Finding Positive Meaning in Negative Experiences Engages
Ventral Striatal and Ventromedial Prefrontal Regions
Associated with Reward Valuation
Bruce P. Doré1, Chelsea Boccagno1, Daisy Burr1, Alexa Hubbard2, Kan Long1,
Jochen Weber1, Yaakov Stern1, and Kevin N. Ochsner1
D
o
w
n
l
o
a
d
e
d
f
r
o
m
Abstract
■ Neuroimaging research has identified systems that facilitate
minimizing negative emotion, but how the brain is able to
transform the valence of an emotional response from nega-
tive to positive is unclear. Behavioral and psychophysiological
studies suggest a distinction between minimizing reappraisal,
which entails diminishing the arousal elicited by negative stim-
uli, and positive reappraisal, which instead changes the emo-
tional valence of arousal from negative to positive. Here we
show that successful minimizing reappraisal tracked with de-
creased activity in the amygdala, but successful positive re-
appraisal tracked with increased activity in regions involved
in computing reward value, including the ventral striatum
and ventromedial pFC (vmPFC). Moreover, positive reappraisal
enhanced positive connectivity between vmPFC and amygdala,
and individual differences in positive connectivity between
vmPFC and amygdala, ventral striatum, dorsomedial pFC, and
dorsolateral pFC predicted greater positive reappraisal success.
These data broaden models of emotion regulation as quanti-
tative dampening of negative emotion and identify activity in a
network of brain valuation, arousal, and control regions as a
neural basis for the ability to create positive meaning from nega-
tive experiences. ■
INTRODUCTION
Distressing life events are an unavoidable aspect of human
experience. Many areas of research converge to demon-
strate that how we respond to these events matters for
our long-term mental and physical well-being (Gross,
2015; Folkman, 2008; Bonanno, 2004). Beyond simply
dampening negative reactions to stressors, the ability to
find positive meaning in potentially traumatic circum-
stances has been consistently identified as a protective
factor against the harmful effects of stress and a critical
pathway to resilience—the maintenance or enhance-
ment of function in the face of potential trauma (Russo,
Murrough, Han, Charney, & Nestler, 2012; Helgeson,
Reynolds, & Tomich, 2006; Tugade & Fredrickson, 2004).
For example, behavioral studies show that expressing
positive emotion when speaking about a recent bereave-
ment predicts improved psychological functioning over
several years (Bonanno & Keltner, 1997) and finding ben-
efits or life lessons in experiencing a heart attack predicts
improvement in health and decreased risk for a sub-
sequent attack (Affleck, Tennen, Croog, & Levine, 1987).
Despite its clear importance, virtually nothing is known
about the neural mechanisms that underlie the ability to
1Columbia University, 2New York University
© 2016 Massachusetts Institute of Technology
find positive meaning and, in turn, generate positive emo-
tion in the face of adversity. Addressing this gap in the
literature builds our basic understanding of the neuro-
biology underlying human emotional capabilities and
helps clarify the precise brain processes that may fail in
psychopathology (Kring & Sloan, 2009).
Focusing on the positive implications of a distressing
experience can be understood as an instance of re-
appraisal, a strategy for emotion regulation that entails
reframing the meaning of an emotional situation. Labora-
tory research has typically considered reappraisal as a uni-
form strategy, ignoring potentially crucial differences
between different ways of reappraising (Buhle et al., 2014;
Ochsner, Silvers, & Buhle, 2012). In particular, a funda-
mental distinction can be made between minimizing re-
appraisal, which entails focusing on unemotional aspects
of a negative experience to dampen negative feelings, and
positive reappraisal, which entails focusing on positive as-
pects of a negative experience to enhance positive feelings
(McRae & Mauss, in press; McRae, Ciesielski, & Gross, 2012).
Prior neuroimaging studies indicate that reappraisal of
negative stimuli elicits increased activity in brain regions
associated with domain-general cognitive control, includ-
ing ventrolateral pFC (vlPFC), dorsolateral pFC (dlPFC),
dorsomedial pFC (dmPFC), and lateral parietal cortex,
alongside decreased activity in brain regions associated
Journal of Cognitive Neuroscience 29:2, pp. 235–244
doi:10.1162/jocn_a_01041
l
l
/
/
/
/
j
f
/
t
t
i
t
.
:
/
/
h
t
t
p
:
/
D
/
o
m
w
i
n
t
o
p
a
r
d
c
e
.
d
s
f
i
r
o
l
m
v
e
h
r
c
p
h
a
d
i
i
r
r
e
.
c
c
t
.
o
m
m
/
j
e
d
o
u
c
n
o
/
c
a
n
r
a
t
r
i
t
i
c
c
l
e
e
–
p
–
d
p
d
2
f
9
/
2
2
9
2
/
3
2
5
/
1
2
9
3
5
5
2
/
3
1
2
7
6
8
o
5
c
7
n
9
_
5
a
/
_
j
0
o
1
c
0
n
4
1
_
a
p
_
d
0
1
b
0
y
4
g
1
u
.
e
p
s
t
d
o
f
n
b
0
y
8
S
M
e
I
p
T
e
m
L
i
b
b
e
r
r
a
2
r
0
i
2
3
e
s
/
j
/
t
.
f
u
s
e
r
o
n
1
7
M
a
y
2
0
2
1
with emotional responding, including the amygdala (Buhle
et al., 2014). However, converging lines of evidence
suggest that positive reappraisal may engage distinct neural
systems.
First, although early theories of amygdala function
associated this region with negative emotions like fear
(LeDoux, 1998), more recent work has shown that the
amygdala responds more generally to material that is rel-
evant to our goals, including stimuli that are positive,
arousing, novel, ambiguous, or surprising (Cunningham
& Brosch, 2012; Whalen, 1998). Second, behavioral and
psychophysiological studies have observed that, unlike
minimizing reappraisal, positive reappraisal does not di-
minish arousal elicited by negative stimuli but instead
changes the emotional valence of this arousal from neg-
ative to positive (McRae et al., 2012; Shiota & Levenson,
2012). Third, neuroscience research has characterized a
system of brain regions, including the ventral striatum
(which includes nucleus accumbens) and ventromedial
pFC (vmPFC) that are involved in computing reward value
and thought to underlie positive emotional responding
across a wide variety of contexts (Bartra, McGuire, &
Kable, 2013; Haber & Knutson, 2010; Sabatinelli, Bradley,
Lang, Costa, & Versace, 2007; Mobbs, Greicius, Abdel-
Azim, Menon, & Reiss, 2003).
To the extent that positive reappraisal successfully
elicits positive emotion, it should not modulate the amyg-
dala, but rather the ventral striatum and vmPFC regions
associated with reward. Moreover, vmPFC and amygdala
are reciprocally interconnected via dense white matter
projections (Freese & Amaral, 2009) and have been ob-
served to show both negative ( Johnstone, van Reekum,
Urry, Kalin, & Davidson, 2007) and positive patterns of
coactivation (Erk et al., 2010; Banks, Eddy, Angstadt,
Nathan, & Phan, 2007) during emotion regulation. How-
ever, it is not yet clear what this coactivation reflects.
Following theories of vmPFC as a region computing an
integrative and contextually sensitive signal for positive
value (Roy, Shohamy, & Wager, 2012; Rangel & Hare,
2010), it may be that positive connectivity between amyg-
dala and vmPFC reflects the operation of a neural circuit
supporting changes in the valence of affective arousal from
negative to positive. If so, vmPFC should show enhanced
positive connectivity with the amygdala during positive
reappraisal.
Taken together, these separate lines of work suggest
that positive reappraisal and minimizing reappraisal may
show similarities in engagement of control-related pre-
frontal and parietal regions, but differences in the extent
to which they modulate brain regions associated with
affective arousal and positive value as well as the extent
to which they rely on vmPFC to amygdala connectivity.
We used behavioral and fMRI methods to address
these questions. In a preliminary behavioral analysis, we
used affect ratings made in the fMRI scanner and text
data we collected in a separate cohort of participants to
examine the effects of these experimental conditions on
emotional experience and the linguistic content of each
type of reappraisal. In our neuroimaging study, we tested
four hypotheses about the neural mechanisms of positive
versus minimizing reappraisals. First, we hypothesized
that positive and minimizing reappraisal would engage
common regions of prefrontal and parietal cortex. Sec-
ond, we hypothesized that positive reappraisal would
modulate activity in the amygdala to a lesser extent than
minimizing reappraisal. Third, we hypothesized that pos-
itive reappraisal would modulate activity in the ventral
striatum and vmPFC to a greater extent than minimizing
reappraisal. Finally, we hypothesized that successful pos-
itive reappraisal would rely on enhanced connectivity be-
tween the amygdala and the vmPFC.
METHODS
Participants
Participants were 20 adults (12 women, 8 men) recruited
from the New York City area (mean age = 24.6, SD = 4.5)
and screened to confirm that they could read and speak
fluently in English, had normal or corrected-to-normal
vision, had never been diagnosed with a psychiatric dis-
order, did not report current depressive symptoms (i.e.,
scored below 16 on the Center for Epidemiologic Studies
Depression Scale), and had no conditions that contra-
indicated magnetic resonance imaging (MRI). A separate
cohort of 20 adults (11 women, 9 men; mean age = 21.5,
SD = 3.1) completed a behavioral study in which they
typed texts representing the content of their reappraisals
and natural responses. All study procedures were approved
by the Columbia University institutional review board.
Image Acquisition
Data were collected with a 3T MR750 (GE Healthcare Sys-
tems, Milwaukee, WI) magnet and a 32-channel RF head coil.
Structural volumes were acquired using a high-resolution
T1-weighted sagittal 3-D BRAVO sequence yielding 1 mm3
isotropic voxel size. Functional volumes were acquired
using a T2*-sensitive EPI sequence with a repetition time of
2000 msec, an echo time of 25 msec, a 77° flip angle, and a
field of view of 19.2 cm consisting of 45 interleaved 3-mm
slices acquired parallel to the AC–PC axis. Four runs of
185 repetition times were collected. Each run began with
8 sec of fixation, and the corresponding four volumes were
discarded.
Task Design
Participants completed an experimental task consisting
of six functional runs. One half (three consecutive runs)
of the experimental task was devoted to positive re-
appraisal (with positive reappraisal trials, negative image
trials, and neutral image trials intermixed), and the other
half (three consecutive runs) was devoted to minimizing
236
Journal of Cognitive Neuroscience
Volume 29, Number 2
D
o
w
n
l
o
a
d
e
d
f
r
o
m
l
l
/
/
/
/
j
t
t
f
/
i
t
.
:
/
/
h
t
t
p
:
/
D
/
o
m
w
i
n
t
o
p
a
r
d
c
e
.
d
s
f
i
r
o
l
m
v
e
h
r
c
p
h
a
d
i
i
r
r
e
.
c
c
t
.
o
m
m
/
j
e
d
o
u
c
n
o
/
c
a
n
r
a
t
r
i
t
i
c
c
l
e
e
–
p
–
d
p
d
2
f
9
/
2
2
9
2
/
3
2
5
/
1
2
9
3
5
5
2
/
3
1
2
7
6
8
o
5
c
7
n
9
_
5
a
/
_
j
0
o
1
c
0
n
4
1
_
a
p
_
d
0
1
b
0
y
4
g
1
u
.
e
p
s
t
d
o
f
n
b
0
y
8
S
M
e
I
p
T
e
m
L
i
b
b
e
r
r
a
2
r
0
i
2
3
e
s
/
j
/
t
.
f
u
s
e
r
o
n
1
7
M
a
y
2
0
2
1
reappraisal (with minimizing reappraisal trials, negative
image trials, and neutral image trials intermixed). The
order of positive and minimizing reappraisal was counter-
balanced across participants. Each run consisted of 15 trials
each: five negative image reappraisal trials, five negative
image natural response trials, and five neutral image trials.
The trial sequence, consisting of cue, image, ISI, rating
period (counterbalanced order), and intertrial interval, is
represented in Figure 1. Aversive images (mean valence =
2.49; mean arousal = 5.71) and neutral images (mean
valence = 5.41; mean arousal = 3.54) were selected from
the International Affective Picture System (Lang, Greenwald,
Bradley, & Hamm, 1993). Aversive images were counter-
balanced to experimental condition across participants.
Before scanning, participants completed experimenter-
guided training modules for both types of reappraisal,
which included examples of acceptable positive and mini-
mizing reappraisals and opportunities to practice. For
positive reappraisal, participants were told to think
about the depicted situations by focusing on potential
positive aspects or outcomes, and for minimizing re-
appraisal, to think about potential neutral aspects or
outcomes. For natural response trials, participants were
told to view and think about the images as they normally
would. Stimuli were presented with E-Prime 1.2 (Psychology
Software Tools, Pittsburgh, PA). Participants made behav-
ioral responses on a five-button response pad.
A separate cohort of participants completed a behav-
ioral study in which they received identical training pro-
cedures, viewed a subset of the same images (38 of the
60 used in the MRI study; mean valence = 2.41; mean
arousal = 5.97), and were asked to type for each image
a short phrase or sentence representing the content of
their natural response, minimizing reappraisal, or positive
reappraisal.
Behavioral Analysis
Behavioral ratings of negative and positive affect tended
to be negatively correlated within participants (mean r =
−.36 for natural response; mean r = −.43 for positive
reappraisal; mean r = −.29 for minimizing reappraisal).
These ratings were differenced (positive − negative) to
yield the overall valence of reported experience on each
trial of the task. Next, valence reports were averaged
within participants and analyzed with planned t tests to
test hypotheses about effects of positive reappraisal and
Figure 1. Trial sequence for the fMRI reappraisal task.
minimizing reappraisal on the valence of affective expe-
rience. Ratings of negative and positive affect were also used
to compute a positive reappraisal success score for each par-
ticipant, reflecting a combined score for success in changing
affective valence by down-regulating negative affect (mean
Natural Response rating − mean Positive Reappraisal rating)
and up-regulating positive affect (mean Positive Reappraisal
rating − mean Natural Response rating).
Reappraisal and natural response texts (generated by a
separate cohort) were processed with Linguistic Inquiry
and Word Count (liwc.net), a prominent dictionary-based
text analysis software, yielding proportions of words from
Linguistic Inquiry and Word Count negative emotion
(negemo) and positive emotion ( posemo) categories
used by participants on each trial. These text proportions
were also averaged within participants and analyzed with
planned t tests. Finally, texts were also processed with a
dictionary-free text analytic method (Fellows, 2014) to
descriptively visualize relative frequencies of particular
words across positive reappraisal, minimizing reappraisal,
and natural response conditions.
fMRI Analysis
Preprocessing/General Linear Model
Data preprocessing was conducted with SPM8 (Wellcome
Department of Cognitive Neurology, University College
London) and consisted of slice-time correction, realign-
ment, coregistration of functional and structural images,
and normalization to the standard Montreal Neurological
Institute brain by segmentation of the structural image
and applying the parameters from this step during warping.
Normalized images were interpolated to 3 mm3 voxels and
smoothed with a 6-mm Gaussian kernel.
First-level (individual) general linear modelanalyses
were implemented in NeuroElf v1.0 (neuroelf.net), using
robust estimation via iteratively reweighted least squares
(see Wager, Keller, Lacey, & Jonides, 2005), to reduce the
influence of time-series outliers (e.g., sudden head motion
or other artifactual changes in signal intensity) on the esti-
mated model parameters. Cue, stimulus, and response
periods of each trial were modeled as boxcar functions
convolved with the canonical hemodynamic response
function. Separate stimulus regressors were entered for
positive reappraisal, natural response (within positive re-
appraisal block), neutral images (within positive reappraisal
block), minimizing reappraisal, natural response (within
minimizing reappraisal block) conditions, and neutral
images (within minimizing reappraisal block) as well as
condition-centered parametric regressors for trial-by-
trial reports of valence (i.e., positive affect rating minus
negative affect rating) in each of these four conditions.
Motion parameters and a high pass temporal filter for
128 sec were added as regressors of no interest.
Second-level (group) random-effects analyses were
implemented in NeuroElf v1.0. All activation peaks are
Doré et al.
237
D
o
w
n
l
o
a
d
e
d
f
r
o
m
l
l
/
/
/
/
j
f
/
t
t
i
t
.
:
/
/
h
t
t
p
:
/
D
/
o
m
w
i
n
t
o
p
a
r
d
c
e
.
d
s
f
i
r
o
l
m
v
e
h
r
c
p
h
a
d
i
i
r
r
e
.
c
c
t
.
o
m
m
/
j
e
d
o
u
c
n
o
/
c
a
n
r
a
t
r
i
t
i
c
c
l
e
e
–
p
–
d
p
d
2
f
9
/
2
2
9
2
/
3
2
5
/
1
2
9
3
5
5
2
/
3
1
2
7
6
8
o
5
c
7
n
9
_
5
a
/
_
j
0
o
1
c
0
n
4
1
_
a
p
_
d
0
1
b
0
y
4
g
1
u
.
e
p
s
t
d
o
f
n
b
0
y
8
S
M
e
I
p
T
e
m
L
i
b
b
e
r
r
a
2
r
0
i
2
3
e
s
/
j
/
.
f
t
u
s
e
r
o
n
1
7
M
a
y
2
0
2
1
reported in standard Montreal Neurological Institute space.
ROIs for bilateral amygdala (L −25, −2, −18; R 27, −2,
−28; 5324 mm3) and nucleus accumbens (L −9, 11, −7;
R 9, 12, −6; 1422 mm3) were defined anatomically, using
volumes from the Harvard–Oxford probabilistic atlas
(thresholded at 25% probability), and an ROI for vmPFC
(peak 0, 46, −6; 4860 mm3) was defined on the basis of
a meta-analysis identifying brain regions carrying a mono-
tonic, modality-independent signal for subjective reward
value (shown in Figure 9 of Bartra et al., 2013). An addi-
tional anatomically and functionally constrained amygdala
ROI was defined by computing the contrast of [natural
response (both blocks)] > [neutral images (both blocks)]
within the mask for bilateral amygdala at p < .05, iden-
tifying a cluster of 33 contiguous voxels within the left
amygdala. This targeted ROI allowed us to test for modu-
lation of the region of the amygdala that was responsive
to the presentation of aversive images. For effect size
estimation across entire ROIs, beta estimates were ex-
tracted, and means and standard errors were computed.
For search analyses within ROIs, small-volume correction
was applied to achieve a corrected p value of <.05, using
Gaussian random field theory to estimate the number of
independent resolution elements in each ROI. For whole-
brain analyses, cluster-extent thresholding was applied,
using AlphaSim Monte Carlo simulation to achieve a
whole-brain FWE rate corrected p value of <.05, with a
primary threshold of p = .005 (i.e., z ≈ 2.6) and smooth-
ness parameters (9.7–11.4 mm) estimated from the re-
siduals of each statistical map to determine a minimum
number of contiguous voxels, k (from 103 to 142 for indi-
vidual contrasts; 10 for a conjunction map).
Functional Connectivity
To examine changes in coactivation across brain regions,
we conducted a psychophysiological interaction (PPI)
analysis. As a seed, we used the region of vmPFC that
parametrically tracked affect ratings for positive re-
appraisal to a greater extent than natural response and
minimizing reappraisal trials, as identified in a whole-
brain parametric analysis. For this PPI, regressors were
entered for each experimental condition, the seed region
time series, and interaction terms for the seed region time
series and the experimental conditions. In a group-level
test, we contrasted the PPI terms for the positive re-
appraisal condition over the natural response and mini-
mizing reappraisal conditions (i.e., [1 −0.5 −0.5]) to
identify regions of the brain that change in connectivity
with vmPFC for positive reappraisal relative to the other
task conditions. Next, we correlated this PPI map with suc-
cess scores to identify regions for which connectivity was
correlated with behavioral success in positive reappraisal.
RESULTS
Manipulation Check: Positive Reappraisal and
Minimizing Reappraisal Have Distinct Effects on
Affective Experience and Linguistic Behavior
First, we asked whether positive reappraisal, minimizing
reappraisal, and natural responses were associated with
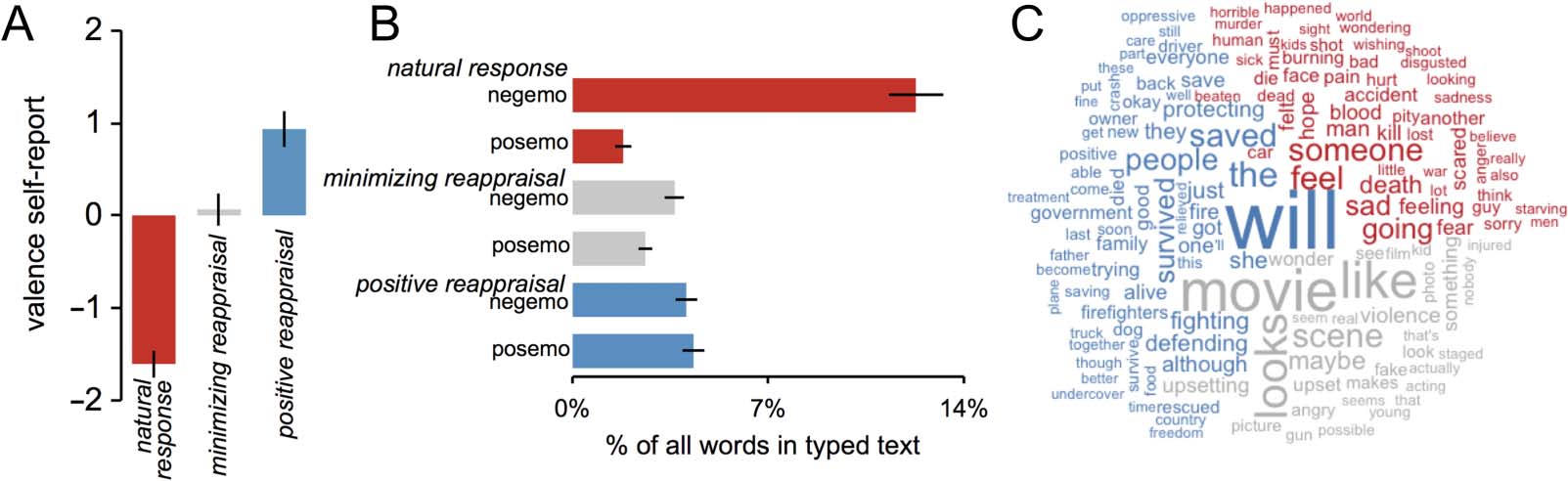
differences in self-reported affect. As expected, relative
to the natural response condition, when using minimiz-
ing reappraisal participants reported feeling less negative
(reaching neutral valence), t(19) = 10.71, p < .0001, and
when using positive reappraisal participants reported
feeling less negative and more positive (i.e., engendering
positively valenced responses), t(19) = 18.16, p < .0001
(see Figure 2A).
To provide insight into the content of reappraisals, in
a separate cohort of participants we asked whether word
use differed in brief typed texts representing the content
of their natural responses, positive reappraisals, and
minimizing reappraisals. We found an interaction of
Condition (natural response, minimizing reappraisal, posi-
tive reappraisal) and Word category (negative emotion
words, positive emotion words), F(1, 19) = 57.87, p <
.0001, on proportion of emotion words used in these
texts. Compared with natural response texts, we found
that positive reappraisal texts had fewer negative emo-
tion words, t(19) = 10.31, p < .0001, and more positive
Figure 2. (A) Effects of experimental condition on self-reported valence of affective experience. (B) Effects of experimental condition on use of
words from negative emotion (negemo) and positive emotion (posemo) categories. (C) Comparison cloud visualizing individual words that show the
highest relative frequencies of use for natural response (red), minimizing reappraisal (gray), and positive reappraisal (blue) conditions. Means ± SE.
238
Journal of Cognitive Neuroscience
Volume 29, Number 2
D
o
w
n
l
o
a
d
e
d
f
r
o
m
l
l
/
/
/
/
j
t
t
f
/
i
t
.
:
/
/
h
t
t
p
:
/
D
/
o
m
w
i
n
t
o
p
a
r
d
c
e
.
d
s
f
i
r
o
l
m
v
e
h
r
c
p
h
a
d
i
i
r
r
e
.
c
c
t
.
o
m
m
/
j
e
d
o
u
c
n
o
/
c
a
n
r
a
t
r
i
t
i
c
c
l
e
e
-
p
-
d
p
d
2
f
9
/
2
2
9
2
/
3
2
5
/
1
2
9
3
5
5
2
/
3
1
2
7
6
8
o
5
c
7
n
9
_
5
a
/
_
j
0
o
1
c
0
n
4
1
_
a
p
_
d
0
1
b
0
y
4
g
1
u
.
e
p
s
t
d
o
f
n
b
0
y
8
S
M
e
I
p
T
e
m
L
i
b
b
e
r
r
a
2
r
0
i
2
3
e
s
/
j
.
/
f
t
u
s
e
r
o
n
1
7
M
a
y
2
0
2
1
D
o
w
n
l
o
a
d
e
d
f
r
o
m
l
l
/
/
/
/
j
f
/
t
t
i
t
.
:
/
/
h
t
t
p
:
/
D
/
o
m
w
i
n
t
o
p
a
r
d
c
e
.
d
s
f
i
r
o
l
m
v
e
h
r
c
p
h
a
d
i
i
r
r
e
.
c
c
t
.
o
m
m
/
j
e
d
o
u
c
n
o
/
c
a
n
r
a
t
r
i
t
i
c
c
l
e
e
-
p
-
d
p
d
2
f
9
/
2
2
9
2
/
3
2
5
/
1
2
9
3
5
5
2
/
3
1
2
7
6
8
o
5
c
7
n
9
_
5
a
/
_
j
0
o
1
c
0
n
4
1
_
a
p
_
d
0
1
b
0
y
4
g
1
u
.
e
p
s
t
d
o
f
n
b
0
y
8
S
M
e
I
p
T
e
m
L
i
b
b
e
r
r
a
2
r
0
i
2
3
e
s
/
j
/
.
f
t
u
s
e
r
o
n
1
7
M
a
y
2
0
2
1
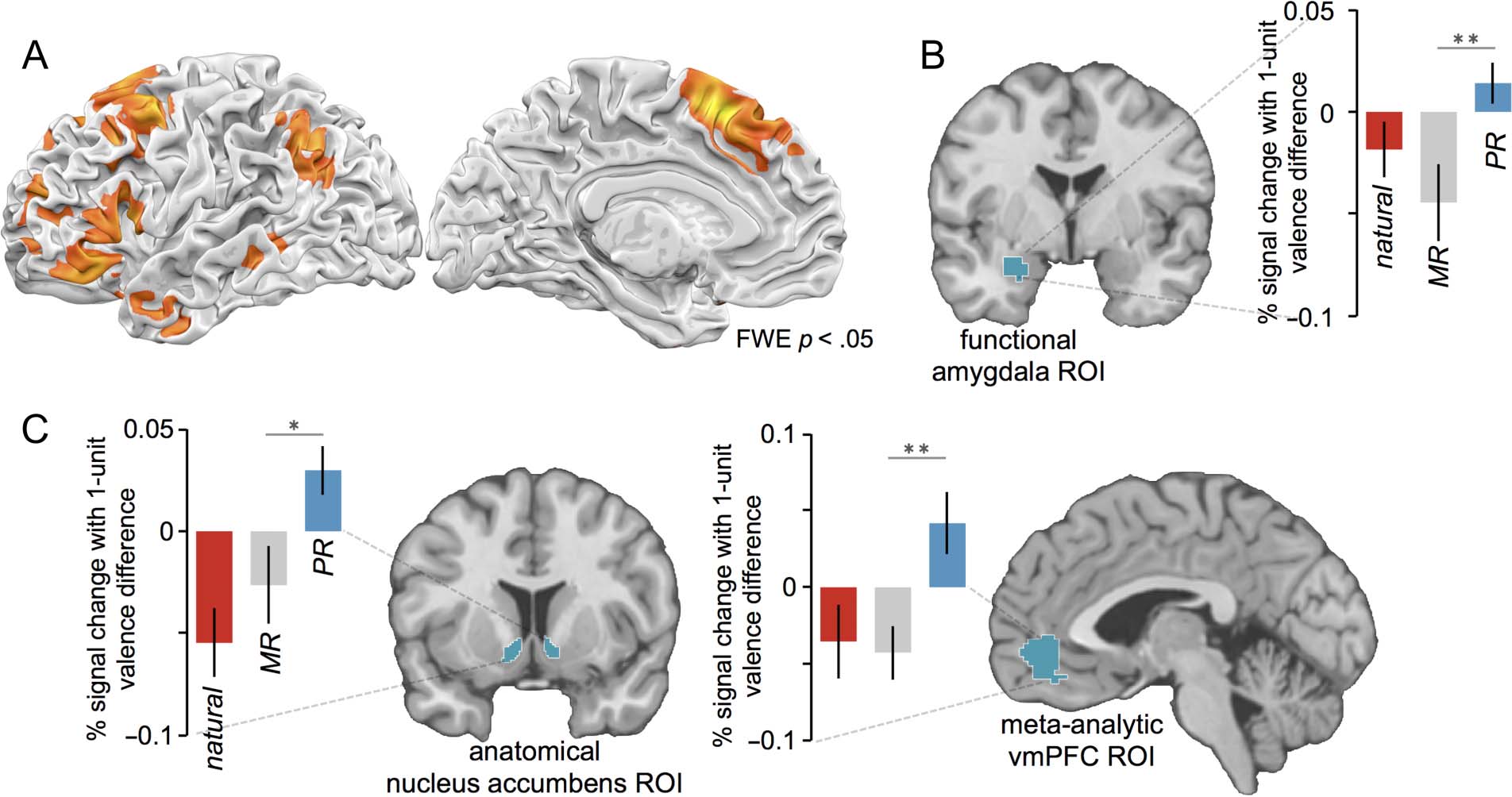
Figure 3. (A) Common regions of activation for minimizing and positive reappraisal, identified with a conjunction of reappraisal contrasts.
(B) Mean betas (±SE ) extracted from an anatomically and functionally constrained amygdala ROI indicate more negative trial-to-trial modulation
of the amygdala for minimizing reappraisal than positive reappraisal. (C) Mean betas (±SE ) extracted from an anatomical nucleus accumbens ROI
and a meta-analytically defined vmPFC ROI indicate more positive trial-to-trial modulation for positive reappraisal than minimizing reappraisal.
emotion words, t(19) = 3.47, p = .002. Similarly, compared
with natural response texts, minimizing reappraisal texts
had fewer negative emotion words, t(19) = 8.46, p <
.0001, and more positive emotion words at a trending level
of significance, t(19) = 1.85, p = .08. Finally, positive re-
appraisal texts had more positive emotion words than did
minimizing reappraisal texts, t(19) = 2.96, p = .008 (see
Figure 2B). Next we used a data-driven text analytic method
to identify words occurring at disproportionately high fre-
quency for texts from a given condition. Words identified
by this analysis are displayed in Figure 2C, a descriptive
visualization of differences in word use across condition
(text size reflects relative frequency for a given experimental
condition relative to the other two).
Hypothesis 1: Positive Reappraisal and Minimizing
Reappraisal Engage Common Regions of
Prefrontal and Parietal Cortex
Next we turned to data from the fMRI study, focusing on
the period of image presentation during which reapprai-
sals are being implemented. To identify the regions of
the brain associated with implementing positive reap-
praisal and minimizing reappraisal, we computed con-
trasts of both positive reappraisal > natural response
(within the positive reappraisal block) and minimizing
reappraisal > natural response (within the minimizing
reappraisal block) conditions.
For positive reappraisal, we found activation in bilat-
eral vlPFC (L −48, 27, −18; R 51, 24, −15), bilateral
dlPFC (L −33, 9, 51; R 18, 60, 30), bilateral dmPFC (L −3,
27, 33; R 6, 21, 33), left posterior parietal cortex (−45,
−57, 30), bilateral temporal lobe (L −57, −24, −15;
R 54, −12, −33), left dorsal caudate (−15, 9, 12) and left
putamen (−15, 12, 0). For minimizing reappraisal, we
found activation in left vlPFC (−48, 30, −9), left dlPFC
(−48, 18, 33), bilateral dmPFC (L −3, 27, 36; R 12, 18,
57), and left posterior parietal (−54, −66, 24). To identify
neural mechanisms common to both types of reappraisal,
we computed the conjunction of these two contrasts, re-
vealing activity in left vlPFC (−51, 15, 3), left dlPFC (−39,
21, 42), bilateral dmPFC (L −3, 27, 36; R 12, 18, 57), left
posterior parietal cortex (−51, −57, 36), and both anterior
(−42, −3, −42) and posterior (−60, −33, −6) regions of
left lateral temporal cortex (see Figure 3A). Considering
differences in the brain mechanisms associated with the
state of implementing positive reappraisals and mini-
mizing reappraisals, an interaction contrast of [positive
reappraisal > natural response] > [minimizing reappraisal >
natural response] resulted in no significant whole brain
clusters.
Hypothesis 2: Minimizing Reappraisal Modulates
Activity in the Amygdala More so than
Positive Reappraisal
Next we asked whether the two types of reappraisal dif-
fered in the extent to which they modulated activity in
the amygdala from trial to trial. We examined betas for
parametric valence regressors from an anatomically and
Doré et al.
239
functionally constrained ROI within the left amygdala,
representing the region of the amygdala sensitive to
the presentation of negative images. We found an inter-
action between Reappraisal tactic (positive reappraisal,
minimizing reappraisal) and Trial type (reappraisal
trial, natural response trial), F(1, 19) = 5.55, p = .03,
for the parametric betas extracted from the amygdala.
Critically, there was a difference, t(19) = 2.87, p =
.01, between the parametric effects for minimizing re-
appraisal (mean b = −.044, 95% CI [−.081, −.007])
and positive reappraisal (mean b = .014, 95% CI
[−.005, .034]), indicating that activity in this amygdala
ROI was negatively modulated from trial to trial for min-
imizing reappraisal (such that less amygdala activity was
apparent for trials for which less negatively valenced
affect was reported), but not for positive reappraisal
(see Figure 3B).
Hypothesis 3: Positive Reappraisal Modulates
Activity in the Ventral Striatum and vmPFC
More so than Minimizing Reappraisal or
Natural Responding
Next we asked whether positive reappraisal differs from
minimizing reappraisal and natural responding in the
extent to which it modulates activity in regions of the
brain associated with reward. For positive reappraisal,
we observed positive parametric tracking of affective
valence within the nucleus accumbens (L −9, 21, −9;
R 12, 15, −12; SVC p < .05) and vmPFC (6, 33, −15; SVC
p < .05) ROIs, such that greater activity was observed
in these regions for trials where participants reported
greater positive affect.
Extracting average parametric betas from bilateral
nucleus accumbens revealed an interaction between Re-
appraisal tactic and Trial type, F(1, 19) = 10.48, p =
.004. Critically, there was a difference, t(19) = 2.63, p =
.02, between parametric effects for positive reappraisal
(mean b = .030, 95% CI [.006, .053]) versus minimizing
reappraisal (mean b = −.026, 95% CI [−.063, .011])
and a difference, t(19) = 4.55, p = .0002, between para-
metric effects for positive reappraisal versus the inter-
mixed natural response condition (mean b = −.055,
95% CI [−.088, −.022]; see Figure 3C). Extracting average
parametric betas from the vmPFC revealed an interaction
between Reappraisal tactic and Trial type, F(1, 19) = 7.67,
p = .01. Critically, there was a difference, t(19) = 3.43,
p = .003, between parametric effects for positive re-
appraisal (mean b = .042, 95% CI [.002, .081]) versus
minimizing reappraisal (mean β = −.043, 95% CI [−.077,
−.001]) and a difference, t(19) = 2.60, p = .02, between
parametric effects for positive reappraisal versus the inter-
mixed natural response condition (mean b = −.035, 95%
CI [−.083, .012]; see Figure 3C).
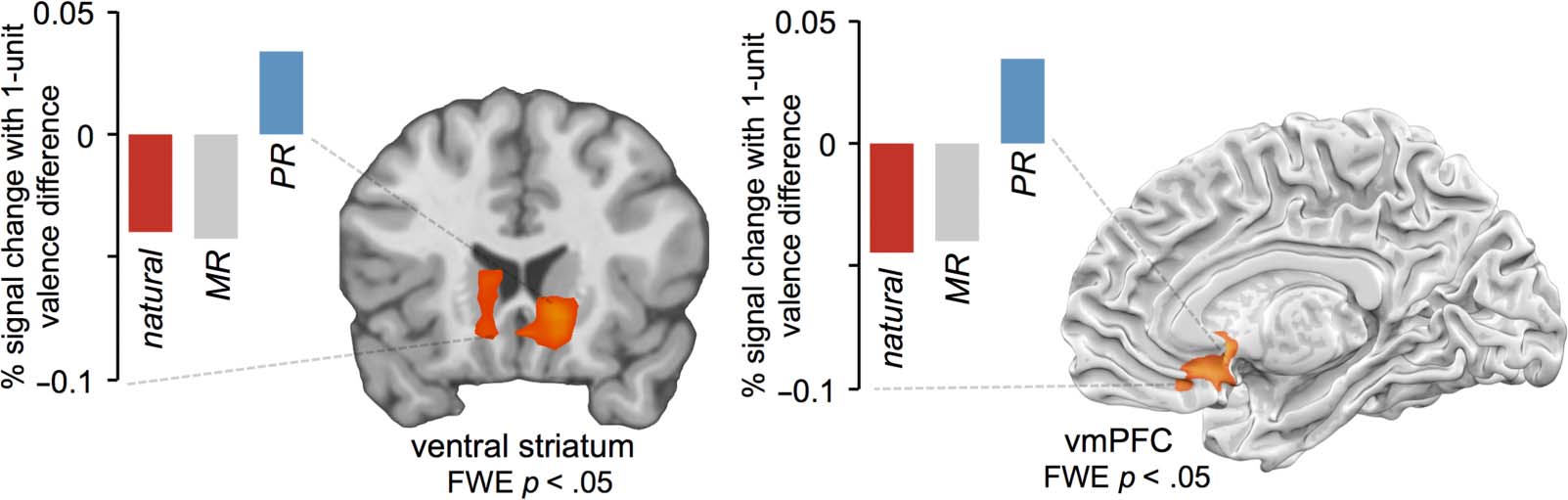
To follow up on these targeted ROI-based analyses, we
computed a whole-brain contrast of parametric maps to
identify regions, across the entire brain, that linearly
tracked affective valence ratings more for the positive
reappraisal condition than for the minimizing reappraisal
or intermixed natural response conditions (positive re-
appraisal > minimizing reappraisal + natural response).
Because stimuli were counterbalanced across conditions,
this analysis identified regions that tracked affect differ-
entially as a result of the manipulation of experimental
condition rather than as a result of stimulus factors (like
trial-to-trial variation in image intensity). This analysis re-
vealed significantly more positive parametric tracking of
affect for positive reappraisal (compared with minimizing
reappraisal and natural response conditions) within bi-
lateral ventral striatum (L 12, 18, −6; R −12, 21, −9) and
a subgenual region of right vmPFC (9, 27, −15; see Fig-
ure 4) and no other regions. In a follow-up analysis, we
asked what regions of the brain showed a full crossover
interaction of parametric effects [positive reappraisal −
intermixed natural response > minimizing reappraisal −
intermixed natural response]—similarly, this revealed
activity in ventral striatum (L −9, 18, −9; R 12, 18, −6)
and vmPFC (3, 21, −9) but no other regions. These
results indicate that ventral striatal and vmPFC regions
of the brain positively track reported affect for the positive
reappraisal condition to a greater extent than for the
minimizing reappraisal and natural response conditions.
Figure 4. Regions tracking trial-to-trial affect for positive reappraisal condition more so than minimizing reappraisal or natural response,
identified with a whole-brain contrast of parametric maps corrected at FWE p < .05. Mean betas shown for descriptive visualization—because
these betas were derived from clusters derived from a whole-brain search, we omit error bars and significance tests.
240
Journal of Cognitive Neuroscience
Volume 29, Number 2
D
o
w
n
l
o
a
d
e
d
f
r
o
m
l
l
/
/
/
/
j
t
t
f
/
i
t
.
:
/
/
h
t
t
p
:
/
D
/
o
m
w
i
n
t
o
p
a
r
d
c
e
.
d
s
f
i
r
o
l
m
v
e
h
r
c
p
h
a
d
i
i
r
r
e
.
c
c
t
.
o
m
m
/
j
e
d
o
u
c
n
o
/
c
a
n
r
a
t
r
i
t
i
c
c
l
e
e
-
p
-
d
p
d
2
f
9
/
2
2
9
2
/
3
2
5
/
1
2
9
3
5
5
2
/
3
1
2
7
6
8
o
5
c
7
n
9
_
5
a
/
_
j
0
o
1
c
0
n
4
1
_
a
p
_
d
0
1
b
0
y
4
g
1
u
.
e
p
s
t
d
o
f
n
b
0
y
8
S
M
e
I
p
T
e
m
L
i
b
b
e
r
r
a
2
r
0
i
2
3
e
s
/
j
t
f
/
.
u
s
e
r
o
n
1
7
M
a
y
2
0
2
1
D
o
w
n
l
o
a
d
e
d
f
r
o
m
l
l
/
/
/
/
j
t
t
f
/
i
t
.
:
/
/
h
t
t
p
:
/
D
/
o
m
w
i
n
t
o
p
a
r
d
c
e
.
d
s
f
i
r
o
l
m
v
e
h
r
c
p
h
a
d
i
i
r
r
e
.
c
c
t
.
o
m
m
/
j
e
d
o
u
c
n
o
/
c
a
n
r
a
t
r
i
t
i
c
c
l
e
e
-
p
-
d
p
d
2
f
9
/
2
2
9
2
/
3
2
5
/
1
2
9
3
5
5
2
/
3
1
2
7
6
8
o
5
c
7
n
9
_
5
a
/
_
j
0
o
1
c
0
n
4
1
_
a
p
_
d
0
1
b
0
y
4
g
1
u
.
e
p
s
t
d
o
f
n
b
0
y
8
S
M
e
I
p
T
e
m
L
i
b
b
e
r
r
a
2
r
0
i
2
3
e
s
/
j
.
f
/
t
u
s
e
r
o
n
1
7
M
a
y
2
0
2
1
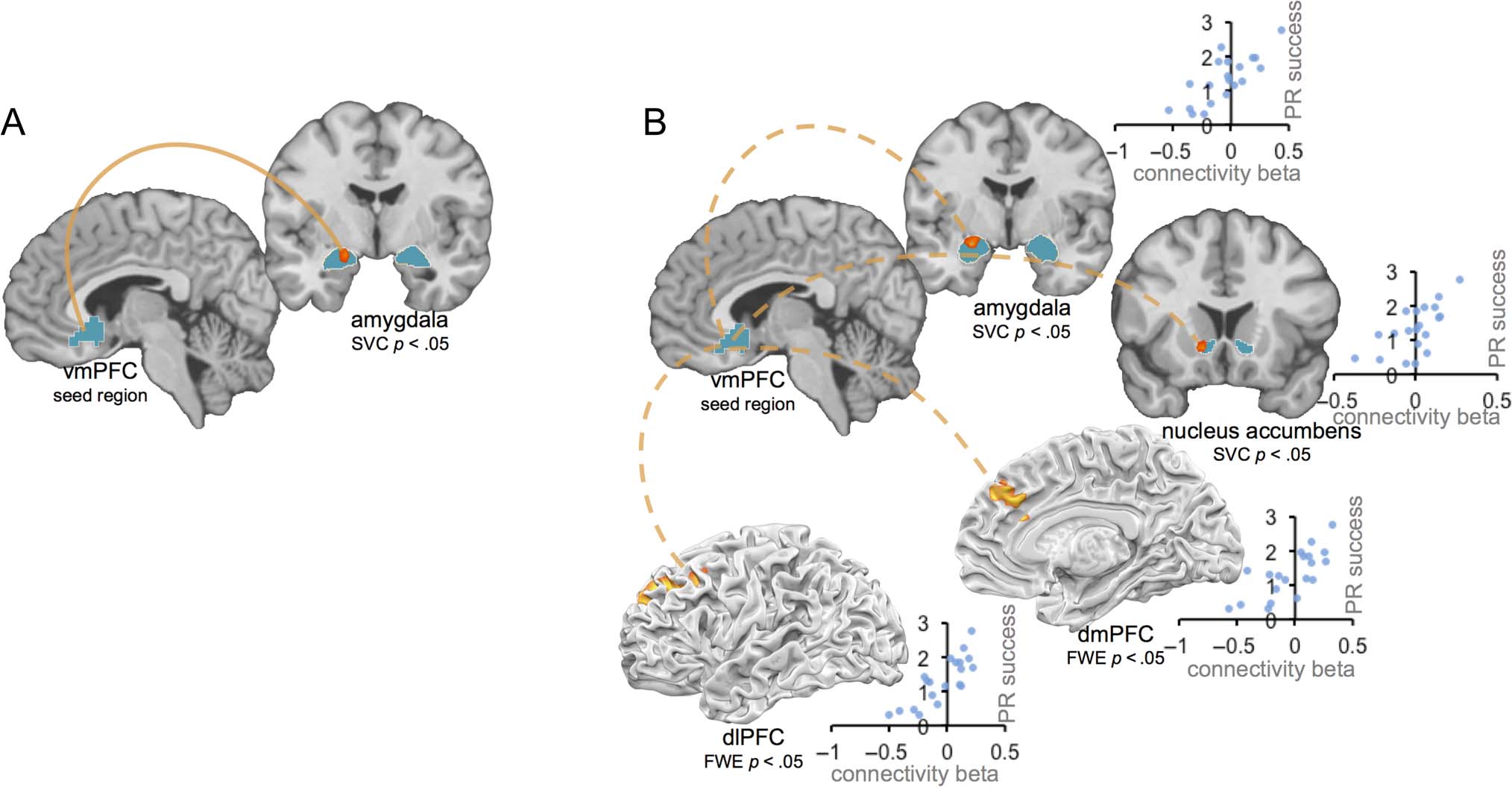
Figure 5. (A) Connectivity between vmPFC seed region and amygdala is enhanced for positive reappraisal, relative to natural response condition.
(B) More positive connectivity between vmPFC seed region and amygdala, nucleus accumbens, dmPFC, and dlPFC regions predicts greater
positive reappraisal success. Activations within amygdala and ventral striatum ROIs displayed at p < .005 uncorrected, and positive reappraisal
success by connectivity beta scatterplots shown for descriptive visualization.
Hypothesis 4a: Positive Reappraisal Enhances
Positive Connectivity between vmPFC
and Amygdala
Hypothesizing that positive reappraisal relies on en-
hanced connectivity between the vmPFC and amygdala,
we next conducted analyses that probed patterns of func-
tional coactivation across the brain. In a functional con-
nectivity (PPI) analysis, we used the region of the vmPFC
identified in the contrast of parametric maps above (rep-
resenting the vmPFC region tracking positive affect for
positive reappraisal) as a seed and asked what regions of
the brain show enhanced coactivation with this vmPFC
seed for positive reappraisal relative to minimizing re-
appraisal and natural response trials. Consistent with our
hypothesis, we found that vmPFC showed enhanced con-
nectivity during positive reappraisal with left amygdala
(peak −18, −6, −15; SVC p < .05; see Figure 5A). In a cor-
responding whole-brain analysis, no regions showed in-
creased connectivity at significant or trend-level thresholds.
Hypothesis 4b: More Positive vmPFC Connectivity
with Amygdala, Striatum, dmPFC, and dlPFC
Correlates with Positive Reappraisal Success
Lastly, we conducted a follow-up analysis to ask whether
differences in connectivity between vmPFC and other
brain regions underlie person-to-person variability in
positive reappraisal success. To do this, we computed a
map reflecting, for each voxel, the correlation between
each participant’s vmPFC connectivity beta (for positive
reappraisal vs. minimizing reappraisal and natural re-
sponse trial types) and his or her positive reappraisal suc-
cess score (reflecting success in up-regulating positive
affect and down-regulating negative affect on positive
reappraisal trials). We first examined a priori ROIs for
amygdala and nucleus accumbens and found that per-
son-to-person differences in positive reappraisal success
were correlated with the extent to which vmPFC showed
positive connectivity with bilateral amygdala (L −21, −3,
−12; R 18, −9, −12; SVC p < .05) and bilateral nucleus
accumbens (L −12, 15, −6; R 18, 9, −12; SVC p < .05).
Next we did a corresponding whole-brain analysis and
found that person-to-person differences in positive reap-
praisal success also were correlated with connectivity
betas for dmPFC (−3, 36, 33) and left dlPFC (−30, 30,
36; see Figure 5B). These analyses indicate that person-
to-person differences in connectivity between the vmPFC
and brain regions associated with affective arousal, posi-
tive value, and cognitive control were correlated with posi-
tive reappraisal success.
DISCUSSION
A primary form of resilient coping involves finding posi-
tive meaning in negative life experiences. Here we per-
formed the first test of the neural systems underlying
Doré et al.
241
this ability. Behaviorally, we found that positive reappraisal
increased positive emotion and changed use of valenced
linguistic content to a greater extent than did minimizing
reappraisal. With fMRI, we found that successful mini-
mizing reappraisal decreased activity in the amygdala, but
successful positive reappraisal increased activity in ventral
striatum and vmPFC regions associated with reward and
positive affect such that activity in these regions tracked re-
ports of more positive emotional experience. Finally, we
found that that positive reappraisal was associated with
enhanced connectivity between vmPFC and amygdala
and person-to-person differences in connectivity between
vmPFC and amygdala, ventral striatum, dmPFC, and dlPFC
predicted overall success in using positive reappraisal.
Implications for Neural Models of
Emotion Regulation
Where brain models of emotion regulation have previ-
ously highlighted the importance of interacting brain
systems for top–down control and bottom–up generation
of emotion, the results of this study extend these models
in three ways. First, our results indicate a specific role
for ventral striatal and vmPFC regions as a mechanism
specific to positive reappraisal and not minimizing re-
appraisal or simply looking at negative images. Previous
studies have not been designed to detect this pattern and
may have failed to do so because they used reappraisal
instructions that emphasized minimizing reappraisal (or
did not clearly distinguish between minimizing and pos-
itive reappraisal) or because they applied a contrast-based
analytic approach, rather than using parametric analyses
to systematically relate reports of affective experience to
underlying brain systems.
Second, brain models of emotion have highlighted that
regulatory strategies can bring about decreased activity
in the amygdala, concluding that diminished activity in
this region reflects a decrease in negative emotion (see
Denny, Ochsner, Weber, & Wager, 2014; Ohira et al.,
2006). However, our data suggest that modulation of
the amygdala is not the only brain indicator of successful
regulation of negative emotion. Using positive reappraisal
to enhance the positive meaning of a stimulus maintains
motivational relevance and affective arousal but shifts
experienced emotional valence from negative to positive
(McRae et al., 2012; Shiota & Levenson, 2012). Our data
identify engagement of ventral striatum and vmPFC re-
ward regions and enhanced vmPFC–amygdala positive
coupling as neural mechanisms that underpin this shift
in valence.
Third, the present data shed new light on models of
emotion regulation and self-regulation more generally,
positing that prefrontal control systems act in opposition
to systems involved in emotion generation. On such
views, increasing activity in one system, in the vmPFC,
for example, down-regulates activity in regions of the
other system, such as the amygdala (e.g., Diekhof, Geier,
Falkai, & Gruber, 2011; Schiller & Delgado, 2010). How-
ever, we did not observe that vmPFC down-regulates
activity in the amygdala during positive reappraisal but
rather that vmPFC and amygdala showed enhanced
positive coupling, and the extent of this coupling was
positively correlated with successfully changing one’s
emotional response from negative to positive. Thus,
our data align better with theories describing the vmPFC
as a nexus point that integrates information from cortical
and subcortical brain regions (Roy et al., 2012; Rangel
& Hare, 2010). Beyond the amygdala, our data suggest
that positive reappraisal success relies on vmPFC con-
nectivity with a suite of regions associated with affective
arousal, cognitive control, and positive value (see Barrett
& Satpute, 2013).
Implications for the Study of Healthy and Clinical
Variability in Affective Experience
Our findings also raise novel questions for understanding
clinical and subclinical variability in affective processes.
Previous work suggests that emotion regulation ability
can moderate the relationship between life stressors
and depressive symptoms (Troy, Wilhelm, Shallcross, &
Mauss, 2010), that regulation of positive affect may reflect
a transdiagnostic mechanism of emotional disturbance
(Carl, Soskin, Kerns, & Barlow, 2013), and that people
suffering from depression show reduced ability to sustain
affective responses to positive stimuli (Heller et al., 2009).
We found that individual differences in positive reap-
praisal success related to functional connectivity of vmPFC,
with dlPFC, dmPFC, amygdala, and ventral striatum, sug-
gesting that these regions form a functional network that
supports the ability to generate positive emotion in the
face of negative stimuli.
A crucial future direction will be to connect models of
the brain mechanisms underlying positive meaning to
the broader literature on resilience and recovery from
stressful life events. Although everyone experiences
stressors, how we respond to them can range from pro-
tracted disability to functional maintenance and growth.
Positive reappraisal ability, supported by vmPFC connec-
tivity with amygdala, ventral striatum, and pFC, could be
one factor that underlies such variability. Future work
that integrates models of genetic, environmental, and neu-
ral underpinnings of variable responses to stress may help
us understand where resilience comes from and how to
optimize it (see Russo et al., 2012; Bonanno, 2004). Impor-
tantly, future studies of this kind will benefit from larger
sample sizes, enabling more precise estimation of within-
and (especially) between-person brain–behavior relation-
ships and detection of smaller magnitude effects.
Conclusion
When life takes a turn for the worse, we cannot only
minimize our negative reactions but fundamentally
242
Journal of Cognitive Neuroscience
Volume 29, Number 2
D
o
w
n
l
o
a
d
e
d
f
r
o
m
l
l
/
/
/
/
j
f
/
t
t
i
t
.
:
/
/
h
t
t
p
:
/
D
/
o
m
w
i
n
t
o
p
a
r
d
c
e
.
d
s
f
i
r
o
l
m
v
e
h
r
c
p
h
a
d
i
i
r
r
e
.
c
c
t
.
o
m
m
/
j
e
d
o
u
c
n
o
/
c
a
n
r
a
t
r
i
t
i
c
c
l
e
e
-
p
-
d
p
d
2
f
9
/
2
2
9
2
/
3
2
5
/
1
2
9
3
5
5
2
/
3
1
2
7
6
8
o
5
c
7
n
9
_
5
a
/
_
j
0
o
1
c
0
n
4
1
_
a
p
_
d
0
1
b
0
y
4
g
1
u
.
e
p
s
t
d
o
f
n
b
0
y
8
S
M
e
I
p
T
e
m
L
i
b
b
e
r
r
a
2
r
0
i
2
3
e
s
/
j
/
.
f
t
u
s
e
r
o
n
1
7
M
a
y
2
0
2
1
transform them to experience positive emotions like
hope, gratitude, and love. Although negativity-minimizing
and positivity-enhancing forms of emotion regulation are
similar in that they rely on brain systems for controlled
processing, they are distinct in that minimizing reap-
praisal modulates brain systems associated with affective
arousal, whereas positive reappraisal modulates brain
systems associated with reward value. Here we describe
these distinct pathways to regulatory success and con-
sider their implications for our broader understanding
of emotion and its regulation. We hope that future work
will expand on our approach to uncover the basic bio-
logical mechanisms that underlie our ability to respond
flexibly and adaptively to the emotional challenges of
everyday life.
Acknowledgments
We thank Ehjeen Kim for assistance with data collection; Laura
Braunstein, Noam Zerubavel, Michael Gilead, Bryan Denny, and
the rest of the Columbia SCAN lab for guidance and helpful
discussion of this work; NIA grant R01AG043463-01 and Conte
grant PAR-11-126 for support to K. O., and NIA grant
R01AG026158-5 to Y. S. B. P. D. and K. N. O. designed the re-
search. B. P. D., C. B., D. B., A. H., and K. L., collected the data.
B. P. D. and J. W. analyzed the data. B. P. D. wrote the manu-
script with critical contributions from K. N. O., J. W., and Y. S.
Reprint requests should be sent to Bruce P. Doré, Department of
Psychology, Columbia University, Schermerhorn Hall, New York,
NY 10027, or via e-mail: brucedore@gmail.com.
REFERENCES
Affleck, G., Tennen, H., Croog, S., & Levine, S. (1987). Causal
attribution, perceived benefits, and morbidity after a heart
attack: An 8-year study. Journal of Consulting and Clinical
Psychology, 55, 29–35.
Banks, S. J., Eddy, K. T., Angstadt, M., Nathan, P. J., & Phan,
K. L. (2007). Amygdala–frontal connectivity during emotion
regulation. Social Cognitive and Affective Neuroscience, 2,
303–312.
Barrett, L. F., & Satpute, A. B. (2013). Large-scale brain networks
in affective and social neuroscience: Towards an integrative
functional architecture of the brain. Current Opinion in
Neurobiology, 23, 361–372.
Bartra, O., McGuire, J. T., & Kable, J. W. (2013). The valuation
system: A coordinate-based meta-analysis of BOLD fMRI
experiments examining neural correlates of subjective
value. Neuroimage, 76, 412–427.
Bonanno, G. A. (2004). Loss, trauma and human resilience:
Conceptual and empirical connections and separateness.
American Psychologist, 59, 20–28.
Bonanno, G. A., & Keltner, D. (1997). Facial expressions of
emotion and the course of conjugal bereavement. Journal of
Abnormal Psychology, 106, 126–137.
Buhle, J. T., Silvers, J. A., Wager, T. D., Lopez, R., Onyemekwu,
C., Kober, H., et al. (2014). Cognitive reappraisal of emotion:
A meta-analysis of human neuroimaging studies. Cerebral
Cortex, 24, 2981–2990.
Carl, J. R., Soskin, D. P., Kerns, C., & Barlow, D. H. (2013).
Positive emotion regulation in emotional disorders: A
theoretical review. Clinical Psychology Review, 33,
343–360.
Cunningham, W. A., & Brosch, T. (2012). Motivational salience
amygdala tuning from traits, needs, values, and goals.
Current Directions in Psychological Science, 21, 54–59.
Denny, B. T., Ochsner, K. N., Weber, J., & Wager, T. D. (2014).
Anticipatory brain activity predicts the success or failure of
subsequent emotion regulation. Social Cognitive and
Affective Neuroscience, 9, 403–411.
Diekhof, E. K., Geier, K., Falkai, P., & Gruber, O. (2011).
Fear is only as deep as the mind allows: A coordinate-based
meta-analysis of neuroimaging studies on the regulation of
negative affect. Neuroimage, 58, 275–285.
Erk, S., Mikschl, A., Stier, S., Ciaramidaro, A., Gapp, V., Weber,
B., et al. (2010). Acute and sustained effects of cognitive
emotion regulation in major depression. Journal of
Neuroscience, 30, 15726–15734.
Fellow, I. (2013). Wordcloud: Word Clouds, R package version
2.4. URL: http://CRAN.R-project.org/package=wordcloud.
Folkman, S. (2008). The case for positive emotions in the
stress process. Anxiety, Stress, and Coping, 21, 3–14.
Freese, J. L., & Amaral, D. G. (2009). Neuroanatomy of the
primate amygdala. In P. J. Whalen & E. A. Phelps (Eds.),
The human amygdala. New York: Guilford Press.
Gross, J. J. (2015). The extended process model of emotion
regulation: Elaborations, applications, and future directions.
Psychological Inquiry, 26, 130–137.
Haber, S. N., & Knutson, B. (2010). The reward circuit:
Linking primate anatomy and human imaging.
Neuropsychopharmacology, 35, 4–26.
Helgeson, V. S., Reynolds, K. A., & Tomich, P. L. (2006). A
meta-analytic review of benefit finding and growth. Journal
of Consulting and Clinical Psychology, 74, 797–816.
Heller, A. S., Johnstone, T., Shackman, A. J., Light, S. N.,
Peterson, M. J., Kolden, G. G., et al. (2009). Reduced capacity
to sustain positive emotion in major depression reflects
diminished maintenance of fronto-striatal brain activation.
Proceedings of the National Academy of Sciences, U.S.A.,
106, 22445–22450.
Johnstone, T., van Reekum, C. M., Urry, H. L., Kalin, N. H., &
Davidson, R. J. (2007). Failure to regulate: Counterproductive
recruitment of top–down prefrontal-subcortical circuitry
in major depression. Journal of Neuroscience, 27, 8877–8884.
Kring, A. M., & Sloan, D. M. (Eds.) (2009). Emotion regulation
and psychopathology: A transdiagnostic approach to
etiology and treatment. New York: Guilford Press.
Lang, P. J., Greenwald, M. K., Bradley, M. M., & Hamm, A. O.
(1993). Looking at pictures: Affective, facial, visceral, and
behavioral reactions. Psychophysiology, 30, 261–273.
LeDoux, J. (1998). Fear and the brain: Where have we been,
and where are we going?. Biological Psychiatry, 44,
1229–1238.
McRae, K., Ciesielski, B., & Gross, J. J. (2012). Unpacking
cognitive reappraisal: Goals, tactics, and outcomes. Emotion,
12, 250–255.
McRae, K., & Mauss, I. B. (in press). Increasing positive emotion
in negative contexts: Emotional consequences neural
correlates and implications for resilience. In J. Green,
I. Morrison, & M. Seligman (Eds.), Positive neuroscience.
Oxford: Oxford University Press.
Mobbs, D., Greicius, M. D., Abdel-Azim, E., Menon, V., &
Reiss, A. L. (2003). Humor modulates the mesolimbic
reward centers. Neuron, 40, 1041–1048.
Ochsner, K. N., Silvers, J. A., & Buhle, J. T. (2012). Functional
imaging studies of emotion regulation: A synthetic review
and evolving model of the cognitive control of emotion.
Annals of the New York Academy of Sciences, 1251,
E1–E24.
Ohira, H., Nomura, M., Ichikawa, N., Isowa, T., Iidaka, T., Sato,
A., et al. (2006). Association of neural and physiological
Doré et al.
243
D
o
w
n
l
o
a
d
e
d
f
r
o
m
l
l
/
/
/
/
j
t
t
f
/
i
t
.
:
/
/
h
t
t
p
:
/
D
/
o
m
w
i
n
t
o
p
a
r
d
c
e
.
d
s
f
i
r
o
l
m
v
e
h
r
c
p
h
a
d
i
i
r
r
e
.
c
c
t
.
o
m
m
/
j
e
d
o
u
c
n
o
/
c
a
n
r
a
t
r
i
t
i
c
c
l
e
e
-
p
-
d
p
d
2
f
9
/
2
2
9
2
/
3
2
5
/
1
2
9
3
5
5
2
/
3
1
2
7
6
8
o
5
c
7
n
9
_
5
a
/
_
j
0
o
1
c
0
n
4
1
_
a
p
_
d
0
1
b
0
y
4
g
1
u
.
e
p
s
t
d
o
f
n
b
0
y
8
S
M
e
I
p
T
e
m
L
i
b
b
e
r
r
a
2
r
0
i
2
3
e
s
/
j
/
t
f
.
u
s
e
r
o
n
1
7
M
a
y
2
0
2
1
responses during voluntary emotion suppression.
Neuroimage, 29, 721–733.
Rangel, A., & Hare, T. (2010). Neural computations associated
with goal-directed choice. Current Opinion in Neurobiology,
20, 262–270.
Roy, M., Shohamy, D., & Wager, T. D. (2012). Ventromedial
prefrontal-subcortical systems and the generation of affective
meaning. Trends in Cognitive Sciences, 16, 147–156.
Russo, S. J., Murrough, J. W., Han, M. H., Charney, D. S., &
Nestler, E. J. (2012). Neurobiology of resilience. Nature
Neuroscience, 15, 1475–1484.
Sabatinelli, D., Bradley, M. M., Lang, P. J., Costa, V. D., &
Versace, F. (2007). Pleasure rather than salience activates
human nucleus accumbens and medial prefrontal cortex.
Journal of Neurophysiology, 98, 1374–1379.
Schiller, D., & Delgado, M. R. (2010). Overlapping neural
systems mediating extinction, reversal and regulation of fear.
Trends in Cognitive Sciences, 14, 268–276.
Shiota, M. N., & Levenson, R. W. (2012). Turn down the volume
or change the channel? Emotional effects of detached versus
positive reappraisal. Journal of Personality and Social
Psychology, 103, 416–429.
Troy, A. S., Wilhelm, F. H., Shallcross, A. J., & Mauss, I. B.
(2010). Seeing the silver lining: Cognitive reappraisal ability
moderates the relationship between stress and depressive
symptoms. Emotion, 10, 783.
Tugade, M. M., & Fredrickson, B. L. (2004). Resilient individuals
use positive emotions to bounce back from negative
emotional experiences. Journal of Personality and Social
Psychology, 86, 320–333.
Wager, T. D., Keller, M. C., Lacey, S. C., & Jonides, J. (2005).
Increased sensitivity in neuroimaging analyses using robust
regression. Neuroimage, 26, 99–113.
Whalen, P. J. (1998). Fear, vigilance, and ambiguity: Initial
neuroimaging studies of the human amygdala. Current
Directions in Psychological Science, 7, 177–188.
D
o
w
n
l
o
a
d
e
d
f
r
o
m
l
l
/
/
/
/
j
t
t
f
/
i
t
.
:
/
/
h
t
t
p
:
/
D
/
o
m
w
i
n
t
o
p
a
r
d
c
e
.
d
s
f
i
r
o
l
m
v
e
h
r
c
p
h
a
d
i
i
r
r
e
.
c
c
t
.
o
m
m
/
j
e
d
o
u
c
n
o
/
c
a
n
r
a
t
r
i
t
i
c
c
l
e
e
-
p
-
d
p
d
2
f
9
/
2
2
9
2
/
3
2
5
/
1
2
9
3
5
5
2
/
3
1
2
7
6
8
o
5
c
7
n
9
_
5
a
/
_
j
0
o
1
c
0
n
4
1
_
a
p
_
d
0
1
b
0
y
4
g
1
u
.
e
p
s
t
d
o
f
n
b
0
y
8
S
M
e
I
p
T
e
m
L
i
b
b
e
r
r
a
2
r
0
i
2
3
e
s
/
j
t
/
f
.
u
s
e
r
o
n
1
7
M
a
y
2
0
2
1
244
Journal of Cognitive Neuroscience
Volume 29, Number 2