JE., Reetz, K., Laird, UN. R., Schulz,
J.. B., & Eickhoff, S. B. (2012). Modelling neural correlates of
working memory: A coordinate-based meta-analysis. NeuroImage,
60, 830–846. EST CE QUE JE: https://doi.org/10.1016/j.neuroimage
.2011.11.050, PMID: 22178808, PMCID: PMC3288533
Sapir, S. (2014). Multiple factors are involved in the dysarthria
associated with Parkinson’s disease: A review with implications
for clinical practice and research. Journal of Speech and Hearing
Research, 57(4), 1330–1343. EST CE QUE JE: https://doi.org/10.1044/2014
_JSLHR-S-13-0039, PMID: 24686571
Segawa, J., Masapollo, M., Tong, M., Forgeron, D.J., & Guenther, F. H.
(2019). Chunking of phonological units in speech sequencing.
Brain and Language, 195, 104636. EST CE QUE JE: https://est ce que je.org/10.1016
/j.bandl.2019.05.001, PMID: 31202179, PMCID: PMC6686190
Segawa, J., Tourville, J.. UN., Beal, D. S., & Guenther, F. H. (2015).
The neural correlates of speech motor sequence learning. Journal
of Cognitive Neuroscience, 27(4), 819–831. EST CE QUE JE: https://doi.org
/10.1162/jocn_a_00737, PMID: 25313656, PMCID: PMC4344924
Skodda, S. (2012). Effect of deep brain stimulation on speech perfor-
mance in Parkinson’s disease. Parkinson’s Disease, 2012, 850596.
EST CE QUE JE: https://doi.org/10.1155/2012/850596, PMID: 23227426,
PMCID: PMC3512320
Forgeron, S. M., & Nichols, T. E. (2009). Threshold-free cluster enhance-
ment: Addressing problems of smoothing, threshold dependence
and localisation in cluster inference. NeuroImage, 44(1), 83–98.
EST CE QUE JE: https://doi.org/10.1016/j.neuroimage.2008.03.061, PMID:
18501637
Smits-Bandstra, S., De Nil, L. F., & Saint-Cyr, J.. (2006). Speech and
nonspeech sequence skill learning in adults who stutter. Journal
of Fluency Disorders, 31(2), 116–136. EST CE QUE JE: https://est ce que je.org/10
.1016/j.jfludis.2006.04.003, PMID: 16735062
Smits-Bandstra, S., & Gracco, V. (2013). Verbal implicit sequence
learning in persons who stutter and persons with Parkinson’s
maladie. Journal of Motor Behavior, 45(5), 381–393. EST CE QUE JE: https://
doi.org/10.1080/00222895.2013.812058, PMID: 23844763,
PMCID: PMC4210384
Sternberg, S., Monsell, S., Knoll, R.. L., & Wright, C. E. (1978). The latency
and duration of rapid movement sequences: Comparisons of speech
and typing. In G. E. Stelmach (Ed.), Information processing in motor
control and learning (pp. 117–152). Academic Press. EST CE QUE JE: https://
doi.org/10.1016/B978-0-12-665960-3.50011-6
Tourville, J.. UN., & Guenther, F. H. (2003). A cortical and cerebellar
parcellation system for speech studies (Boston University Technical
Report CAS/CNS-03-022). Université de Boston.
Tourville, J.. UN., Reilly, K. J., & Guenther, F. H. (2008). Neural mecha-
nisms underlying auditory feedback control of speech. NeuroImage,
39(3), 1429–1443. EST CE QUE JE: https://doi.org/10.1016/j.neuroimage
.2007.09.054, PMID: 18035557, PMCID: PMC3658624
Toyomura, UN., Fujii, T., & Kuriki, S. (2015). Effect of an 8-week
practice of externally triggered speech on basal ganglia activity
of stuttering and fluent speakers. NeuroImage, 109, 458–468.
EST CE QUE JE: https://doi.org/10.1016/j.neuroimage.2015.01.024, PMID:
25595501
Vahdat, S., Fogel, S., Benali, H., & Doyon, J.. (2017). Network-wide
reorganization of procedural memory during NREM sleep revealed
by fMRI. eLife, 11, e24987. EST CE QUE JE: https://doi.org/10.7554/eLife
.24987
Neurobiology of Language
136
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
e
d
toi
n
o
/
je
/
je
un
r
t
je
c
e
–
p
d
F
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
un
_
0
0
0
2
7
p
d
/
.
je
F
b
oui
g
toi
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neural correlates of speech motor sequence learning
Vakil, E., Kahan, S., Huberman, M., & Osimani, UN. (2000). Motor
and non-motor sequence learning in patients with basal ganglia
lesions: The case of serial reaction time (SRT). Neuropsychologie,
38, 1-dix. EST CE QUE JE: https://doi.org/10.1016/S0028-3932(99)00058-5
Watkins, K. E., Forgeron, S. M., Davis, S., & Howell, P.. (2008).
Structural and functional abnormalities of the motor system in
developmental stuttering. Cerveau, 131, 50–59. EST CE QUE JE: https://est ce que je
.org/10.1093/ brain/awm241, PMID: 17928317, PMCID:
PMC2492392
Whitfield-Gabrieli, S., & Nieto-Castañón, UN. (2012). CONN: UN
functional connectivity toolbox for correlated and anticorrelated
brain networks. Brain Connectivity, 2(3), 125–141. EST CE QUE JE: https://
doi.org/10.1089/brain.2012.0073, PMID: 22642651
Wiener, N. (1949). Extrapolation, interpolation, and smoothing of
stationary time series. AVEC Presse. EST CE QUE JE: https://doi.org/10.7551
/mitpress/2946.001.0001
Lequel, Y., Jia, F., Fox, P.. T., Siok, W., & Tan, L. H. (2019). Abnormal
neural response to phonological working memory demands in
persistent developmental stuttering. Human Brain Mapping, 40(1),
214–225. EST CE QUE JE: https://doi.org/10.1002/ hbm.24366, PMID:
30145850, PMCID: PMC6865627
Yaruss, J.. S., & Conture, E. G. (1993). F2 transitions during sound/
syllable repetitions of children who stutter and predictions of
stuttering chronicity. Journal of Speech, Language, and Hearing
Research, 36(5), 883–896. EST CE QUE JE: https://doi.org/10.1044/jshr
.3605.883, PMID: 8246477
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
e
d
toi
n
o
/
je
/
je
un
r
t
je
c
e
–
p
d
F
/
/
/
/
2
1
1
0
6
1
8
9
9
6
7
7
n
o
_
un
_
0
0
0
2
7
p
d
.
/
je
F
b
oui
g
toi
e
s
t
t
o
n
0
8
S
e
p
e
m
b
e
r
2
0
2
3
Neurobiology of Language
137