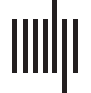RECHERCHE
Characterizing heterogeneity in early adolescent
reward networks and individualized associations
with behavioral and clinical outcomes
Matthew Mattoni
, David V. Forgeron, and Thomas M. Olino
Department of Psychology and Neuroscience, Temple University, Philadelphia, Pennsylvanie, Etats-Unis
Mots clés: Effective connectivity, Heterogeneity, Adolescence, Reward, Depression, Substance use
un accès ouvert
journal
ABSTRAIT
Associations between connectivity networks and behavioral outcomes such as depression are
typically examined by comparing average networks between known groups. Cependant, neural
heterogeneity within groups may limit the ability to make inferences at the individual level as
qualitatively distinct processes across individuals may be obscured in group averages. Ce
study characterizes the heterogeneity of effective connectivity reward networks among 103
early adolescents and examines associations between individualized features and multiple
behavioral and clinical outcomes. To characterize network heterogeneity, we used extended
unified structural equation modeling to identify effective connectivity networks for each
individual and an aggregate network. We found that an aggregate reward network was a poor
representation of individuals, with most individual-level networks sharing less than 50% of the
group-level network paths. We then used Group Iterative Multiple Model Estimation to identify
a group-level network, subgroups of individuals with similar networks, and individual-level
réseaux. We identified three subgroups that appear to reflect differences in network maturity,
but this solution had modest validity. Enfin, we found numerous associations between
individual-specific connectivity features and behavioral reward functioning and risk for
substance use disorders. We suggest that accounting for heterogeneity is necessary to use
connectivity networks for inferences precise to the individual.
RÉSUMÉ DE L'AUTEUR
Associations between brain connectivity and behavioral phenotypes are typically examined
by comparing group averages (par exemple., clinical vs. control). This approach presumes that the
group-average network reflects individuals. Cependant, if individuals’ network structures are
highly heterogenous, averaging across them will create a group-level network that does not
generalize to individuals, preventing valid inferences of associations with behavioral
differences. Ici, we showed that a group-level reward network in early adolescents poorly
reflected individuals. We then used GIMME to identify reward network features that were
specific to individuals. These network features were associated with multiple reward-related
résultats, including familial risk for substance use disorder (R2 = 31%). A focus on the
individual, rather than groups, may be necessary for valid inferences of individual behavioral
differences from fMRI connectivity.
Citation: Mattoni, M., Forgeron, D. V., &
Olino, T. M.. (2023). Characterizing
heterogeneity in early adolescent
reward networks and individualized
associations with behavioral and
clinical outcomes. Réseau
Neurosciences, 7(2), 787–810. https://est ce que je
.org/10.1162/netn_a_00306
EST CE QUE JE:
https://doi.org/10.1162/netn_a_00306
Informations complémentaires:
https://doi.org/10.1162/netn_a_00306
Reçu: 8 Septembre 2022
Accepté: 6 Janvier 2023
Intérêts concurrents: Les auteurs ont
a déclaré qu'aucun intérêt concurrent
exister.
Auteur correspondant:
Matthew Mattoni
matt.mattoni@temple.edu
Éditeur de manipulation:
Olaf Sporns
droits d'auteur: © 2023
Massachusetts Institute of Technology
Publié sous Creative Commons
Attribution 4.0 International
(CC PAR 4.0) Licence
La presse du MIT
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
/
t
/
e
d
toi
n
e
n
un
r
t
je
c
e
–
p
d
je
F
/
/
/
/
/
7
2
7
8
7
2
1
1
8
4
4
9
n
e
n
_
un
_
0
0
3
0
6
p
d
.
t
F
b
oui
g
toi
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
Adolescent reward network heterogeneity
Reward network:
Connectivity between brain regions
associated with approach
motivation, wanting, and positive
emotions related to anticipating or
consuming a reward.
Heterogeneity:
Qualitative differences in
connectivity network patterns that
are not attributable to random error
and result in distinct network
structures.
Effective connectivity:
Hypothesized underlying directional
network that models how activation
in one region influences others to
explain the observed correlations in
BOLD activity.
Ergodicity:
The ability to generalize group-level
statistics (par exemple., averages) à
individuals.
INTRODUCTION
Adolescent reward network processing is associated with several health-related outcomes,
including risk-taking behaviors, depression, and substance use problems (Casey et al., 2008).
Research has traditionally compared aggregate network models between known groups, tel
as individuals with and without target disorders. This approach implicitly assumes that each
group is a homogenous population such that individuals can be represented by a single aggre-
gate network. Cependant, there is increasing evidence that there is substantial heterogeneity in
neural networks within groups that are defined by a single behavioral phenotype (Drysdale
et coll., 2017; Feczko & Fair, 2020; Marquand et al., 2016; Price et al., 2017b). This heteroge-
neity may result in a group-average model that is not representative of some, or all, individuals,
limiting the ability to use a case-controlled framework to make inferences at the individual
level. Alternativement, estimating networks that are more precise to the individual can improve
the ability to make inferences specific to the individual, rather than the group. Dans cette étude, nous
parse heterogeneity in adolescent effective connectivity reward networks using multiple
methods and examine associations between individual-specific network features and
reward-related behavioral outcomes. D'abord, we examine the applicability of the aggregate net-
work structure to that for each individual. Deuxième, we derive subgroups of participants who
share similar connectivity network features. Enfin, we examine how features of networks,
including subgroups and individual connectivity paths, are associated with outcomes including
reward-related behaviors, depression, and risk for alcohol and substance use problems.
Case-controlled study designs can provide insight into broad neural differences between
groupes. Cependant, translational utility of neuroimaging (par exemple., diagnosis, treatment selection,
etc.) ultimately requires that models exhibit group-to-individual generalizability. Model
group-to-individual generalizability is termed ergodicity, and requires that individuals conform
to a similar model (Fisher et al., 2018; Molenaar, 2004). Early examinations of neural networks
have found that individuals exhibit qualitatively distinct traitlike patterns that are not reflected
by an aggregate neural model (Gratton et al., 2020; Laumann et al., 2015; Medaglia et al.,
2011; Seitzman et al., 2019), preventing group-to-individual generalizability. The inability
to make inferences on an individual level may be a key factor in the limited clinical utility
of fMRI scans (Zhuo et al., 2019), despite the numerous reviews and meta-analyses that have
implicated neural reward processing dysfunction in adolescent depression at a group level
(Fischer et al., 2019; Keren et al., 2018; Miller et al., 2015; O’Callaghan & Stringaris,
2019). En outre, heterogeneity across individuals may also limit inferences in nonclinical
cognitive neuroscience research if the group-level model obscures qualitative differences in
network functioning which may represent distinct psychological processes across individuals
(par exemple., Demidenko et al., 2022).
One alternative approach to case-controlled designs is identifying biological subgroups that
represent more homogenous network patterns across individuals and then examining behav-
ioral differences between them (Feczko & Fair, 2020). Ainsi, subgrouping approaches identify
groups of individuals based on similarity in network functioning, rather than behavior (par exemple.,
depression). En outre, as subgroups serve as an intermediate between the individual and
the group, they may be more representative of specific individuals than group-level models,
and increase the precision of resulting inferences to the individual. Multiple studies have used
subgroups to identify more precise neural network associations with depression (Drysdale
et coll., 2017; Liang et al., 2020), attention deficit/hyperactivity disorder (Costa Dias et al.,
2015), and alcohol and substance use disorders (Kashyap et al., 2020; Zhu et al., 2022). These
études (further reviewed in the Supporting Information) have demonstrated that subgroups
have external validity in their associations with clinical functioning. De plus, as they are
Neurosciences en réseau
788
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
t
/
/
e
d
toi
n
e
n
un
r
t
je
c
e
–
p
d
je
F
/
/
/
/
/
7
2
7
8
7
2
1
1
8
4
4
9
n
e
n
_
un
_
0
0
3
0
6
p
d
t
.
F
b
oui
g
toi
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
Adolescent reward network heterogeneity
intended to represent individuals more similar in network models, they may better represent
individuals than group-level models. Cependant, few studies have assessed the internal validity
of the identified subgroup solution (Brucar et al., 2023), an important step to ensure that the
identified subgroups are capturing true network differences, rather than noise.
The development of Group Iterative Multiple Model Estimation (GIMME; Beltz & Gates, 2017;
Gates et al., 2014; Gates & Molenaar, 2012) has furthered research examining network het-
erogeneity by identifying group-level networks with paths common to the sample, data-driven
subgroups, and individualized networks. GIMME searches for unobserved network structures
in intensive time series data. Rather than averaging across participants, GIMME only adds
paths to a group-level network that are statistically significant for the majority of individuals
in a specified sample. GIMME then uses a community detection algorithm to also search for
subgroups of individuals with similar network features, et, finally, adds paths that are signif-
icant for each individual considered independently.
Several studies have used GIMME to study associations between subgroups of network
functioning and clinical outcomes. Price et al. (2017un) used GIMME to identify two subgroups
of resting-state connectivity in a sample of adults with depression. They found that a subgroup
with an intradefault mode network (DMN) path from the perigenual anterior cingulate cortex
to the posterior cingulate cortex (PCC) and a path from the dorsal anterior cingulate cortex
(ACC) to the right insula represented the majority of patients, and a subgroup defined by addi-
tional paths to the parietal lobe represented more participants who identified as female, had
comorbid anxiety disorders, and had more recurrent depression. In a separate study of adults
with and without depression, Price et al. (2017b) identified two subgroups of network connec-
tivity after positive mood induction. They found that one subgroup had fewer connectivity
paths and decreased connectivity in ventral affective network and DMN paths relative to
the other subgroup, and individuals in this subgroup had a higher rate of depression diagnosis
and higher symptom severity. While most studies have focused on adult samples and resting-
state connectivity, Demidenko et al. (2022) recently examined network heterogeneity during a
reward task in a sample of older adolescents and early adults (âge moyen = 19 années). Ils
identified two subgroups, one of which had increased connectivity paths in reward, cognitive
control, and salience networks, and another which had fewer subgroup-level paths that were
concentrated in cortical regions. Participants in the subgroup characterized by increased net-
work density had increased self-reported sensation-seeking behavior. Within this subgroup,
connectivity between the ventromedial prefrontal cortex and right orbitofrontal cortex was
positively associated with sensation-seeking behavior and connectivity between the right orbi-
tofrontal cortex and right ventral striatum was negatively associated with sensation-seeking
behavior.
Studies have used GIMME to identify subsets of individuals more similar in their network
functioning and have found differences between subgroups on measures of clinical function-
ing, increasing the ability to make inferences on an individual level. Cependant, little work has
been done assessing heterogeneity of networks at the group-level before subgroup identifica-
tion, or examining network heterogeneity in adolescent samples. Ici, we explore heteroge-
neity of reward networks in 103 early adolescents (âge moyen = 11.32 années, SD = 1.46) et
examine associations between network features and behavioral outcomes, depression, et
risk for alcohol and substance use problems. The study of reward networks in adolescence
is an important extension as adolescence is a key risk period for the onset of multiple mental
disorders such as depression (Costello et al., 2011; Solmi et al., 2021) and substance use prob-
lems (Grant & Dawson, 1998; Poudel & Gautam, 2017), which are in part characterized by
alterations in reward functioning during adolescence (Casey et al., 2019; Forbes & Dahl, 2012;
Neurosciences en réseau
789
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
/
t
/
e
d
toi
n
e
n
un
r
t
je
c
e
–
p
d
je
F
/
/
/
/
/
7
2
7
8
7
2
1
1
8
4
4
9
n
e
n
_
un
_
0
0
3
0
6
p
d
.
t
F
b
oui
g
toi
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
Adolescent reward network heterogeneity
Heitzeg et al., 2015). D'abord, we characterize qualitative network heterogeneity between
individuals by examining whether an aggregate connectivity network is representative of
connectivity networks identified in individuals. Deuxième, we identify data-driven subgroups
of individuals with similar reward networks, test the robustness of this solution, and examine
if subgroup membership is associated with reward-related outcomes. Enfin, we test the effect
of network features on an individual level by examining associations between individualized
connectivity paths and reward-related outcomes in the sample using regularized regressions.
MÉTHODES
The study was preregistered on Open Science Framework (https://doi.org/10.17605/osf.io
/6n5j2). Deviations from the preregistration include the Early Adolescent Temperament
Questionnaire pleasure intensity scale not being available from parent report and the additions
of exploratory analyses examining network associations with risk for alcohol and substance use
disorders and behavioral outcomes at 27-month follow-up after acquisition of supplemental
funding. Time series data and open code for reproducing analyses are available on Open
Science Framework (https://doi.org/10.17605/osf.io/7dgp4).
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
/
/
t
e
d
toi
n
e
n
un
r
t
je
c
e
–
p
d
je
F
/
/
/
/
/
7
2
7
8
7
2
1
1
8
4
4
9
n
e
n
_
un
_
0
0
3
0
6
p
d
t
.
F
b
oui
g
toi
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
Study Participants and Exclusion Criteria
Participants came from the Temple Adolescent Depression Study. The study was approved by
Temple University’s Institutional Review Board (IRB no. 23174) and consent forms were signed
prior to participation. Youth between ages 9 et 14 years with a primary caregiver were
eligible for participation. Youth with a history of neurological disorder, head injury, pervasive
developmental disorders, and/or intellectual functioning less than 70 as assessed by the
Kaufman Brief Intelligence Test (Kaufman & Kaufman, 2013) were ineligible for study partic-
ipation. Youth also were ineligible for participation if they had a history of bipolar disorder,
psychosis spectrum disorders, developmental disorders or disabilities, neurological or cardio-
vascular diseases that affected central nervous system blood flow, were taking any psychotro-
pic medications at the time of recruitment or scan, or were not able to complete an MRI scan
safely. In total, 175 participants consented and completed the MRI portion of the study. Par-
ticipants were excluded in a four-stage quality control process to reduce the effect of noise,
particularly motion. D'abord, participants were excluded if there were scanning issues (N = 5),
incidental radiological findings (N = 1), ou <75% behavioral compliance with the scanning
task (N = 32). Second, participants were excluded based on manual inspection of MRIQC
(Esteban et al., 2017) outputs for artifacts such as severe ringing or signal loss (N = 15). Third,
participants were excluded after fmriprep preprocessing if their temporal signal-to-noise ratio
(tSNR) or framewise displacement (FD) value was less than or greater than 1.5 times the inter-
quartile range for each parameter, respectively (N = 5). Finally, participants were excluded if
more than 25% of their frames had a FD value of at least 2 mm (N = 12). In total, 70 of the
initial 175 participants were excluded, resulting in a sample of 105. The final sample had a
mean age of 11.32 years (SD = 1.46), 58% were female, 48% were White, 36% were Black or
African American, 1% were Asian, 11% were multiracial, and 4% preferred not to identify a
race; 6% of the sample were Hispanic.
fMRI Acquisition and Scanning Task
Neuroimaging data were acquired using a 3T Philips Ingenia scanner. BOLD functional
images were acquired with a gradient echo planar imaging sequence and covered 34 axial
slices (3 mm thick; TR = 2,000 ms, TE = 25 ms, field of view = 20 cm, matrix = 64 × 64).
Network Neuroscience
790
Adolescent reward network heterogeneity
We used a Card Guessing task (Forbes et al., 2006) that is frequently used in studies of
monetary incentives (see Forbes et al., 2009, for task schematic). This event-related task exam-
ines responses to monetary gains and losses. Each trial includes both anticipation and outcome
periods, and participants receive win, loss, or no-change feedback for each trial. Participants
were told that their performance determines a monetary reward to be received after the scan,
such that they would receive $1 for each win, lose 50 cents for each loss, and no change for neutral outcomes. Trials were presented in a pseudorandom order with predetermined out- comes. During each 17-second trial, participants had 4 seconds to select whether the value of a visually presented card with a possible value of 1–9 will be higher or lower than 5. After a choice was made, the trial type (reward or loss) is presented visually for 6 seconds (anticipa- tion). The “actual” numerical value of the card is briefly displayed (500 ms), followed by out- come feedback (500ms), and, finally, a crosshair is presented for 7 seconds (outcome). The task included a jittered intertrial interval that averages 4 seconds. The task included 24 trials and lasted 8 minutes 2 seconds (239 acquisitions). The participants were unaware of the fixed outcome probabilities and were led to believe that outcomes were solely due to chance. The participants’ engagement and motivation were maintained by verbal encouragement during practice and between tasks in the magnet. Behavioral Measures Several child-about-self and parent-about-child measures were examined as reward-related behavioral outcomes. Measures related to reward approach behaviors, inhibitory control, and clinical functioning and risk were selected as tests of external validity (i.e., whether indi- vidual differences in identified reward-related network features were associated with differ- ences in reward-related behavioral outcomes). For parent report, we relied on reports from primary caretakers (99% mothers). When reports were unavailable for primary caretakers, we relied on reports from secondary caretakers. Correlations between behavioral measures are reported in the Supporting Information. Descriptive statistics of each behavioral measure for the included sample and excluded participants are provided in the Supporting Information. l D o w n o a d e d f r o m h t t p : / / d i r e c t . m i t . / / t e d u n e n a r t i c e - p d l f / / / / / 7 2 7 8 7 2 1 1 8 4 4 9 n e n _ a _ 0 0 3 0 6 p d . t Discounting rate. The Delay Discounting task (McClure et al., 2004) assessed child preference for smaller, but immediate, rewards relative to larger, delayed rewards. Higher reward sensi- tivity reflects a greater preference for the immediate reward, indexed by the log of the dis- counting rate (log(k)). Participants chose between immediate and delayed rewards (e.g., $400 today or $1,000 in a week) considered roughly equal over four delay intervals (1 week,
1 month, 6 months, 1 year).
Early Adolescent Temperament Questionnaire (EATQ). The EATQ (Ellis & Rothbart, 2001) is a
103-item self-report measure assessing temperament and self-regulation in children and
adolescents. Items are rated on a five-point Likert scale, ranging from “Almost always untrue,”
to “Almost always true.” EATQ Pleasure Sensitivity subscale contains seven items assessing
pleasure related to activities or stimuli involving low intensity, rate, complexity, novelty, and
incongruity. The Pleasure Sensitivity subscale is only assessed in the child self-report EATQ
(α = .85, ω = .85). The EATQ Inhibitory Control subscale is included in the child self- and
parent report EATQ. Inhibitory control represents the capacity to suppress inappropriate
responses (youth α = 62, ω = .49; parent α = .64, ω = .60).
f
b
y
g
u
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
Pleasure Scale for Children (PSC). The PSC (Kazdin, 1989) is a 39-item self-report measure
assessing hedonic responses to rewarding events and activities in children. Youth were asked
to indicate on a three-point Likert scale if that activity would make them feel “very happy,”
Network Neuroscience
791
Adolescent reward network heterogeneity
“happy,” or if it “wouldn’t matter.” The PSC was only administered to child participants (α =
.97, ω = .96).
Behavioral inhibition system/ behavioral activation system (BIS/ BAS). The BIS/ BAS (Carver &
White, 1994) is a 24-item self-report measure assessing appetitive and aversive motivation.
Participants rate each item on a four-point Likert scale ranging from “Very true for me” to “Very
false for me.” We used the Drive, Reward Responsiveness, and Fun seeking subscales of the
BAS. Both children and parents completed the BIS/BAS about the child. The BAS Drive sub-
scale contains four items that reflect persistent pursuit of desired goals (youth α = .75, ω = .78;
parent α = 86, ω = .81). The BAS Reward Responsiveness subscale contains five items that
reflect positive responses to the occurrence or anticipation of reward (youth α = 82, ω =
.75; parent α = .79, ω = .74). The BAS Fun Seeking subscale contains four items that reflect
desire for new rewards and a willingness to approach a potentially rewarding event spuriously
(youth α = .74, ω = .68; parent α = .72, ω = .68).
Child Depression Inventory (CDI). The CDI (Kovacs, 1985) is a 27-item self-report measure
assessing the cognitive, affective, and behavioral symptoms of depression in children and ado-
lescents. Participants endorse statements about their experience of depressive symptoms on a
three-point scale (youth α = .95, ω = .91; parent α = .92, ω = .90).
Mood and Feelings Questionnaire (MFQ). The MFQ (Angold et al., 1995) is 33-item self-report
measure assessing recent depressive symptoms. Participants endorse whether depressive
symptoms are “Not True,” “Sometimes True,” or “True” of them in the past two weeks (youth
α = .98, ω = .96; parent α = 95, ω = .94).
Exploratory Measures
Alcohol expectations (AE) scale. The AE scale contains 15 items that measure adolescent atti-
tudes and behavioral expectations for alcohol use. Participants endorse degree of agreement
with statements about expectations after drinking (e.g., “After a few drinks of alcohol, I would
be more likely to be courageous/calm/sociable”) on a five-point Likert scale ranging from
“Strongly Disagree” to “Strongly Agree.”
Familial risk for substance use problems. Familial risk was assessed by maternal history of diag-
nosis of alcohol and/or substance use disorder was assessed by the Structured Clinical Inter-
view for the DSM-5 (SCID-5; First et al., 2016).
Follow-up measures. Follow-up measures of each of the above outcome variables (excluding
familial risk) were assessed at a 27-month follow-up assessment. There was high attrition due
to the COVID-19 pandemic. Follow-up sample sizes ranged from 41 (child-reported inhibitory
control) to 59 (child-reported depression) and are fully described in the Supporting Information.
fMRI Preprocessing and Time Series Extraction
Preprocessing was performed using fmriprep 20.2.6 (Esteban et al., 2019, 2022a; RRID:
SCR_016216), which is based on Nipype 1.7.0 (Esteban et al., 2022b; Gorgolewski et al.,
2011; RRID:SCR_002502). Fmriprep’s fieldmap-less distortion correction was used. BOLD
runs were slice-time corrected with a middle slice reference and resampled onto the default
MNI152NLin2009cAsym space. Full fmriprep preprocessing details are provided in the Sup-
porting Information.
Further preprocessing was conducted in CONN toolbox, version 20.b ( Whitfield-Gabrieli
& Nieto-Castanon, 2012) in MATLAB 2017a. Minimally preprocessed data from fmriprep were
Network Neuroscience
792
l
D
o
w
n
o
a
d
e
d
f
r
o
m
h
t
t
p
:
/
/
d
i
r
e
c
t
.
m
i
t
.
/
/
t
e
d
u
n
e
n
a
r
t
i
c
e
-
p
d
l
f
/
/
/
/
/
7
2
7
8
7
2
1
1
8
4
4
9
n
e
n
_
a
_
0
0
3
0
6
p
d
t
.
f
b
y
g
u
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
Adolescent reward network heterogeneity
euSEM:
Approach to effective connectivity
that models contemporaneous and
lagged relationships between nodes,
as well as effects of exogenous
variables.
Functional connectivity:
Correlation between BOLD activity
of two brain regions or voxels.
Idiographic:
Study of a single individual (N = 1)
rather than a group.
spatially smooth with a 6-mm full-width at half-maximum Gaussian kernel. Data were then
denoised using the first six aCompCor components, three translational motion regressors
and their first derivatives, three rotational motion regressors and their first derivatives, the full
set of cosine regressors, spike regressors defined by volumes with greater than 1.5-mm FD or
2.0 standardized DVARS values, and nonsteady-state outliers, as calculated by fmriprep. Time
series were then extracted from 15 regions of interest (ROIs) using CONN’s default Harvard-
Oxford probabilistic atlas (Desikan et al., 2006). The ROIs were the ACC, PCC, ventromedial
prefrontal cortex (vmPFC; average of frontal medial cortex and subcallosal cortex), and the
bilateral orbitofrontal cortex (OFC), insula, caudate, putamen, nucleus accumbens (NAcc),
and amygdala.
Analysis Plan
All statistical analyses on fMRI timeseries were conducted in R 4.0.1. Package versions are
listed in the Supporting Information.
Effective connectivity network modeling. We used several related iterative extended unified
structural equation modeling (euSEM) processes to parse effective connectivity network het-
erogeneity at different levels of data aggregation. Effective connectivity is distinct from
correlation-based functional connectivity in that it tests a directional model as the mechanism
that generated the data, improving the ability to test hypotheses about how the brain is func-
tioning (Friston, 2011). Effective connectivity modeling with euSEM differs from Dynamic
Causal Modeling (DCM; Friston et al., 2003), a common approach to effective connectivity
modeling, in two key ways. First, euSEM, as implemented here, is a data-driven approach,
while DCM is confirmatory. Second, euSEM convolves activation with the hemodynamic
response function, but does not assume relationships between neural and hemodynamic pro-
cesses. However, several simulations have shown euSEM outperforming DCM in uncovering
true network structures (Gates et al., 2011; Gates & Molenaar, 2012). For each network in this
study, estimation begins with a null model, adds the contemporaneous, lagged, or autoregres-
sive connectivity path that best improves model fit based on modification indices, until no
further edges would do so. For all networks, nodes were the 15 ROIs and the reward antici-
pation and reward outcome exogenous task events that were convolved with the hemody-
namic response function.
Aggregate and idiographic networks. First, to assess how well a group-level model reflects each
individual, we used euSEM to identify idiographic directed effective connectivity network
models for the group aggregate and each individual (Gates et al., 2010, 2011). The aggregate
group-level model was estimated using the aggSEM function in the GIMME package (Gates &
Molenaar, 2012). This function concatenates time series from each participant into a single
time series, then iteratively adds network paths that significantly improve model fit based
on modification indices, beginning with an empty null model. To estimate idiographic network
models, we used the indSEM function in the GIMME package in R. Using the same network
identification process as the aggregate model, indSEM iteratively estimates networks for each
individual independently, using no information from the group. We then examined how many
paths of each individual-level network paths were present in the aggregate model.
Subgroup networks. Second, we examined the potential presence of subgroups of individuals
with more homogenous network features using GIMME (Gates et al., 2017). GIMME is an iter-
ative model building application that relies on different levels of data aggregation. Simulations
have shown that GIMME effectively detects more true edges and fewer spurious relationships
Network Neuroscience
793
l
D
o
w
n
o
a
d
e
d
f
r
o
m
h
t
t
p
:
/
/
d
i
r
e
c
t
.
m
i
t
.
/
/
t
e
d
u
n
e
n
a
r
t
i
c
e
-
p
d
l
f
/
/
/
/
/
7
2
7
8
7
2
1
1
8
4
4
9
n
e
n
_
a
_
0
0
3
0
6
p
d
.
t
f
b
y
g
u
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
Adolescent reward network heterogeneity
than many other connectivity approaches, with the core distinction being that GIMME explic-
itly accounts for sample heterogeneity (Gates & Molenaar, 2012). Group-level network paths
were first identified for the whole sample using paths that are significant for the majority of
individuals (default 75%). Subgroups of individuals with similar network properties were then
identified based on beta weights and modification indices of group-level paths, using a
Walktrap community detection algorithm (Pons & Latapy, 2005). Subgroup-level paths were
then iteratively added to each individual in the subgroup with a default majority threshold of
>50%. Subgrouping GIMME has been shown to accurately capture subgroup classification in
Monte Carlo simulations and improve the detection of the presence and direction of effects
compared to the default GIMME process (Beltz & Gates, 2017).
To examine robustness of the identified subgroup solution, we used the perturbR package
(Gates et al., 2019). Perturbr (1) assesses the stability of the subgrouping solution after itera-
tively randomly changing edges in the matrix and (2) compares the modularity of the obtained
similarity matrix against simulated matrices to test if the obtained value is greater than what
would be expected by chance. Similarity matrices are simulated by a weighted extension of
the Erdos-Renyi binary random matrix approach which maintains statistical equivalence of
nodes (c'est à dire., ROI), thereby maintaining the weighted properties of the original similarity matrix
(Garlaschelli, 2009; Gates et al., 2019). The preregistered robustness criteria were (1) le
similarity matrix requiring at least 20% of its edges being perturbed before 20% of individuals
were placed in different clusters, et (2) the modularity for the original solution being greater
than or equal to the 95th percentile of modularity obtained simulated matrices. A solution that
passes these criteria will be regarded as robust to noise and modular (c'est à dire., independent).
Individual networks. Enfin, as group-level information has been shown to improve detection
of individual-level paths (Gates et al., 2017; Gates & Molenaar, 2012), we examined individ-
ualized networks estimated using the group-level model as the null. GIMME completes its
model identification process by using the group and subgroup (if identified) networks as a null
model for each individual, and then iteratively adds individual-level paths. Since the
individual-level search follows the subgroup estimation process, we will only identify
individual-level networks following the subgroup search if the subgroup solution passes the
preregistered robustness criteria. If the subgroup solution does not pass the preregistered
robustness criteria, we will estimate the individual-GIMME networks without a subgroup
recherche.
Associations with behavioral outcomes. After identifying connectivity network models, nous
examined how network features were associated with behavioral reward-related outcomes.
To decrease the number of comparisons, we reduced the BAS subscales, PSC, and EATQ
pleasure sensitivity subscale to a single reward sensitivity factor, and the CDI and MFQ to a
depression aggregate score. These processes were completed separately for child- and parent-
reported measures. Exploratory factor analysis (EFA) solutions for reward sensitivity are
described in the Supporting Information. For depression, we standardized sum scores of the
CDI and MFQ and averaged them. If one scale score was missing for a reporter, the other sum
score was used by itself. Scores on the child- and parent-reported reward sensitivity factor,
enfant- and parent-reported depression aggregate, child discounting rate, and child- et
parent-reported inhibitory control were the final behavioral outcomes.
For idiographic and individual-GIMME models, we examined network features’ behavioral
associations using adaptive lasso (Zou, 2006) in the glmnet R package. Adaptive lasso is an L1-
regularized regression method that uses different regularization penalties for each coefficient.
As coefficients are permitted to be penalized to 0, adaptive lasso also performs feature
794
Adaptive lasso:
Regression method where
coefficients are differentially
penalized to reduce bias. Penalties
can minimize coefficients to 0,
performing feature selection.
Neurosciences en réseau
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
/
/
t
e
d
toi
n
e
n
un
r
t
je
c
e
–
p
d
je
F
/
/
/
/
/
7
2
7
8
7
2
1
1
8
4
4
9
n
e
n
_
un
_
0
0
3
0
6
p
d
.
t
F
b
oui
g
toi
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
Adolescent reward network heterogeneity
selection, a useful tool due to the large number of features provided by GIMME. Adaptive lasso
provides a final set of features (c'est à dire., directed connectivity paths) that are associated relevant
outcome variable and their respective beta weights. Ici, we followed a similar procedure
as Dajani et al. (2020), but included binary features of whether a path was present or absent
(c'est à dire., statistically significant) for idiographic and individual-GIMME paths, rather than their beta
weight, which would have been missing from individuals without that path. For both idio-
graphic and individual-GIMME paths, paths were only considered if they were present for
at least 20% of the sample (21 individuals). For GIMME group-level paths, beta weights were
used since they are estimated for every individual. Adaptive penalties were determined by a
ridge regression with 10-fold cross-validation, and adaptive lasso was then fit with a 10-fold
cross-validation. All independent variables were standardized. We also calculated R2 and
adjusted-R2 values for the final set of features for each outcome variable. Finalement, ce
approach aims to identify what set of connectivity paths explain the maximum variance in
a given behavioral outcome.
Behavioral differences between subgroups were tested using an analysis of variance
(ANOVA) to examine omnibus differences with a Benjamini–Hochberg false discovery rate
correction (Benjamini & Hochberg, 1995) to adjust for multiple comparisons. Post hoc pair-
wise comparisons examined differences between subgroups if omnibus tests were statistically
significant.
RÉSULTATS
In initial GIMME models, path estimates for two participants yielded extreme outlier values.
These two participants were removed, and all network models were reestimated with the
remaining 103 participants.
Idiographic and Aggregate Networks
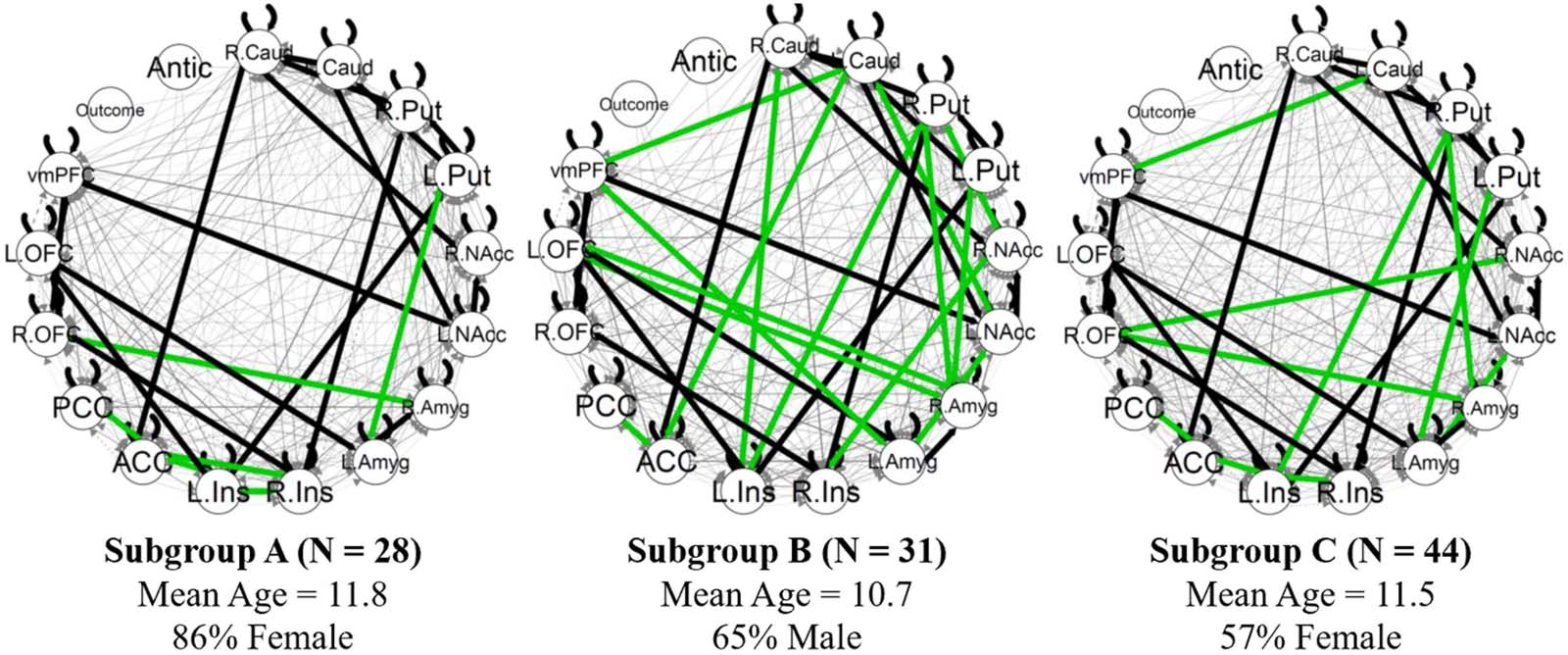
Chiffre 1 shows the aggregate directed connectivity network. Connectivity paths that were
either significant in the aggregate network or at least 20% of the sample (21 individuals) idi-
ographic networks are listed in the Supporting Information. The median idiographic network
only shared 10 of the 24 aggregate paths (mean = 9.4) et 85 of the 103 participants (83%)
shared less than half (<12) of the aggregate paths. Additionally, the two idiographic paths that
were significant for the largest number of individuals were not significant in the aggregate net-
work. The idiographic network most resembling the aggregate network had 17 of the 24 paths,
and two idiographic networks had 16 of the 24 paths. Visual comparisons between the aggre-
gate model, the closest resembling individual model, the median-resembling individual
model, and the least-resembling individual model are shown in the Supporting Information.
Overall, the aggregate level network was not a good representation of individual-level
networks.
Subgrouping GIMME
GIMME identified three subgroups (Figure 2). Subgroup characteristics are described in
Table 1. Subgroup 1 has the least dense connectivity network; it has four cortical paths
between the bilateral insular regions, the ACC, and the PCC, and two subcortical paths,
one from the left putamen to the left amygdala and another from the right OFC to the right
amygdala. Participants in Subgroup 2 had the densest connectivity network, with 13 of the
14 subgroup-level paths involving subcortical regions. Participants in Subgroup 3 had an inter-
mediately dense network, with 6 of the 10 paths involving striatal regions. Participants in
Network Neuroscience
795
l
D
o
w
n
o
a
d
e
d
f
r
o
m
h
t
t
p
:
/
/
d
i
r
e
c
t
.
m
i
t
.
/
/
t
e
d
u
n
e
n
a
r
t
i
c
e
-
p
d
l
f
/
/
/
/
/
7
2
7
8
7
2
1
1
8
4
4
9
n
e
n
_
a
_
0
0
3
0
6
p
d
t
.
f
b
y
g
u
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
Adolescent reward network heterogeneity
Figure 1. Aggregate connectivity network. Nodes are 15 ROIs and two exogenous task events
convolved with hemodynamic response function. Red edges indicate significant positive connec-
tivity paths for the averaged group. Blue edges indicate negative paths. Edge thickness corresponds
with beta weight. L. = left. R. = right; Caud = caudate; Put = putamen; NAcc = nucleus accumbens.
Amyg = amygdala; Ins = insula; ACC = anterior cingulate cortex; PCC = posterior cingulate cortex;
OFC = orbitofrontal cortex; vmPFC = ventromedial prefrontal cortex; Antic = anticipation.
Subgroup 1were significantly older than participants in other subgroups and were predomi-
nantly female. Participants in Subgroup 2 were mostly male and were significantly younger
than participants in Subgroup 1, but not significantly younger than participants in Subgroup 3.
Subgroup 3 had the most participants, had a near even split between sexes, and had partici-
pants that were significantly older than those in Subgroup 2, but not significantly different in age
Figure 2. GIMME subgroups. Black edges reflect connectivity paths significant for the group (same for all subgroups). Green edges reflect
subgroup-specific connectivity paths. Gray edges reflect individual-level paths identified after the subgroup search. L. = left; R. = right; Caud =
caudate; Put = putamen; NAcc = nucleus accumbens; Amyg = amygdala; Ins = insula; ACC = anterior cingulate cortex; PCC = posterior
cingulate cortex; OFC = orbitofrontal cortex; vmPFC = ventromedial prefrontal cortex; Antic = anticipation.
Network Neuroscience
796
l
D
o
w
n
o
a
d
e
d
f
r
o
m
h
t
t
p
:
/
/
d
i
r
e
c
t
.
m
i
t
.
/
/
t
e
d
u
n
e
n
a
r
t
i
c
e
-
p
d
l
f
/
/
/
/
/
7
2
7
8
7
2
1
1
8
4
4
9
n
e
n
_
a
_
0
0
3
0
6
p
d
.
t
f
b
y
g
u
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
Adolescent reward network heterogeneity
Number of individuals
Table 1. GIMME subgroup characteristics
Subgroup 1
28
Subgroup 2
31
Subgroup 3
44
Number of subgroup paths
6
14
10
Omnibus test
–
–
Age mean (SD)
Percent female (no.)
11.76 (1.53)a
10.73 (1.32)b
11.49 (1.40)b
F(2,98) = 4.18*
86% (24)a
35% (11)b
57% (25)c
χ2(2) = 16.36**
Framewise displacement in mm (SD)
0.27 (0.13)a
1.23 (0.50)b
0.62 (0.34)c
F(2,98) = 26.13**
Note. For each row, subgroups that do not share the same subscript are different at p < .05.
*p < .05
**p < .001.
than participants in Subgroup 1. The subgroups appear to reflect reward network maturity, with
more mature subgroups having denser networks, older participants, and predominantly female
participants.
In tests of the preregistered subgroup validity criteria, the obtained subgroup solution was
stable (i.e., robust to noise), passing the first criterion, but had low modularity, failing the
second criterion. Using the Variation of Information criteria (see Supporting Information),
approximately 35% edges had to be perturbed before 20% of participants were placed into
different clusters. Additionally, according to the adjusted Rand index, the solutions when
20% of edges were perturbed were more similar to the original model than a model with
20% of cluster assignments randomly swapped (t = 102, p < .001). These indices suggest that
the Walktrap clustering algorithm reliably produced the same subgrouping solution as noise
iteratively increased, until about one third of paths were perturbed. However, the subgroup
solution had very low modularity (.03). Furthermore, sensitivity analyses indicated that
participants in different subgroups significantly differed in scanner movement, raising the
possibility that subgroups were identified based on movement, rather than distinct network
properties.
Overall, the obtained subgroups were robust to noise, but have very low modularity. Thus,
we present the subgroup solution and examine differences between subgroups on reward out-
comes, as previous studies have solely utilized robustness to noise to assess validity of GIMME
subgroups (Kaurin et al., 2022). However, as the subgroup solution did not pass our preregis-
tered modularity criteria, we also estimate GIMME without subgroups for examination of
individual-level paths.
Individual GIMME
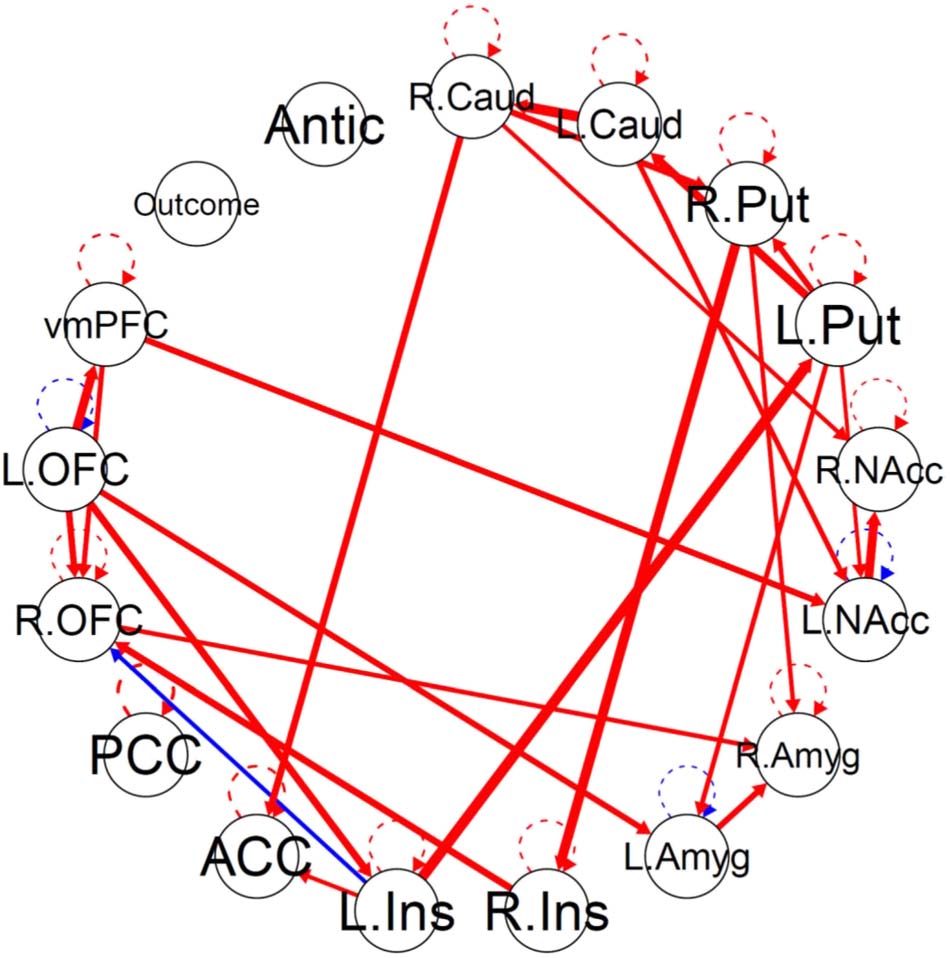
Figure 3 shows the GIMME group-level network without a subgroup search. There were 19
group-level paths (Table 2). The paths with the strongest weights, excluding lateralized paths
of same region, were from the left insula to the left putamen, from the right insula to the right
putamen, from the left OFC to the left amygdala, from the left putamen to the left caudate, and
from the left OFC to the vmPFC. GIMME identified five group-level paths that were not iden-
tified in the aggregate model: from the right insula to the right putamen, from the left insula to
the left OFC, from the right insula to the left insula, from the right putamen to the right caudate,
and from the right OFC to the left OFC. Conversely, the aggregate group model identified 10
paths that were not present in the GIMME group model. No group-level paths involving the
reward outcome or anticipation were identified.
Network Neuroscience
797
l
D
o
w
n
o
a
d
e
d
f
r
o
m
h
t
t
p
:
/
/
d
i
r
e
c
t
.
m
i
t
.
t
/
/
e
d
u
n
e
n
a
r
t
i
c
e
-
p
d
l
f
/
/
/
/
/
7
2
7
8
7
2
1
1
8
4
4
9
n
e
n
_
a
_
0
0
3
0
6
p
d
.
t
f
b
y
g
u
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
Adolescent reward network heterogeneity
Figure 3. GIMME group network. (A) The GIMME output network, with black edges representing group-level paths and gray edges repre-
senting individual-level paths. (B) The same group-level paths projected onto a smoothed MNI glass brain. Edge thickness corresponds to the
path’s beta weight. The brain network was visualized with the BrainNet Viewer (Xia et al., 2013). L. = Left. R; = Right; Caud = caudate; Put =
putamen; NAcc = nucleus accumbens; Amyg = amygdala; Ins = insula; ACC = anterior cingulate cortex; PCC = posterior cingulate cortex;
OFC = orbitofrontal cortex; vmPFC = ventromedial prefrontal cortex; Antic = anticipation.
l
D
o
w
n
o
a
d
e
d
f
r
o
m
h
t
t
p
:
/
/
d
i
r
e
c
t
.
m
i
t
.
/
t
/
e
d
u
n
e
n
a
r
t
i
c
e
-
p
d
l
f
/
/
/
/
/
7
2
7
8
7
2
1
1
8
4
4
9
n
e
n
_
a
_
0
0
3
0
6
p
d
.
t
f
b
y
g
u
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
GIMME networks fit the data well for the majority of participants. Using fit criteria of com-
parative fit index (CFI) ≥ .90, nonnormal fit index (NNFI) ≥ .90, root-mean-square error of
approximation (RMSEA) ≤ .08, and standardized root-mean-squared residual (SRMR) ≤ .08,
100 participants met all four criteria, 1 participant met two of the four criteria, and 2 partic-
ipants did not meet any of the criteria. Individual models from GIMME also demonstrated sub-
stantial network heterogeneity, such that the presence of various individual-level paths differed
widely across adolescents. Table 3 lists the 17 individual-level paths that were significant for at
least 21 participants. The most common individual-level paths were from the right caudate to
the right NAcc and from the right putamen to the right amygdala. A visualization of differences
in individual-level paths across select participants is presented in the Supporting Information.
We also estimated GIMME models without including exogenous task regressors, and the
resulting models were nearly identical. All GIMME outputs are provided on OSF (osf.io
/zymq5).
Associations With Behavioral Outcomes
There were no significant differences in any of the preregistered outcomes—discounting rate,
reward sensitivity, inhibitory control, and depression—across subgroups. The two participants
that did not meet two out of four fit criteria were excluded from further analyses. Table 4 dis-
plays results of associations between GIMME network features and behavioral outcomes using
adaptive lasso. Network paths were associated with age, sex, discounting rate, child- and
parent-reported reward sensitivity, and child- and parent-reported inhibitory control, and
child-reported depression. Adaptive lasso did not identify any paths that were associated with
parent-reported depression. Based on R2, network features were most associated with child-
reported reward sensitivity (adjusted R2 = .19, 10 paths) and parent-reported inhibitory control
(adjusted R2 = .16, 7 paths). The individual-level path from the vmPFC to the ACC was asso-
ciated with the greatest number of outcomes, including discounting rate, child-reported
reward sensitivity-child report, and parent-reported inhibitory control. Sensitivity analyses
accounting for scanner movement were conducted by assessing associations after inclusion
Network Neuroscience
798
Adolescent reward network heterogeneity
From
Left insula
Left caudate
Right insula
Table 2. GIMME group-level paths
To
Left putamen
Right caudate
Right putamen
Beta (SD)
0.77 (0.19)
0.63 (0.22)
0.59 (0.20)
Left orbitofrontal cortex
Ventromedial prefrontal cortex
0.52 (0.33)
Ventromedial prefrontal cortex
Left nucleus accumbens
Left nucleus accumbens
Right nucleus accumbens
Ventromedial prefrontal cortex
Right orbitofrontal cortex
Left putamen
Left orbitofrontal cortex
Left caudate
Left amygdala
Right insula
Right caudate
Right orbitofrontal cortex
Anterior cingulate cortex
Right orbitofrontal cortex
Right amygdala
Left insula
Left caudate
Left putamen
Left amygdala
Right insula
Right putamen
Left orbitofrontal cortex
Left nucleus accumbens
Right putamen
Right amygdala
Left insula
Right caudate
Right orbitofrontal cortex
Left orbitofrontal cortex
Note. Paths are listed in the order identified iteratively by GIMME, using modification indices.
0.40 (0.20)
0.66 (0.22)
0.38 (0.21)
0.57 (0.32)
0.59 (0.60)
0.44 (0.20)
0.42 (0.25)
0.30 (0.24)
0.49 (0.20)
0.36 (0.23)
0.45 (0.23)
0.46 (0.24)
0.66 (0.66)
0.32 (0.23)
0.50 (0.30)
of mean FD for each participant. Results were largely similar after including mean FD. Full
results are presented in the Supporting Information.
Adaptive lasso results using idiographic network features are presented in the Supporting
Information. Results and overall trends were similar to those using GIMME networks, although
the number of significant features and the strength of associations tended to be higher using
idiographic network features.
Post Hoc Analyses
The final adaptive lasso model explained 31% of the variance in having parental history of
alcohol and/or substance use disorder using 13 paths. The group-level path from the ventro-
medial prefrontal cortex to the right orbitofrontal cortex and the individual-level paths from the
right putamen to the right caudate and from the right caudate to the right nucleus accumbens
had the strongest associations. No paths were associated with child alcohol expectancies at
baseline. Since participants at baseline may be too young (mean age = 11.32) for alcohol
expectancies, we also explored associations with alcohol expectancies at 27-month follow-
Network Neuroscience
799
l
D
o
w
n
o
a
d
e
d
f
r
o
m
h
t
t
p
:
/
/
d
i
r
e
c
t
.
m
i
t
.
/
t
/
e
d
u
n
e
n
a
r
t
i
c
e
-
p
d
l
f
/
/
/
/
/
7
2
7
8
7
2
1
1
8
4
4
9
n
e
n
_
a
_
0
0
3
0
6
p
d
.
t
f
b
y
g
u
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
Adolescent reward network heterogeneity
From
Right caudate
Right putamen
Right insula
Left insula
Table 3. GIMME individual-level paths
To
Right nucleus accumbens
Count
41
Right amygdala
Left OFC
Right putamen
Posterior cingulate cortex
Anterior cingulate cortex
Left insula
Left insula
Left putamen
Left amygdala
Anterior cingulate cortex
Left amygdala
Anterior cingulate cortex
Posterior cingulate cortex
Left amygdala
Left putamen
Right caudate
vmPFC
Right insula
Right putamen
Right caudate
Right nucleus accumbens
Left nucleus accumbens
Left nucleus accumbens
Left insula
Anterior cingulate cortex
Right amygdala
Left insula
Left caudate
Right insula
Note. Count of the number of individuals that the path was statistically significant. Paths only listed if significant
in at least 20% of sample (>21 individuals).
up with adaptive lasso. The final model explained 23% of the variance in alcohol expectan-
cies at follow-up using five paths. The individual-level paths from the left amygdala to the left
NAcc and from the ACC to the PCC had the strongest associations.
As split-sample testing was not used, we also conducted post hoc linear regressions with each
selected feature to obtain estimates of standard error and significance tests. Although adaptive
lasso feature selection is not a test of statistical significance, a large number of selected features
had significant associations in the linear regressions. Adjusted R2 values also tended to be higher
in the linear regression models. Full results are presented in the Supporting Information.
Follow-up analyses examined whether identified baseline features for each measure con-
tinued to be significantly associated with the respective measure at 27-month follow-up. Nous
tested the significance of each of the baseline features for the respective 27-month measure
using a general linear model. Follow-up child-reported reward sensitivity was significantly
associated with the significance of the right NAcc to right insula path at baseline. En plus,
follow-up parent-reported inhibitory control was significantly associated with the significance
of the ACC to PCC path at baseline. These associations were no longer significant after con-
trolling for the false discovery with a Benjamini–Hochberg correction (Benjamini & Hochberg,
1995). No other baseline features were significantly associated with respective measures at
27-month follow-up.
Neurosciences en réseau
800
37
31
30
28
27
26
26
23
23
22
22
22
21
21
21
21
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
/
/
t
e
d
toi
n
e
n
un
r
t
je
c
e
–
p
d
je
F
/
/
/
/
/
7
2
7
8
7
2
1
1
8
4
4
9
n
e
n
_
un
_
0
0
3
0
6
p
d
t
.
F
b
oui
g
toi
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
Adolescent reward network heterogeneity
Résultat
Age
Tableau 4. GIMME network associations with behavioral outcomes
Depuis
Left nucleus
Accumbens
Ventromedial
prefrontal cortex
À
Right nucleus Accumbens
Beta Path level
−0.17 Group
Total R2 Adjusted R2
0.22
0.16
Right orbitofrontal cortex
−0.12 Group
Right caudate
Cortex cingulaire antérieur
−0.20 Group
Right insula
Left insula
Right caudate
Left caudate
Right putamen
Amygdale droite
Left putamen
Left nucleus accumbens
0.10 Groupe
−0,02
Indiv
−0.22
Indiv
−0.13
Indiv
Sex
Right insula
Left Insula
Right putamen
0.32 Groupe
0.16
0.13
Left orbitofrontal cortex
0.17 Groupe
Right orbitofrontal
Left orbitofrontal cortex
0.26 Groupe
cortex
Left putamen
Left nucleus accumbens
0.27
Indiv
Discounting rate
Left orbitofrontal
Ventromedial prefrontal
0.67 Groupe
0.16
0.14
Reward sensitivity
(child report)
cortex
cortex
Right putamen
Left insula
0.15
Indiv
Ventromedial
prefrontal cortex
Cortex cingulaire antérieur
0.22
Indiv
Left orbitofrontal
Ventromedial prefrontal
0.09 Groupe
0.27
0.19
cortex
Right insula
cortex
Right orbitofrontal cortex
−0.08 Group
Right caudate
Cortex cingulaire antérieur
0.03 Groupe
Left insula
Left orbitofrontal cortex
−0.09 Group
Right orbitofrontal
Left orbitofrontal cortex
0.21 Groupe
cortex
Right caudate
Right nucleus
accumbens
Left caudate
Right insula
Left insula
Cortex cingulaire antérieur
Posterior cingulate
Cortex cingulaire antérieur
0.01
Indiv
0.17
Indiv
−0.10
Indiv
−0.16
Indiv
cortex
Ventromedial
Prefrontal cortex
Cortex cingulaire antérieur
−0.24
Indiv
Neurosciences en réseau
801
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
/
t
/
e
d
toi
n
e
n
un
r
t
je
c
e
–
p
d
je
F
/
/
/
/
/
7
2
7
8
7
2
1
1
8
4
4
9
n
e
n
_
un
_
0
0
3
0
6
p
d
t
.
F
b
oui
g
toi
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
Adolescent reward network heterogeneity
Résultat
Reward sensitivity
(parent report)
Inhibitory control
(child report)
Inhibitory control
(parent report)
Depression
(child report)
Depression
(parent report)
Baseline alcohol
expectancies*
Follow-up alcohol
expectancies*
Depuis
Left insula
Right insula
Right insula
Tableau 4.
(a continué )
À
Left putamen
Right putamen
Amygdale droite
Right caudate
Left insula
Beta Path level
0.08 Groupe
Total R2 Adjusted R2
0.08
0.05
0.05 Groupe
−0.08
Indiv
−1.01
Indiv
0.10
0.07
Left insula
Cortex cingulaire antérieur
0.42
Indiv
Posterior cingulate
Cortex cingulaire antérieur
−0.19
Indiv
cortex
Left insula
Ventromedial
prefrontal cortex
Left putamen
−0.51 Group
0.22
0.16
Right orbitofrontal cortex
−0.21 Group
Left caudate
Left nucleus accumbens
0.36 Groupe
Left amygdala
Amygdale droite
0.16 Groupe
Right caudate
Left caudate
Anterior Cingulate
Posterior cingulate cortex
−0.28
Indiv
−0.59
Indiv
cortex
Ventromedial
prefrontal cortex
Cortex cingulaire antérieur
−0.20
Indiv
Left amygdala
Amygdale droite
−0.04 Group
.02
0.01
–
–
–
–
–
–
–
–
–
–
–
–
Right orbitofrontal
Left orbitofrontal cortex
0.42 Groupe
0.27
0.23
cortex
Right putamen
Left insula
Left amygdala
Left nucleus accumbens
Right insula
Amygdale droite
Anterior cingulate
Posterior cingulate cortex
cortex
0.09
Indiv
−3.02
Indiv
1.45
Indiv
−3.24
Indiv
Familial risk for substance
Ventromedial
Right orbitofrontal cortex
0.59 Groupe
.40
.31
use problems*
prefrontal cortex
Left insula
Right insula
Left orbitofrontal cortex
Left insula
Right putamen
Right caudate
−0.46 Group
−0.34 Group
−0.59 Group
Neurosciences en réseau
802
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
/
/
t
e
d
toi
n
e
n
un
r
t
je
c
e
–
p
d
je
F
/
/
/
/
/
7
2
7
8
7
2
1
1
8
4
4
9
n
e
n
_
un
_
0
0
3
0
6
p
d
t
.
F
b
oui
g
toi
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
Adolescent reward network heterogeneity
Résultat
Depuis
Right caudate
À
Left caudate
Tableau 4.
(a continué )
Right caudate
Right nucleus accumbens
Right caudate
Left insula
Total R2 Adjusted R2
Beta Path level
−0.34
Indiv
−0.58
Indiv
−0.55
Indiv
Right insula
Left orbitofrontal cortex
0.26
Indiv
Left insula
Left insula
Left insula
Right putamen
Left amygdala
Cortex cingulaire antérieur
Anterior cingulate
Posterior cingulate cortex
cortex
0.20
Indiv
0.24
Indiv
−0.24
Indiv
−0.25
Indiv
Posterior cingulate
Cortex cingulaire antérieur
0.30
Indiv
cortex
Note. Positive relationship with sex indicates association with being male. Group-level features used each participant’s beta weight. Individual-level features
used binary significance of that path for each individual. Indiv = Individual.
*Post hoc test.
DISCUSSION
This study examined qualitative reward network functioning between early adolescents and
tested the relationship between individualized reward network features and reward-related
behavioral outcomes, depression, and risk for substance use disorder. Results showed substan-
tial heterogeneity in reward network function between adolescents, indicating that a group-
level model was not representative of individuals. Connectivity paths between striatal and
prefrontal regions were associated with multiple behavioral outcomes and had the strongest
relationships with child-reported reward sensitivity, parent-reported inhibitory control, et
familial history of alcohol and/or substance use disorder. These findings caution the reliance
on group aggregate networks for studying behavioral phenotypes such as clinical disorders
and suggest a viable alternative in the focus of individualized network features.
The majority of individual-level networks shared less than half of the connectivity paths that
were significant in the group aggregate model, indicating that the aggregate model was a poor
representation of individuals. This model nonconformity prevents generalizing findings from
group-level analyses to individuals (Molenaar, 2004). This substantially diminishes the clinical
utility of network neuroscience findings as individual-level inferences are not valid. De plus,
implications extend beyond clinical use of fMRI to basic research, where group averages may
obscure individual differences in qualitative network functioning during a task that reflect dis-
tinct underlying psychological processes. While this result is concerning for a field that largely
relies on group averages, it is consistent with previous findings that individuals exhibit traitlike
network features that are not captured in an aggregate network (Gratton et al., 2020; Seitzman
et coll., 2019). See Medaglia et al. (2011) for a discussion of nonergodicity in network neuro-
science and Fisher et al. (2018) for a discussion of the threat of nonergodicity in psychological
processes more broadly.
An increasingly common approach to examine heterogeneity in network function is the
identification of subgroups of individuals who are more homogeneous in their network func-
tioning. Subgrouping approaches capture multidimensional information in a single categorical
Neurosciences en réseau
803
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
t
/
/
e
d
toi
n
e
n
un
r
t
je
c
e
–
p
d
je
F
/
/
/
/
/
7
2
7
8
7
2
1
1
8
4
4
9
n
e
n
_
un
_
0
0
3
0
6
p
d
t
.
F
b
oui
g
toi
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
Adolescent reward network heterogeneity
variable, rather than interactions between all ROIs that would require very large samples
(Feczko et al., 2019). Ici, we used Walktrap community detection in GIMME to identify
three distinct subgroups. The subgroups reflected differences in reward network maturity, comme
they differed in reward network density, âge, and sex. Cependant, the subgroups do not resem-
ble those previously identified by Demidenko et al. (2022), who used GIMME to examine
reward network functioning in a sample of older adolescents and early adults. Differences
in identified subgroups may be due to differences between the studies in the developmental
étapes, ROI, atlases, and tasks. The Card Guessing task used here is a reward decision-making
task, and the monetary incentive delay task used by Demidenko et al. (2022) is an instrumental
reward task (Richards et al., 2013).
En outre, while unsupervised clustering methods will identify a solution, the internal and
external validity of the solution must be examined to determine utility (Brucar et al., 2023).
Ici, we found modest evidence of subgroup validity. Testing internal validity, the identified
subgroups do not appear to be a result of noise, as they showed robustness to increased levels
of path perturbations. Cependant, the subgroups had low modularity, suggesting high overlap
between them, likely due to the large number of group-level paths. En plus, the subgroups
significantly differed in mean FD, leaving uncertainty to whether they capture differences in
reward network maturity, mouvement, or both. Testing external validity, individuals across sub-
groups did not significantly differ in reward-related behavior outcomes. Ensemble, these results
highlight the importance of assessing both internal validity, such as robustness to noise, differ-
ences in movement, and solution modularity, as well as external validity such as differences in
behavioral outcomes when using data-driven network subgroup approaches.
The group-level GIMME network contained 19 total connectivity paths, which largely con-
sisted of ipsilateral connections between subcortical reward-related regions and prefrontal
régions, as well as connections between homologous regions across hemispheres. This is a
denser group-level network than that identified in Demidenko et al. (2022), potentially due
to this sample being younger, as adolescence is characterized by a shift from diffuse connectivity
to stronger, focalized connectivity networks (Khundrakpam et al., 2016). Shared group-level
paths between the two studies include those from the right OFC to the right amygdala, from the
left amygdala to the right amygdala, and from the right OFC to the left OFC.
In contrast to a subgroup-level focus, analyses focusing on the external utility of individu-
alized connectivity paths to behavioral outcomes showed promise. We assessed relationships
between individual-level network features and behavioral outcomes using adaptive lasso in
two ways. For group-level paths that were significant for at least 75% of individuals, we tested
associations between individual differences in connectivity strength and behavioral outcomes.
For individual-level connectivity paths, we tested whether a path being significant in each
individual was associated with behavioral outcomes. Surtout, this approach focused anal-
yses on network features that are precise to each individual, rather than a group average.
Using adaptive lasso, a regularized regression technique that performs feature selection and
penalized coefficient estimation specific to the sample, we found that individualized features
were associated with multiple reward-related functions and risk for substance use problems,
but not with depressive symptoms. Consistent with previous research on delay discounting
(Anandakumar et al., 2018), we found that connectivity between regions involved with cog-
nitive control regions was associated with discounting rate. We also observed a pattern where
increased group-level connectivity and number of individual-level connections between sub-
cortical to cortical regions were associated with child-reported increased reward sensitivity
and decreased inhibitory control, while increased connectivity/number of connections
between cortical regions were associated with decreased reward sensitivity and increased
Neurosciences en réseau
804
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
t
/
/
e
d
toi
n
e
n
un
r
t
je
c
e
–
p
d
je
F
/
/
/
/
/
7
2
7
8
7
2
1
1
8
4
4
9
n
e
n
_
un
_
0
0
3
0
6
p
d
.
t
F
b
oui
g
toi
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
Adolescent reward network heterogeneity
inhibitory control. These results are consistent with previous findings of separate systems for
adolescent reward functioning and cognitive control (Casey et al., 2008).
Clinically, network features were associated with parental history of alcohol and/or sub-
stance use disorder, a key risk factor for future adolescent substance use problems (Hoffmann
& Cerbone, 2002). The relationships were characterized by decreased connectivity between
several subcortical regions and the insula, and increased connectivity between the vmPFC and
right OFC. These results are consistent with previous findings that adolescents with familial risk
for alcohol and substance use problems have decreased connectivity in frontostriatal regions
(for reviews, see Cservenka, 2016, for alcohol and Heitzeg et al., 2015, and Squeglia &
Cservenka, 2017, for substance use). Results suggest particular relevance of individualized
reward network functioning in the risk for adolescent substance use problems. En outre,
the utility of individualized network features here provides further support for an increased focus
on precision functional connectivity estimation, rather than group-level estimation (Gratton
et coll., 2020). In contrast, although previous studies have found associations between reward-
related brain function and depression (Forbes & Dahl, 2012), we did not find robust associa-
tion. It is possible that brain-behavior associations in the early adolescence sample used here
are more specific to reward-based behavioral dimensions (par exemple., discounting rate, reward sensi-
tivité) or specific clinical symptoms (par exemple., anhedonia), rather than aggregate symptom scores.
Strengths and Limitations
Strengths of this study include preregistration, open data and code, examination of the internal
validity of the subgrouping solution, and assessment of multiple distinct child- and parent-
reported reward-related behaviors and clinical outcomes. This study also had several limita-
tions that future research should address. D'abord, the length of the scan may limit power to reliably
detect network structures at the individual level. Precision imaging studies have suggested
that scans may require more than 40-minutes to reliably estimate individual’s networks (c'est à dire.,
retest reliability > 0.9; Gratton et al., 2020), although other work has demonstrated reliability
of individual network differences in shorter scan lengths (Birn et al., 2013; Duda et al., 2023).
En outre, poor reliability of individual-level networks has downstream consequences on
the reliability of associations with individual differences in behavioral outcomes. GIMME par-
tially mitigates this concern by estimating individual-level features after identifying group-level
features, and simulations have shown strong reliability in detecting individual-level features
(Beltz & Gates, 2017; Gates & Molenaar, 2012). Future work should emphasize scans with
more measures to increase reliability of network estimation and brain-behavior associations.
De la même manière, longer scans with more task effects can increase power to detect effects of exogenous
events on connectivity, which was low here (discussed in more detail in Demidenko et al.,
2022). Deuxième, the sample size was not large enough to use training and testing samples to
assess reliability of the subgrouping solution in external data or the ability of networks paths to
predict behavioral outcomes. En tant que tel, associations identified by adaptive lasso may be overfit
to the sample used here. Research with larger sample sizes, such as in the Adolescent Brain
Cognitive Development (ABCD) étude (Casey et al., 2018) or Human Connectome Project
(HCP; Van Essen et al., 2013) can address these limitations going forward by testing for pre-
diction in external samples and examining potential model overfit. There was also particularly
low power to detect behavioral associations with follow-up measurements due to attrition related
to COVID-19. Troisième, while the exclusion criteria were selected to reduce noise in the BOLD sig-
nal, it altered the representativeness of the sample’s age due to increased head motion (see Sup-
porting Information). Enfin, the vector autoregression approach that GIMME uses assumes that
network edges are consistent across the time series (Gates et al., 2011). Cependant, this assumption
Neurosciences en réseau
805
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
/
/
t
e
d
toi
n
e
n
un
r
t
je
c
e
–
p
d
je
F
/
/
/
/
/
7
2
7
8
7
2
1
1
8
4
4
9
n
e
n
_
un
_
0
0
3
0
6
p
d
.
t
F
b
oui
g
toi
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
Adolescent reward network heterogeneity
may conflict with connectivity at different stages of the task used in this study (c'est à dire., win, perte,
neutral changes) or in other GIMME studies (par exemple., Dajani et al., 2020; Demidenko et al., 2022;
McCormick et al., 2019). Further work with precision imaging approaches may increase the
power to model multiple exogenous task events to help address this limitation.
Conclusion
Dans l'ensemble, our results show substantial qualitative heterogeneity in reward networks across early
adolescents such that an aggregate model does not adequately represent individual-level
models. These findings suggest that group-average comparisons may have limited utility to
examine associations between network functioning and behavioral and clinical outcomes,
as findings may not be generalizable to the individual. While previous studies have identified
subgroups of distinct network processes and detected clinical differences between them, nous
found little validity in a subgrouping solution and no behavioral differences between sub-
groupes. In contrast, individualized network features were associated with multiple reward-
related behavioral outcomes and risk for substance use problems, showing promise for
detecting brain-behavior relationships specific to the individual.
REMERCIEMENTS
Training in GIMME methodology was gained through Dr. Kathleen Gates’s workshop at the
University of Pittsburgh’s Summer Methodology Series.
INFORMATIONS À L'APPUI
Supporting information for this article is available at https://doi.org/10.1162/netn_a_00306.
CONTRIBUTIONS DES AUTEURS
Matthew Mattoni: Conceptualisation; Conservation des données; Analyse formelle; Acquisition de financement;
Enquête; Méthodologie; Gestion de projet; Visualisation; Writing – Original draft;
Writing – Review & édition. David V. Forgeron: Méthodologie; Surveillance; Writing – Review &
édition. Thomas M. Olino: Conceptualisation; Analyse formelle; Acquisition de financement; Method-
ology; Gestion de projet; Surveillance; Rédaction – révision & édition.
INFORMATIONS SUR LE FINANCEMENT
Thomas M. Olino, National Institute of Mental Health (https://dx.doi.org/10.13039
/100000025), Award ID: R01 MH107495. Matthew Mattoni, Weinstein Family/Civic Founda-
tion Award. Publication of this article was funded in part by the Temple University Libraries
Open Access Publishing Fund.
RÉFÉRENCES
Anandakumar, J., Mills, K. L., Earl, E. UN., Irwin, L., Miranda-
Dominguez, O., Demeter, D. V., Walton-Weston, UN., Karalunas,
S., Nigg, J., & Fair, D. UN. (2018). Individual differences in func-
tional brain connectivity predict temporal discounting preference
in the transition to adolescence. Developmental Cognitive Neu-
roscience, 34, 101–113. https://doi.org/10.1016/j.dcn.2018.07
.003, PubMed: 30121543
Angold, UN., Costello, E. J., Messer, S. C., & Pickles, UN. (1995).
Development of a short questionnaire for use in epidemiological
studies of depression in children and adolescents. International
Journal of Methods in Psychiatric Research, 5(4), 237–249.
Beltz, UN. M., & Gates, K. M.. (2017). Network mapping with
GIMME. Multivariate Behavioral Research, 52(6), 789–804.
https://doi.org/10.1080/00273171.2017.1373014, PubMed:
29161187
Benjamini, Y., & Hochberg, Oui. (1995). Controlling the false discov-
ery rate: A Practical and powerful approach to multiple testing.
Journal of the Royal Statistical Society: Série B (Methodological),
Neurosciences en réseau
806
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
/
t
/
e
d
toi
n
e
n
un
r
t
je
c
e
–
p
d
je
F
/
/
/
/
/
7
2
7
8
7
2
1
1
8
4
4
9
n
e
n
_
un
_
0
0
3
0
6
p
d
.
t
F
b
oui
g
toi
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
Adolescent reward network heterogeneity
57(1), 289–300. https://doi.org/10.1111/j.2517-6161.1995
.tb02031.x
Birn, R.. M., Molloy, E. K., Patriat, R., Parker, T., Meier, T. B., Kirk,
G. R., Naïr, V. UN., Meyerand, M.. E., & Prabhakaran, V. (2013). Le
effect of scan length on the reliability of resting-state fMRI con-
nectivity estimates. NeuroImage, 83, 550–558. https://est ce que je.org/10
.1016/j.neuroimage.2013.05.099, PubMed: 23747458
Brucar, L. R., Feczko, E., Fair, D. UN., & Zilverstand, UN. (2023). Actuel
approaches in computational psychiatry for the data-driven
identification of brain-based subtypes. Biological Psychiatry,
93(8), 704–716. https://doi.org/10.1016/j.biopsych.2022.12.020,
PubMed: 36841702
Carver, C. S., & Blanc, T. L. (1994). Behavioral inhibition, behav-
ioral activation, and affective responses to impending reward and
punishment: The BIS/ BAS Scales. Journal of Personality and
Social Psychology, 67(2), 319–333. https://doi.org/10.1037
/0022-3514.67.2.319
Casey, B. J., Cannonier, T., Conley, M.. JE., Cohen, UN. O., Barch,
D. M., Heitzeg, M.. M., Soules, M.. E., Teslovich, T., Dellarco,
D. V., Garavan, H., Orr, C. UN., Wager, T. D., Banich, M.. T., Speer,
N. K., Sutherland, M.. T., Riedel, M.. C., Dick, UN. S., Bjork, J.. M.,
Thomas, K. M., … ABCD Imaging Acquisition Workgroup.
(2018). The Adolescent Brain Cognitive Development (ABCD)
étude: Imaging acquisition across 21 des sites. Developmental Cog-
nitive Neuroscience, 32, 43–54. https://doi.org/10.1016/j.dcn
.2018.03.001, PubMed: 29567376
Casey, B. J., Getz, S., & Galvan, UN. (2008). The adolescent brain.
Developmental Review, 28(1), 62–77. https://est ce que je.org/10.1016/j
.dr.2007.08.003, PubMed: 18688292
Casey, B. J., Heller, UN. S., Gee, D. G., & Cohen, UN. Ô. (2019).
Development of the emotional brain. Neuroscience Letters,
693, 29–34. https://doi.org/10.1016/j.neulet.2017.11.055,
PubMed: 29197573
Costa Dias, T. G., Iyer, S. P., Carpenter, S. D., Cary, R.. P., Wilson,
V. B., Mitchell, S. H., Nigg, J.. T., & Fair, D. UN. (2015). Character-
izing heterogeneity in children with and without ADHD based
on reward system connectivity. Developmental Cognitive Neuro-
science, 11, 155–174. https://doi.org/10.1016/j.dcn.2014.12
.005, PubMed: 25660033
Costello, E. J., Copeland, W., & Angold, UN. (2011). Trends in psy-
chopathology across the adolescent years: What changes when
children become adolescents, and when adolescents become
adultes? Journal of Child Psychology and Psychiatry, and Allied
Disciplines, 52(10), 1015–1025. https://doi.org/10.1111/j.1469
-7610.2011.02446.X, PubMed: 21815892
Cservenka, UN. (2016). Neurobiological phenotypes associated with
a family history of alcoholism. Drug and Alcohol Dependence,
158, 8–21. https://doi.org/10.1016/j.drugalcdep.2015.10.021,
PubMed: 26559000
Dajani, D. R., Odriozola, P., Winters, M., Voorhies, W., Marcano,
S., Baez, UN., Gates, K. M., Dick, UN. S., & Uddin, L. Q. (2020).
Measuring cognitive flexibility with the flexible item selection
task: From fMRI adaptation to individual connectome mapping.
Journal des neurosciences cognitives, 32(6), 1026–1045. https://est ce que je
.org/10.1162/jocn_a_01536, PubMed: 32013686
Demidenko, M.. JE., Huntley, E. D., Weigard, UN. S., Keating, D. P., &
Beltz, UN. M.. (2022). Neural heterogeneity underlying late ado-
lescent motivational processing is linked to individual
differences in behavioral sensation seeking. Journal of Neurosci-
ence Research, 100(3), 762–779. https://doi.org/10.1002/jnr
.25005, PubMed: 35043448
Desikan, R.. S., Ségonne, F., Fischl, B., Quinn, B. T., Dickerson,
B. C., Blacker, D., Buckner, R.. L., Dale, UN. M., Maguire, R.. P.,
Hyman, B. T., Albert, M.. S., & Killiany, R.. J.. (2006). An automated
labeling system for subdividing the human cerebral cortex on
MRI scans into gyral based regions of interest. NeuroImage,
31(3), 968–980. https://doi.org/10.1016/j.neuroimage.2006.01
.021, PubMed: 16530430
Drysdale, UN. T., Grosenick, L., Downar, J., Dunlop, K., Mansouri, F.,
Meng, Y., Fetcho, R.. N., Zebley, B., Oathes, D. J., Etkin, UN.,
Schatzberg, UN. F., Sudheimer, K., Keller, J., Mayberg, H. S.,
Gunning, F. M., Alexopoulos, G. S., Fox, M.. D., Pascual-Leone,
UN., Voss, H. U., … Liston, C. (2017). Resting-state connectivity
biomarkers define neurophysiological subtypes of depression.
Nature Medicine, 23(1), 28–38. https://doi.org/10.1038/nm
.4246, PubMed: 27918562
Duda, M., Iraji, UN., Ford, J.. M., Lim, K. O., Mathalon, D. H.,
Mueller, B. UN., Potkin, S. G., Preda, UN., Van Erp, T. G. M., &
Calhoun, V. D. (2023). Reliability and clinical utility of spatially
constrained estimates of intrinsic functional networks from very
short fMRI scans. Human Brain Mapping, 44(6), 2620–2635.
https://doi.org/10.1002/hbm.26234, PubMed: 36840728
Élise, L. K., & Rothbart, M.. (2001). Early Adolescent Temperament
Questionnaire—Revised (EAT-R). APA PsycTests. https://doi.org
/10.1037/t07624-000
Esteban, O., Birman, D., Shaer, M., Koyejo, O., Poldrack, R., &
Gorgolewski, K. (2017). MRIQC: Advancing the automatic pre-
diction of image quality in MRI from unseen sites. PLoS One.
12(9), e018466 https://doi.org/10.1371/journal.pone.0184661,
PubMed: 28945803
Esteban, O., Markiewicz, C. J., Blair, R.. W., Moodie, C. UN., Isik, UN. JE.,
Erramuzpe, UN., Kent, J.. D., Goncalves, M., DuPre, E., Snyder, M.,
Oya, H., Ghosh, S. S., Wright, J., Durnez, J., Poldrack, R.. UN., &
Gorgolewski, K. J.. (2019). fMRIPrep: A robust preprocessing
pipeline for functional MRI. Nature Methods, 16(1), 111–116.
https://doi.org/10.1038/s41592-018-0235-4, PubMed: 30532080
Esteban, O., Markiewicz, C. J., Brûlures, C., Goncalves, M., Jarecka,
D., Ziegler, E., Berleant, S., Élise, D. G., Pinsard, B., Madison, C.,
Waskom, M., Notter, M.. P., Clark, D., Manhães-Savio, UN., Clark, D.,
Jordan, K., Dayan, M., Halchenko, Oui. O., Loney, F., … Ghosh, S.
(2022un). nipy/nipype: 1.7.1. Zenodo. https://doi.org/10.5281
/zenodo.6415183
Esteban, O., Markiewicz, C. J., Goncalves, M., Kent, J.. D., DuPre,
E., Ciric, R., Salo, T., Pinsard, B., Blair, R.. W., Poldrack, R.. UN., &
Gorgolewski, K. J.. (2022b). fMRIPrep: A robust preprocessing
pipeline for functional MRI. Zenodo. https://doi.org/10.5281
/zenodo.6476576
Feczko, E., & Fair, D. UN. (2020). Methods and challenges for
assessing heterogeneity. Biological Psychiatry, 88(1), 9–17.
https://doi.org/10.1016/j.biopsych.2020.02.015, PubMed:
32386742
Feczko, E., Miranda-Dominguez, O., Marr, M., Graham, UN. M.,
Nigg, J.. T., & Fair, D. UN. (2019). The heterogeneity problem:
Approaches to identify psychiatric subtypes. Tendances cognitives
les sciences, 23(7), 584–601. https://doi.org/10.1016/j.tics.2019.03
.009, PubMed: 31153774
Neurosciences en réseau
807
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
/
/
t
e
d
toi
n
e
n
un
r
t
je
c
e
–
p
d
je
F
/
/
/
/
/
7
2
7
8
7
2
1
1
8
4
4
9
n
e
n
_
un
_
0
0
3
0
6
p
d
t
.
F
b
oui
g
toi
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
Adolescent reward network heterogeneity
D'abord, M.. B., Williams, J.. B. W, Karg, R.. S., & Spitzer, R.. L. (2016).
User’s guide for the SCID-5-CV Structured Clinical Interview for
DSM-5 disorders: Clinical version (pp. XII, 158). American Psy-
chiatric Publishing.
Fischer, UN. S., Ellwood-Lowe, M.. E., Colich, N. L., Cichocki, UN., Ho,
T. C., & Gotlib, je. H. (2019). Reward-circuit biomarkers of risk
and resilience in adolescent depression. Journal of Affective
Disorders, 246, 902–909. https://doi.org/10.1016/j.jad.2018.12
.104, PubMed: 30795497
Pêcheur, UN. J., Medaglia, J.. D., & Jeronimus, B. F. (2018). Lack of
group-to-individual generalizability is a threat to human subjects
recherche. Proceedings of the National Academy of Sciences of the
United States of America, 115(27), E6106–E6115. https://doi.org
/10.1073/pnas.1711978115, PubMed: 29915059
Forbes, E. E., Christopher May, J., Siegle, G. J., Ladouceur, C. D.,
Ryan, N. D., Carter, C. S., Birmaher, B., Axelson, D. UN., & Dahl,
R.. E. (2006). Reward-related decision-making in pediatric major
depressive disorder: Une étude IRMf. Journal of Child Psychology
and Psychiatry, 47(10), 1031–1040. https://doi.org/10.1111/j
.1469-7610.2006.01673.X, PubMed: 17073982
Forbes, E. E., & Dahl, R.. E. (2012). Research review: Altered reward
function in adolescent depression: What, when and how? Journal
of Child Psychology and Psychiatry, 53(1), 3–15. https://doi.org
/10.1111/j.1469-7610.2011.02477.x, PubMed: 22117893
Forbes, E. E., Hariri, UN. R., Martine, S. L., Silk, J.. S., Moyles, D. L.,
Pêcheur, P.. M., Brun, S. M., Ryan, N. D., Birmaher, B., Axelson,
D. UN., & Dahl, R.. E. (2009). Altered striatal activation predicting
real-world positive affect in adolescent major depressive disor-
der. The American Journal of Psychiatry, 166(1), 64–73. https://
doi.org/10.1176/appi.ajp.2008.07081336, PubMed: 19047324
Friston, K. J.. (2011). Functional and effective connectivity: UN
revoir. Brain Connectivity, 1(1), 13–36. https://doi.org/10.1089
/brain.2011.0008, PubMed: 22432952
Friston, K. J., Harrison, L., & Penny, W. (2003). Dynamic causal
modelling. NeuroImage, 19(4), 1273–1302. https://est ce que je.org/10
.1016/s1053-8119(03)00202-7, PubMed: 12948688
Garlaschelli, D. (2009). The weighted random graph model. Nouveau
Journal of Physics, 11(7), 073005. https://doi.org/10.1088/1367
-2630/11/7/073005
Gates, K. M., Pêcheur, Z. F., Arizmendi, C., Henry, T. R., Duffy, K. UN.,
& Mucha, P.. J.. (2019). Assessing the robustness of cluster solu-
tions obtained from sparse count matrices. Psychological
Methods, 24(6), 675–689. https://doi.org/10.1037/met0000204,
PubMed: 30742473
Gates, K. M., voie, S. T., Varangis, E., Giovanello, K., & Guiskewicz,
K. (2017). Unsupervised classification during time-series model
bâtiment. Multivariate Behavioral Research, 52(2), 129–148.
https://doi.org/10.1080/00273171.2016.1256187, PubMed:
27925768
Gates, K. M., & Molenaar, P.. C. M.. (2012). Group search algorithm
recovers effective connectivity maps for individuals in homoge-
neous and heterogeneous samples. NeuroImage, 63(1), 310–319.
https://doi.org/10.1016/j.neuroimage.2012.06.026, PubMed:
22732562
Gates, K. M., Molenaar, P.. C. M., Hillary, F. G., Ram, N., & Rovine,
M.. J.. (2010). Automatic search for fMRI connectivity mapping:
An alternative to Granger causality testing using formal equiva-
lences among SEM path modeling, VAR, and unified SEM.
NeuroImage, 50(3), 1118–1125. https://est ce que je.org/10.1016/j
.neuroimage.2009.12.117, PubMed: 20060050
Gates, K. M., Molenaar, P.. C. M., Hillary, F. G., & Slobounov, S.
(2011). Extended unified SEM approach for modeling
event-related fMRI data. NeuroImage, 54(2), 1151–1158.
https://doi.org/10.1016/j.neuroimage.2010.08.051, PubMed:
20804852
Gates, K. M., Molenaar, P.. C. M., Iyer, S. P., Nigg, J.. T., & Fair, D. UN.
(2014). Organizing Heterogeneous samples using community
detection of GIMME-derived resting state functional networks.
PLoS One, 9(3), e91322. https://doi.org/10.1371/journal.pone
.0091322, PubMed: 24642753
Gorgolewski, K., Brûlures, C., Madison, C., Clark, D., Halchenko, Y.,
Waskom, M., & Ghosh, S. (2011). Nipype: A flexible, lightweight
and extensible neuroimaging data processing framework in
Python. Frontiers in Neuroinformatics, 5, 13. https://est ce que je.org/10
.3389/fninf.2011.00013, PubMed: 21897815
Grant, B. F., & Dawson, D. UN. (1998). Age of onset of drug use and
its association with DSM-IV drug abuse and dependence: Results
from the National Longitudinal Alcohol Epidemiologic Survey.
Journal of Substance Abuse, 10(2), 163–173. https://est ce que je.org/10
.1016/s0899-3289(99)80131-X, PubMed: 9854701
Gratton, C., Kraus, B. T., Vert, D. J., Gordon, E. M., Laumann,
T. O., Nelson, S. M., Dosenbach, N. U. F., & Petersen, S. E.
(2020). Defining individual-specific functional neuroanatomy for
precision psychiatry. Biological Psychiatry, 88(1), 28–39. https://
doi.org/10.1016/j.biopsych.2019.10.026, PubMed: 31916942
Heitzeg, M.. M., Cope, L. M., Martz, M.. E., & Hardee, J.. E. (2015).
Neuroimaging risk markers for substance abuse: Recent findings
on inhibitory control and reward system functioning. Actuel
Addiction Reports, 2(2), 91–103. https://doi.org/10.1007/s40429
-015-0048-9, PubMed: 26236575
Hoffmann, J.. P., & Cerbone, F. G. (2002). Parental substance use
disorder and the risk of adolescent drug abuse: An event history
analyse. Drug and Alcohol Dependence, 66(3), 255–264.
https://doi.org/10.1016/S0376-8716(02)00005-4, PubMed:
12062460
Kashyap, R., Bhattacharjee, S., Yeo, B. T. T., & Chen, S. H. UN.
(2020). Maximizing dissimilarity in resting state detects heteroge-
neous subtypes in healthy population associated with high sub-
stance use and problems in antisocial personality. Cerveau humain
Cartographie, 41(5), 1261–1273. https://doi.org/10.1002/ hbm
.24873, PubMed: 31773817
Kaufman, UN. S., & Kaufman, N. L. (2013). Kaufman Brief Intelli-
gence Test (2nd ed.). Wiley.
Kaurin, UN., Dombrovski, UN. Y., Hallquist, M.. N., & Wright, UN. G. C.
(2022). Integrating a functional view on suicide risk into idio-
graphic statistical models. Behaviour Research and Therapy,
150, 104012. https://doi.org/10.1016/j.brat.2021.104012,
PubMed: 35121378
Kazdin, UN. E. (1989). Evaluation of the Pleasure Scale in the
assessment of anhedonia in children. Journal of the American
Academy of Child and Adolescent Psychiatry, 28(3), 364–372.
https://doi.org/10.1097/00004583-198905000-00010, PubMed:
2738002
Keren, H., O’Callaghan, G., Vidal-Ribas, P., Buzzell, G. UN.,
Brotman, M.. UN., Leibenluft, E., Pan, P.. M., Meffert, L., kaiser,
UN., Wolke, S., Pine, D. S., & Stringaris, UN. (2018). Reward
Neurosciences en réseau
808
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
/
t
/
e
d
toi
n
e
n
un
r
t
je
c
e
–
p
d
je
F
/
/
/
/
/
7
2
7
8
7
2
1
1
8
4
4
9
n
e
n
_
un
_
0
0
3
0
6
p
d
.
t
F
b
oui
g
toi
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
Adolescent reward network heterogeneity
processing in depression: A conceptual and meta-analytic review
across fMRI and EEG studies. American Journal of Psychiatry,
175(11), 1111–1120. https://doi.org/10.1176/appi.ajp.2018
.17101124, PubMed: 29921146
Khundrakpam, B. S., Lewis, J.. D., Zhao, L., Chouinard-Decorte,
F., & Evans, UN. C. (2016). Brain connectivity in normally devel-
oping children and adolescents. NeuroImage, 134, 192–203.
https://doi.org/10.1016/j.neuroimage.2016.03.062, PubMed:
27054487
Kovacs, M.. (1985). The Children’s Depression, Inventory (CDI).
Psychopharmacology Bulletin, 21(4), 995–998. PubMed:
4089116
Laumann, T. O., Gordon, E. M., Adeyemo, B., Snyder, UN. Z., Joo,
S. J., Chen, M.-Y., Gilmore, UN. W., McDermott, K. B., Nelson,
S. M., Dosenbach, N. U. F., Schlaggar, B. L., Mumford, J.. UN.,
Poldrack, R.. UN., & Petersen, S. E. (2015). Functional system
and areal organization of a highly sampled individual human
brain. Neurone, 87(3), 657–670. https://est ce que je.org/10.1016/j.neuron
.2015.06.037, PubMed: 26212711
Liang, S., Deng, W., Li, X., Greenshaw, UN. J., Wang, Q., Li, M.,
Ma, X., Bai, T.-J., Bo, Q.-J., Cao, J., Chen, G.-M., Chen, W.,
Cheng, C., Cheng, Y.-Q., Cui, X.-L., Duan, J., Fang, Y.-R.,
Gong, Q.-Y., Guo, W.-B., … Li, T. (2020). Biotypes of major
depressive disorder: Neuroimaging evidence from resting-state
default mode network patterns. NeuroImage: Clinique, 28,
102514. https://doi.org/10.1016/j.nicl.2020.102514, PubMed:
33396001
Marquand, UN. F., Rezek, JE., Buitelaar, J., & Beckmann, C. F. (2016).
Understanding heterogeneity in clinical cohorts using normative
models: Beyond case-control studies. Biological Psychiatry,
80(7), 552–561. https://doi.org/10.1016/j.biopsych.2015.12
.023, PubMed: 26927419
McClure, S. M., Laibson, D. JE., Löwenstein, G., & Cohen, J.. D.
(2004). Separate neural systems value immediate and delayed
monetary rewards. Science, 306(5695), 503–507. https://doi.org
/10.1126/science.1100907, PubMed: 15486304
McCormick, E. M., Gates, K. M., & Telzer, E. H. (2019). Model-
based network discovery of developmental and performance-
related differences during risky decision-making. NeuroImage,
188, 456–464. https://doi.org/10.1016/j.neuroimage.2018.12
.042, PubMed: 30579902
Medaglia, J.. D., Ramanathan, D. M., Venkatesan, U. M., & Hillary,
F. G. (2011). The challenge of non-ergodicity in network neuro-
science. Réseau, 22(1–4), 148–153. https://doi.org/10.3109
/09638237.2011.639604, PubMed: 22149675
Miller, C. H., Hamilton, J.. P., Sacchet, M.. D., & Gotlib, je. H.
(2015). Meta-analysis of functional neuroimaging of major
depressive disorder in youth. JAMA Psychiatry, 72(10), 1045–1053.
https://doi.org/10.1001/jamapsychiatry.2015.1376, PubMed:
26332700
Molenaar, P.. C. M.. (2004). A manifesto on psychology as idio-
graphic science: Bringing the person back into scientific psychol-
ogy, this time forever. Measurement: Interdisciplinary Research
and Perspectives, 2(4), 201–218. https://doi.org/10.1207
/s15366359mea0204_1
O’Callaghan, G., & Stringaris, UN. (2019). Reward processing in
adolescent depression across neuroimaging modalities. Zeitschrift
fur Kinder- und Jugendpsychiatrie und Psychotherapie, 47(6),
535–541. https://doi.org/10.1024/1422-4917/a000663, PubMed:
30957688
Pons, P., & Latapy, M.. (2005). Computing communities in large
networks using random walks. In P. Yolum, T. Güngör, F.
Gürgen, & C. Özturan (Éd.), Computer and Information
Sciences—ISCIS 2005 (pp. 284–293). Springer. https://doi.org
/10.1007/11569596_31
Poudel, UN., & Gautam, S. (2017). Age of onset of substance use and
psychosocial problems among individuals with substance use
disorders. BMC Psychiatry, 17(1), 10. https://doi.org/10.1186
/s12888-016-1191-0, PubMed: 28077106
Prix, R.. B., Gates, K., Kraynak, T. E., Thase, M.. E., & Siegle, G. J..
(2017un). Data-driven subgroups in depression derived from
directed functional connectivity paths at rest. Neuropsychophar-
macology, 42(13), 2623–2632. https://doi.org/10.1038/npp
.2017.97, PubMed: 28497802
Prix, R.. B., voie, S., Gates, K., Kraynak, T. E., Horner, M.. S.,
(2017b). Parsing heterogeneity
J..
Thase, M.. E., & Siegle, G.
in the brain connectivity of depressed and healthy adults
during positive mood. Biological Psychiatry, 81(4), 347–357.
https://doi.org/10.1016/j.biopsych.2016.06.023, PubMed:
27712830
Richards, J., Plate, R., & Ernst, M.. (2013). A systematic review of
fMRI reward paradigms used in studies of adolescents vs. adultes:
The impact of task design and implications for understanding
neurodevelopment. Neurosciences & Biobehavioral Reviews,
37(5), 976–991. https://doi.org/10.1016/j.neubiorev.2013.03
.004, PubMed: 23518270
Seitzman, B. UN., Gratton, C., Laumann, T. O., Gordon, E. M.,
Adeyemo, B., Dworetsky, UN., Kraus, B. T., Gilmore, UN. W., Berger,
J.. J., Ortega, M., Nguyen, UN., Vert, D. J., McDermott, K. B.,
Nelson, S. M., Lessov-Schlaggar, C. N., Schlaggar, B. L.,
Dosenbach, N. U. F., & Petersen, S. E. (2019). Trait-like variants
in human functional brain networks. Actes de la Nationale
Académie des Sciences, 116(45), 22851–22861. https://est ce que je.org/10
.1073/pnas.1902932116, PubMed: 31611415
Solmi, M., Radua, J., Olivola, M., Croce, E., Soardo, L., Salazar de
Pablo, G., Il Shin, J., Kirkbride, J.. B., Jones, P., Kim, J.. H., Kim,
J.. Y., Carvalho, UN. F., Seeman, M.. V., Correll, C. U., & Fusar-Poli,
P.. (2021). Age at onset of mental disorders worldwide:
Large-scale meta-analysis of 192 epidemiological studies.
Molecular Psychiatry, 27(1), 281–295. https://est ce que je.org/10.1038
/s41380-021-01161-7, PubMed: 34079068
Squeglia, L. M., & Cservenka, UN. (2017). Adolescence and drug use
vulnerability: Findings from neuroimaging. Current Opinion in
Behavioral Sciences, 13, 164–170. https://est ce que je.org/10.1016/j
.cobeha.2016.12.005, PubMed: 28111629
Van Essen, D. C., Forgeron, S. M., Barch, D. M., Behrens, T. E. J.,
Yacoub, E., Voiture d'Ugur, K., & WU-Minn HCP Consortium. (2013).
The WU-Minn Human Connectome Project: An overview. Neu-
roImage, 80, 62–79. https://doi.org/10.1016/j.neuroimage.2013
.05.041, PubMed: 23684880
Whitfield-Gabrieli, S., & Nieto-Castanon, UN. (2012). Connecticut: A func-
tional connectivity toolbox for correlated and anticorrelated
brain networks. Brain Connectivity, 2(3), 125–141. https://est ce que je
.org/10.1089/brain.2012.0073, PubMed: 22642651
Xia, M., Wang, J., & Il, Oui. (2013). BrainNet viewer: A network
visualization tool for human brain connectomics. PLoS ONE,
Neurosciences en réseau
809
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
/
t
/
e
d
toi
n
e
n
un
r
t
je
c
e
–
p
d
je
F
/
/
/
/
/
7
2
7
8
7
2
1
1
8
4
4
9
n
e
n
_
un
_
0
0
3
0
6
p
d
.
t
F
b
oui
g
toi
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
Adolescent reward network heterogeneity
8(7), e68910. https://doi.org/10.1371/journal.pone.0068910,
PubMed: 23861951
Zhu, T., Becquey, C., Chen, Y., Lejuez, C. W., Li, C.-S. R., & Bi, J..
(2022). Identifying alcohol misuse biotypes from neural connec-
tivity markers and concurrent genetic associations. Translational
Psychiatry, 12(1), 253. https://doi.org/10.1038/s41398-022
-01983-1, PubMed: 35710901
Zhuo, C., Li, G., Lin, X., Jiang, D., Xu, Y., Tian, H., Wang, W., &
Song, X. (2019). The rise and fall of MRI studies in major depres-
sive disorder. Translational Psychiatry, 9, 335. https://est ce que je.org/10
.1038/s41398-019-0680-6, PubMed: 31819044
Zou, H. (2006). The adaptive lasso and its oracle properties. Journal
of the American Statistical Association, 101(476), 1418–1429.
https://doi.org/10.1198/016214506000000735
je
D
o
w
n
o
un
d
e
d
F
r
o
m
h
t
t
p
:
/
/
d
je
r
e
c
t
.
m
je
t
.
/
/
t
e
d
toi
n
e
n
un
r
t
je
c
e
–
p
d
je
F
/
/
/
/
/
7
2
7
8
7
2
1
1
8
4
4
9
n
e
n
_
un
_
0
0
3
0
6
p
d
.
t
F
b
oui
g
toi
e
s
t
t
o
n
0
7
S
e
p
e
m
b
e
r
2
0
2
3
Neurosciences en réseau
810