Fixation-related Brain Potentials during Semantic
Integration of Object–Scene Information
Moreno I. Coco1,2, Antje Nuthmann3, and Olaf Dimigen4
Abstrakt
■ In vision science, a particularly controversial topic is whether
and how quickly the semantic information about objects is avail-
able outside foveal vision. Hier, we aimed at contributing to this
debate by coregistering eye movements and EEG while parti-
cipants viewed photographs of indoor scenes that contained a
semantically consistent or inconsistent target object. Linear de-
convolution modeling was used to analyze the ERPs evoked by
scene onset as well as the fixation-related potentials (FRPs) elic-
ited by the fixation on the target object (T) and by the preceding
fixation (t − 1). Object–scene consistency did not influence the
probability of immediate target fixation or the ERP evoked by
scene onset, which suggests that object–scene semantics was
not accessed immediately. Jedoch, during the subsequent
scene exploration, inconsistent objects were prioritized over con-
sistent objects in extrafoveal vision (d.h., looked at earlier) and were
more effortful to process in foveal vision (d.h., looked at longer).
In FRPs, we demonstrate a fixation-related N300/N400 effect,
whereby inconsistent objects elicit a larger frontocentral nega-
tivity than consistent objects. In line with the behavioral findings,
this effect was already seen in FRPs aligned to the pretarget fixa-
tion t − 1 and persisted throughout fixation t, indicating that the
extraction of object semantics can already begin in extrafoveal
vision. Taken together, the results emphasize the usefulness
of combined EEG/eye movement recordings for understanding
the mechanisms of object–scene integration during natural
viewing. ■
EINFÜHRUNG
In our daily activities—for example, when we search for
something in a room—our attention is mostly oriented to
Objekte. The time course of object recognition and the
role of overt attention in this process are therefore topics
of considerable interest in the visual sciences. In the con-
text of real-world scene perception, the question of what
constitutes an object is a more complex question than in-
tuition would suggest (z.B., Wolfe, Alvarez, Rosenholtz,
Kuzmova, & Sherman, 2011). An object is likely a hierar-
chical construct (z.B., Feldman, 2003), with both low-level
Merkmale (z.B., visual saliency) and high-level properties
(z.B., semantics) contributing to its identity. Entsprechend,
when a natural scene is inspected with eye movements,
the observer’s attentional selection is thought to be based
either on objects (z.B., Nuthmann & Henderson, 2010),
image features (saliency; Itti, Koch, & Niebur, 1998), or some
combination of the two (z.B., Stoll, Thrun, Nuthmann, &
Einhäuser, 2015).
An early and uncontroversial finding is that the recog-
nition of objects is mediated by their semantic consis-
tency. Zum Beispiel, an object that the observer would
not expect to occur in a particular scene (z.B., a tooth-
brush in a kitchen) is recognized less accurately (z.B.,
1The University of East London, 2CICPSI, Faculdade de Psicologia,
Universidade de Lisboa, 3Christian-Albrechts-Universität zu Kiel,
4Humboldt-Universität zu Berlin
Fenske, Aminoff, Gronau, & Bar, 2006; Davenport &
Töpfer, 2004; Biederman, 1972) and looked at for longer
than an expected object (z.B., Cornelissen & Võ, 2017;
Henderson, Weeks, & Hollingworth, 1999; De Graef,
Christiaens, & d’Ydewalle, 1990).
What is more controversial, Jedoch, is the exact time
course along which the meaning of an object is processed
and how this semantic processing then influences the
overt allocation of visual attention (see Wu, Wick, &
Pomplun, 2014, für eine Rezension). Two interrelated questions
are at the core of this debate: (1) How much time is needed
to access the meaning of objects after a scene is displayed,
Und (2) Can object semantics be extracted before the ob-
ject is overtly attended, das ist, while the object is still out-
side high-acuity foveal vision (> 1° eccentricity) or even in
the periphery (> 5° eccentricity)?
Evidence that the meaning of not-yet-fixated objects can
capture overt attention comes from experiments that have
used sparse displays of several standalone objects (z.B.,
Cimminella, Della Sala, & Coco, in press; Nuthmann, von
Groot, Huettig, & Olivers, 2019; Belke, Humphreys,
Watson, Meyer, & Telling, 2008; Moores, Laiti, & Chelazzi,
2003). Zum Beispiel, across three different experiments,
Nuthmann et al. found that the very first saccade in the dis-
play was directed more frequently to objects that were
semantically related to a target object rather than to un-
related objects.
Whether such findings generalize to objects embed-
ded in real-world scenes is currently an open research
© 2019 Massachusetts Institute of Technology. Published under a
Creative Commons Attribution 4.0 International (CC BY 4.0) Lizenz.
Zeitschrift für kognitive Neurowissenschaften 32:4, S. 571–589
https://doi.org/10.1162/jocn_a_01504
D
Ö
w
N
l
Ö
A
D
e
D
l
l
/
/
/
/
J
T
T
F
/
ich
T
.
:
/
/
F
R
Ö
M
D
Ö
H
w
T
N
T
P
Ö
:
A
/
D
/
e
D
M
ich
F
R
T
Ö
P
M
R
C
H
.
P
S
ich
l
D
v
ich
R
e
e
R
C
T
C
.
M
H
A
ich
e
R
D
.
u
C
Ö
Ö
M
C
N
/
J
A
Ö
R
T
C
ich
C
N
e
/
–
A
P
R
D
T
ich
3
2
C
l
4
e
5
–
7
P
1
D
F
2
0
/
1
3
3
2
2
/
4
4
7
/
5
Ö
7
C
1
N
/
_
A
1
_
8
0
6
1
1
5
2
0
7
4
6
P
/
D
J
Ö
B
C
j
N
G
_
u
A
e
_
S
0
T
1
Ö
5
N
0
0
4
8
.
P
S
D
e
F
P
e
B
M
j
B
e
G
R
u
2
e
0
S
2
T
3
/
J
/
F
T
.
Ö
N
0
5
M
A
j
2
0
2
1
question. The size of the visual span—that is, the area of
the visual field from which observers can take in useful
Information (see Rayner, 2014, für eine Rezension)—is large
in scene viewing. For object-in-scene search, it corre-
sponded to approximately 8° in each direction from
fixation (Nuthmann, 2013). This opens up the possibility
that both low- and high-level object properties can be
processed outside the fovea. This is clearly the case for
low-level visual features: Objects that are highly salient
(d.h., visually distinct) are preferentially selected for fixation
(z.B., Stoll et al., 2015). If semantic processing also takes
place in extrafoveal vision, then objects that are inconsis-
tent with the scene context (which are also thought to be
more informative; Antes, 1974) should be fixated earlier
in time than consistent ones (Loftus & Mackworth, 1978;
Mackworth & Morandi, 1967).
Jedoch, results from eye-movement studies on this is-
sue have been mixed. A number of studies have indeed re-
ported evidence for an inconsistent object advantage (z.B.,
Borges, Fernandes, & Coco, 2019; LaPointe & Milliken,
2016; Bonitz & Gordon, 2008; Underwood, Templeman,
Lamming, & Foulsham, 2008; Loftus & Mackworth, 1978).
Among these studies, only Loftus and Mackworth (1978)
have reported evidence for immediate extrafoveal atten-
tional capture (d.h., within the first fixation) by object–scene
semantics. In this study, which used relatively sparse line
drawings of scenes, the mean amplitude of the saccade into
the critical object was more than 7°, suggesting that viewers
could process semantic information based on peripheral
information obtained in a single fixation. Im Gegensatz,
other studies have failed to find any advantage for
inconsistent objects in attracting overt attention (z.B.,
Võ & Henderson, 2009, 2011; Henderson et al., 1999;
De Graef et al., 1990). In these experiments, only mea-
sures of foveal processing—such as gaze duration—
were influenced by object–scene consistency, with lon-
ger fixation times on inconsistent than on consistent
Objekte.
Interessant, a similar controversy exists in the lit-
erature on eye guidance in sentence reading. Obwohl
some degree of parafoveal processing during reading is
uncontroversial, it is less clear whether semantic infor-
mation is acquired from the parafovea (Andrews &
Veldre, 2019, für eine Rezension). Most evidence from studies
involving readers of English has been negative (z.B.,
Rayner, Balota, & Pollatsek, 1986), whereas results from
reading German (z.B., Hohenstein & Kliegl, 2014) Und
Chinese (z.B., Yan, Richter, Shu, & Kliegl, 2009) vorschlagen
that parafoveal processing can advance up to the level of
semantic processing.
The processing of object–scene inconsistencies and its
time course have also been investigated in electrophysi-
ological studies (z.B., Mudrik, Lamy, & Deouell, 2010;
Ganis & Kutas, 2003). In ERPs, it is commonly found that
scene-inconsistent objects elicit a larger negative brain re-
sponse compared with consistent ones. This long-lasting
negative shift typically starts as early as 200–250 msec
after stimulus onset (z.B., Draschkow, Heikel, Võ, Fiebach,
& Sassenhagen, 2018; Mudrik, Shalgi, Lamy, & Deouell,
2014) and has its maximum at frontocentral scalp sites,
in contrast to the centroparietal N400 effect for words
(z.B., Kutas & Federmeier, 2011). The effect was found
for objects that appeared at a cued location after the
scene background was already shown (Ganis & Kutas,
2003), for objects that were photoshopped into the
scene (Coco, Araujo, & Petersson, 2017; Mudrik et al.,
2010, 2014), and for objects that were part of realistic
photographs ( Võ & Wolfe, 2013). These ERP effects of
object–scene consistency have typically been subdivided
into two distinct components: N300 and N400. The earlier
part of the negative response, usually referred to as N300,
has been taken to reflect the context-dependent difficulty
of object identification, whereas the later N400 has been
linked to semantic integration processes after the object is
identified (z.B., Dyck & Brodeur, 2015). The present study
was not designed to differentiate between these two sub-
components, especially considering that their scalp distri-
bution is strongly overlapping or even topographically
indistinguishable (Draschkow et al., 2018). Daher, for rea-
sons of simplicity, we will in most cases simply refer to
all frontocentral negativities as “N400.”
One limiting factor of existing ERP studies is that the
data were gathered using steady-fixation paradigms in
which the free exploration of the scene through eye
movements was not permitted. Stattdessen, the critical object
was typically large and/or located relatively close to the
center of the screen, and ERPs were time-locked to the
onset of the image (z.B., Mudrik et al., 2010). Weil
of these limitations, it remains unclear whether foveation
of the object is a necessary condition for processing
object–scene consistencies or whether such processing
can at least begin in extrafoveal vision.
In the current study, we used fixation-related po-
tentials (FRPs), das ist, EEG waveforms aligned to fixation
onset, to shed new light on the controversial findings of
the role of foveal versus extrafoveal vision in extracting
object semantics, while providing insights into the
patterns of brain activity that underlie them (for reviews
about FRPs, see Nikolaev, Meghanathan, & van Leeuwen,
2016; Dimigen, Sommer, Hohlfeld, Jacobs, & Kliegl, 2011).
FRPs have been used to investigate the brain-electric
correlates of natural reading, as opposed to serial word
presentation, helping researchers to provide finer details
about the online processing of linguistic features (wie zum Beispiel
word predictability; Kliegl, Dambacher, Dimigen, Jacobs,
& Sommer, 2012; Kretzschmar, Bornkessel-Schlesewsky,
& Schlesewsky, 2009) or the dynamics of the perceptual
span during reading (z.B., parafovea-on-fovea effects;
Niefind & Dimigen, 2016). More recently, the coregistra-
tion method has also been applied to investigate active
visual search (z.B., Ušćumlić & Blankertz, 2016; Devillez,
Guyader, & Guérin-Dugué, 2015; Kaunitz et al., 2014;
Brouwer, Reuderink, Vincent, van Gerven, & van Erp, 2013;
Kamienkowski, Ison, Quiroga, & Sigman, 2012), Objekt
572
Zeitschrift für kognitive Neurowissenschaften
Volumen 32, Nummer 4
D
Ö
w
N
l
Ö
A
D
e
D
l
l
/
/
/
/
J
T
T
F
/
ich
T
.
:
/
/
F
R
Ö
M
D
Ö
H
w
T
N
T
P
Ö
:
A
/
D
/
e
D
M
ich
F
R
T
Ö
P
M
R
C
H
.
P
S
ich
l
D
v
ich
R
e
e
R
C
T
C
.
M
H
A
ich
e
R
D
.
u
C
Ö
Ö
M
C
N
/
J
A
Ö
R
T
C
ich
C
N
e
/
–
A
P
R
D
T
ich
3
2
C
l
4
e
5
–
7
P
1
D
F
2
0
/
1
3
3
2
2
/
4
4
7
/
5
Ö
7
C
1
N
/
_
A
1
_
8
0
6
1
1
5
2
0
7
4
6
P
/
D
J
Ö
B
C
j
N
G
_
u
A
e
_
S
0
T
1
Ö
5
N
0
0
4
8
.
P
S
D
e
F
P
e
B
M
j
B
e
G
R
u
2
e
0
S
2
T
3
/
J
.
F
T
/
Ö
N
0
5
M
A
j
2
0
2
1
identification (Rämä & Baccino, 2010), and affective process-
ing in natural scene viewing (Simola, Le Fevre, Torniainen, &
Baccino, 2015).
In this study, we simultaneously recorded eye move-
ments and FRPs during the viewing of real-world scenes
to distinguish between three alternative hypotheses on
object–scene integration that can be derived from the lit-
erature: (A) One glance of the scene is sufficient to extract
object semantics from extrafoveal vision (z.B., Loftus &
Mackworth, 1978), (B) extrafoveal processing of object–
scene semantics is possible but takes some time to unfold
(z.B., Bonitz & Gordon, 2008; Underwood et al., 2008),
Und (C) the processing of object semantics requires foveal
vision, das ist, a direct fixation of the critical object (z.B., Võ &
Henderson, 2009; Henderson et al., 1999; De Graef et al.,
1990). We note that these possibilities are not mutually
exclusive, an issue we elaborate on in the Discussion section.
For the behavioral data, these hypotheses translate as
follows: under Hypothesis A, the probability of immedi-
ate target fixation should reveal that already the first
saccade on the scene goes more often toward inconsis-
tent than consistent objects. Under Hypothesis B, Dort
should be no effect on the first eye movement, aber die
latency to first fixation on the critical object should be shorter
for inconsistent than consistent objects. Under Hypothesis
C, only fixation times on the critical object itself should differ
as a function of object–scene consistency, with longer gaze
durations on inconsistent objects.
For the electrophysiological data analysis, we used a novel
regression-based analysis approach (linear deconvolution
modeling; Cornelissen, Sassenhagen, & Võ, 2019; Dimigen
& Ehinger, 2019; Ehinger & Dimigen, 2019; Kristensen, Rivet,
& Guérin-Dugué, 2017; Schmied & Kutas, 2015B; Dandekar,
Privitera, Carney, & Klein, 2012), which allowed us to con-
trol for the confounding influences of overlapping poten-
tials and oculomotor covariates on the neural responses
during natural viewing. In the EEG, Hypothesis A can be
tested by computing the ERP time-locked to the onset of
the scene on the display, following the traditional ap-
proach. Given that the critical objects in our study were
not placed directly in the center of the screen from which
observers started their exploration of the scene, any effect
of object–scene congruency in this ERP would suggest that
object semantics is rapidly processed in extrafoveal vision,
even before the first eye movement is generated, in line
with Loftus and Mackworth (1978). Under Hypothesis B,
we would not expect to see an effect in the scene-onset
ERP. Stattdessen, we should find a negative brain potential
(N400) for inconsistent as compared with consistent ob-
jects in the FRP aligned to the fixation that precedes the
one that first lands on the critical object. Endlich, Wenn
Hypothesis C is correct, an N400 for inconsistent objects
should only arise once the critical object is foveated, Das
Ist, in the FRP aligned to the target fixation (fixation t). In con-
trast, no consistency effects should appear in the scene-
onset ERP or in the FRP aligned to the pretarget fixation
(fixation t − 1). To preview the results, both the eye move-
ment and the EEG data lend support for Hypothesis B.
METHODEN
Design and Task Overview
We designed a short-term visual working memory change
detection task, illustrated in Figures 1 Und 2. Während der
study phase, participants were exposed to photographs
D
Ö
w
N
l
Ö
A
D
e
D
l
l
/
/
/
/
J
T
T
F
/
ich
T
.
:
/
/
F
R
Ö
M
D
Ö
H
w
T
N
T
P
Ö
:
A
/
D
/
e
D
M
ich
F
R
T
Ö
P
M
R
C
H
.
P
S
ich
l
D
v
ich
R
e
e
R
C
T
C
.
M
H
A
ich
e
R
D
.
u
C
Ö
Ö
M
C
N
/
J
A
Ö
R
T
C
ich
C
N
e
/
–
A
P
R
D
T
ich
3
2
C
l
4
e
5
–
7
P
1
D
F
2
0
/
1
3
3
2
2
/
4
4
7
/
5
Ö
7
C
1
N
/
_
A
1
_
8
0
6
1
1
5
2
0
7
4
6
P
/
D
J
Ö
B
C
j
N
G
_
u
A
e
_
S
0
T
1
Ö
5
N
0
0
4
8
.
P
S
D
e
F
P
e
B
M
j
B
e
G
R
u
2
e
0
S
2
T
3
/
J
T
F
.
/
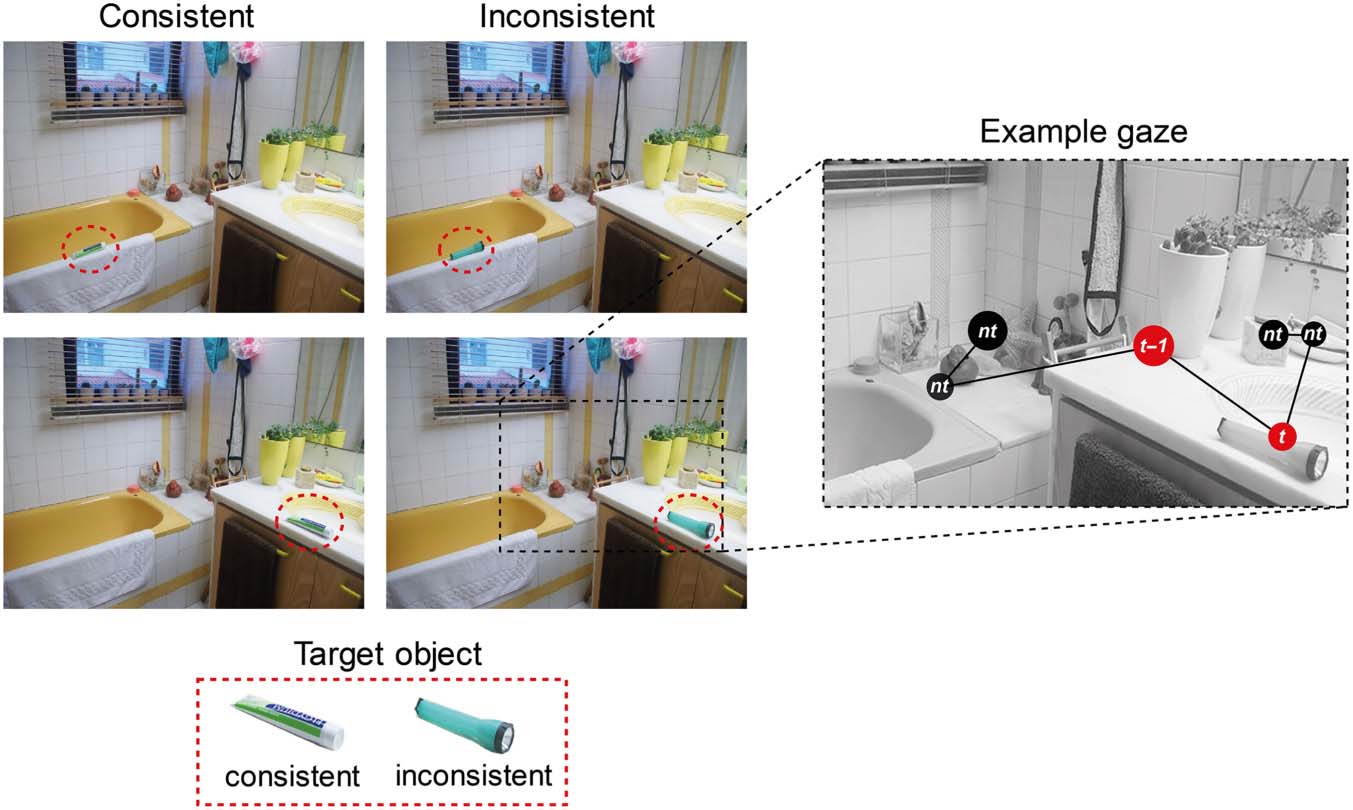
Figur 1. Example stimuli and
conditions in the study.
Participants viewed photographs
of indoor scenes that contained a
target object (highlighted with a
red circle) that was either
semantically consistent (Hier,
toothpaste) or semantically
inconsistent (Hier, flashlight)
with the context of the scene.
The target object could be
placed at different locations
within the scene, on either the
left or right side. The example
gaze path plotted on the right
illustrates the three types of
fixations analyzed in the study:
(A) t – 1, the fixation preceding
the first fixation to the target
Objekt; (B) T, the first fixation to
the target; Und (C) nt, all other
(nontarget) fixations. Fixation
duration is proportional
to the diameter of the circle,
which is red for the critical
fixations and black for the
nontarget fixations.
Ö
N
0
5
M
A
j
2
0
2
1
Coco, Nuthmann, and Dimigen
573
additional two participants were recorded but removed
from the analysis because of excessive scalp muscle
(EMG) activity or skin potentials in the raw EEG. Ethics
approval was obtained from the Psychology Research
Ethics Committee of the University of Edinburgh.
Apparatus and Recording
Scenes were presented on a 19-in. CRT monitor (Iiyama
Vision Master Pro 454) at a vertical refresh rate of 75 Hz.
At the viewing distance of 60 cm, each scene subtended
35.8° × 26.9° (width × height). Eye movements were re-
corded monocularly from the dominant eye using an SR
Research EyeLink 1000 desktop-mounted system at a
sampling rate of 1000 Hz. Eye dominance for each par-
ticipant was determined with a parallax test. A chin-and-
forehead rest was used to stabilize the participant’s head.
Nine-point calibrations were run at the beginning of each
session and whenever the participant’s fixation deviated
by > 0.5° horizontally or > 1° vertically from a drift cor-
rection point presented at trial onset.
The EEG was recorded from 64 active electrodes at a
sampling rate of 512 Hz using BioSemi ActiveTwo am-
plifiers. Four electrodes, located near the left and right
canthus and above and below the right eye, recorded the
EOG. All channels were referenced against the BioSemi
common mode sense (active electrode) and grounded
to a passive electrode. The BioSemi hardware is DC
coupled and applies digital low-pass filtering through
the A/D-converter’s decimation filter, which has a fifth-order
sinc response with a −3 dB point at one fifth of the sample
rate (corresponding approximately to a 100-Hz low-
pass filter).
Offline, the EEG was rereferenced to the average of all
scalp electrodes and filtered using EEGLAB’s (Delorme &
Makeig, 2004) Hamming-windowed sinc finite impulse
response filter (pop_eegfiltnew.m) with default settings.
The lower edge of the filter’s passband was set to 0.2 Hz
(with −6 dB attenuation at 0.1 Hz); and the upper edge,
Zu 30 Hz (with −6 dB attenuation at 33.75 Hz). Eye track-
ing and EEG data were synchronized using shared triggers
sent via the parallel port of the stimulus presentation PC to
the two recording computers. Synchronization was per-
formed offline using the EYE-EEG extension (v0.8) für
EEGLAB (Dimigen et al., 2011). All data sets were aligned
with a mean synchronization error ≤ 2 msec as computed
based on trigger alignment after synchronization.
Materials and Rating
Stimuli consisted of 192 color photographs of indoor
scenes (z.B., bedrooms, bathrooms, offices). Real target
objects were placed in the physical scene, before each
picture was taken with a tripod under controlled lighting
conditions and with a fixed aperture (d.h., there was no
photo-editing). One scene is shown in Figure 1; minia-
ture versions of all stimuli used in this study are found
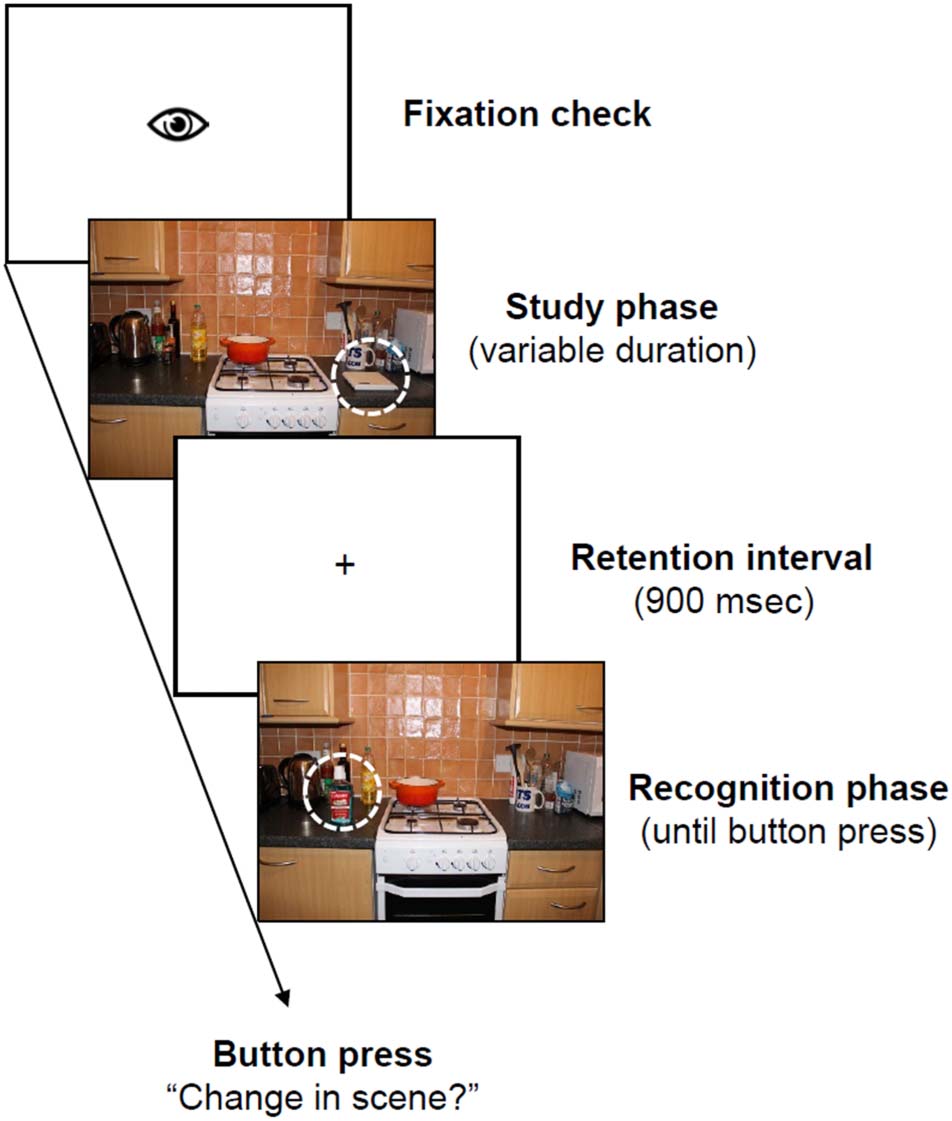
Figur 2. Trial scheme. After a drift correction, the study scene
appeared. The display duration of the scene was controlled by a
gaze-contingent mechanism, and it disappeared, on average, 2000 ms
after the target object was fixated. In the following retention interval,
only a fixation cross was presented. During the recognition phase,
the scene was presented again until participants pressed a button to
indicate whether or not a change had occurred within the scene. Alle
analyses in the present article focus on eye-movement and EEG data
collected during the study phase.
of indoor scenes (z.B., a bathroom), each of which con-
tained a target object that was either semantically consis-
tent (z.B., toothpaste) or inconsistent (z.B., a flashlight)
with the scene context. In the following recognition
Phase, after a short retention interval of 900 ms, Die
same scene was shown again, but in half of the trials,
either the identity, the location, or both the identity and
location of the target object had changed relative to the
study phase.
The participants’ task was to indicate with a keyboard
press whether or not a change had happened to the
scene (see also LaPointe & Milliken, 2016). All eye-
movement and EEG analyses in the present article focus
on the semantic consistency manipulation of the target
object during the study phase.
Teilnehmer
Twenty-four participants (nine men) zwischen dem Alter von
18 Und 33 Jahre (M = 25.0 Jahre) took part in the exper-
iment after providing written informed consent. Sie
were compensated with £7 per hour. All participants
had normal or corrected-to-normal vision. Data from an
574
Zeitschrift für kognitive Neurowissenschaften
Volumen 32, Nummer 4
D
Ö
w
N
l
Ö
A
D
e
D
l
l
/
/
/
/
J
T
T
F
/
ich
T
.
:
/
/
F
R
Ö
M
D
Ö
H
w
T
N
T
P
Ö
:
A
/
D
/
e
D
M
ich
F
R
T
Ö
P
M
R
C
H
.
P
S
ich
l
D
v
ich
R
e
e
R
C
T
C
.
M
H
A
ich
e
R
D
.
u
C
Ö
Ö
M
C
N
/
J
A
Ö
R
T
C
ich
C
N
e
/
–
A
P
R
D
T
ich
3
2
C
l
4
e
5
–
7
P
1
D
F
2
0
/
1
3
3
2
2
/
4
4
7
/
5
Ö
7
C
1
N
/
_
A
1
_
8
0
6
1
1
5
2
0
7
4
6
P
/
D
J
Ö
B
C
j
N
G
_
u
A
e
_
S
0
T
1
Ö
5
N
0
0
4
8
.
P
S
D
e
F
P
e
B
M
j
B
e
G
R
u
2
e
0
S
2
T
3
/
J
F
T
/
.
Ö
N
0
5
M
A
j
2
0
2
1
online at https://osf.io/sjprh/. Of the 192 scenes, 96 war
conceived as change items and 96 were conceived as no-
change items. Each one of the 96 change scenes was created
in four versions. Insbesondere, the scene (z.B., a bathroom)
was photographed with two alternative target objects in
Es, one that was consistent with the scene context (z.B., A
toothbrush) and one that was not (z.B., a flashlight).
Darüber hinaus, each of these two objects was placed at two
alternative locations (left or right side) within the scene
(z.B., either on the sink or on the bathtub). Entsprechend,
three types of change were implemented during the rec-
ognition phase (Congruency, Location, and Both; sehen
Procedure section below).
Each of the 96 no-change scenes was also a real pho-
tograph with either a consistent or an inconsistent object
in it, which was again located in either the left or right
half of the scene. Across the 96 no-change scenes, Die
factors consistency (consistent vs. inconsistent objects)
and location ( left and right) were also balanced.
Jedoch, each no-change scene was unique; das ist, Wir
did not create four different versions of each no-change
scene. The data of the 96 no-change scenes, die Waren
originally conceived to be filler trials, were included to
improve the signal-to-noise ratio of the EEG analyses,
as these scenes also had a balanced distribution of incon-
sistent and consistent objects.
Wie oben erklärt, scenes contained a critical object
that was either consistent or inconsistent with the scene
Kontext. Object consistency was assessed in a pretest rat-
ing study by eight naive participants who were not in-
volved in any other aspect of the study. Each participant
rated all of the no-change scenes as well as one of the four
versions of each change-scene (counterbalanced across
raters). Together with the scene, raters saw a box with
a cropped image of the critical object. They were asked
(A) to write down the name for the displayed object and
(B) to respond to the question “How likely is it that this
object would be found in this room?” using a 6-point
Likert scale (1–6). For the object naming, a mean naming
agreement of 96.35% was obtained. Außerdem, konsis-
tent objects were judged as significantly more likely (M =
5.78, SD = 0.57) to appear in the scene than inconsistent
Objekte (M = 1.88, SD = 1.11), wie von einem bestätigt
independent-samples Kruskal–Wallis H test, χ2(1) =
616.09, P < .001.
In addition, we ensured that there was no difference be-
tween consistent and inconsistent objects on three impor-
tant low-level variables: object size (pixels square), distance
from the center of the scene (degrees of visual angle),
and mean visual saliency of the object as computed using
the Adaptive Whitening Saliency model (Garcia-Diaz,
Fdez-Vidal, Pardo, & Dosil, 2012). Table 1 provides addi-
tional information about the target object. Independent t
tests showed no significant difference between inconsis-
tent and consistent objects in size, t(476) = −1.27, p =
.2; visual saliency, t(476) = 0.82, p = .41; and distance
from the center, t(476) = −1.75, p = .08.
The position of each target object was marked with an
invisible rectangular bounding box, which was used to im-
plement the gaze contingency mechanism (described in
the Procedure section below) and to determine whether
a fixation was inside the target object. The average width
of the bounding box was 6.1° ± 2.0° for consistent objects
and 6.1° ± 2.1° for inconsistent objects (see Table 1); the
average height was 5.1° ± 1.8° and/or 5.4° ± 2.2°, respec-
tively. The average distance of the object centroid from
the center of the scene was 12.1° (± 2.8°) for consistent
and 11.7° (± 3.0°) for inconsistent objects.
Procedure
A schematic representation of the task is shown in
Figure 2. Each trial started with a drift correction of the
eye tracker. Afterward, the study scene was presented
(e.g., a bathroom). The display duration of the study
scene was controlled by a gaze-contingent mechanism
that ensured that participants fixated the target object
(e.g., toothbrush or flashlight) at least once during the
trial. Specifically, the study scene disappeared, on aver-
age, 2000 msec (with a random jitter of ± 200 msec,
drawn from a uniform distribution) after the participant’s
eyes left the invisible bounding box of the target object
(and provided that the target had been fixated for at least
150 msec). The jittered delay of about 2000 msec was im-
plemented to prevent participants from learning to asso-
ciate the last fixated object during the study phase with
the changed object during the recognition phase. If the
participant did not fixate the target object within 10 sec,
the study scene disappeared from the screen and the re-
tention interval was triggered, which lasted for 900 msec.
In the following recognition phase (data not analyzed
here), the scene was presented again, either with (50% of
trials) or without (50% of trials) a change to an object in
the scene. Three types of object changes occurred with
equal probability: Location, Consistency, or Both. In the
(a) Location condition, the target object changed its po-
sition and moved either from left to right or from right to
left to another plausible location within the scene (e.g., a
toothbrush was placed elsewhere within the bathroom
scene). In the (b) Consistency condition, the object re-
mained in the same location but was replaced with an-
other object of opposite semantic consistency (e.g., the
toothbrush was replaced by a flashlight). Finally, in the
(c) Both condition, the object was both replaced and
moved within the scene (e.g., a toothbrush was replaced
by a flashlight at a different location).
During the recognition phase, participants had to indi-
cate whether they noticed any kind of change within the
scene by pressing the arrow keys on the keyboard.
Afterward, the scene disappeared, and the next trial be-
gan. If participants did not respond within 10 sec, a miss-
ing response was recorded.
The type of change between trials was fully counterba-
lanced using a Latin Square rotation. Specifically, the 96
Coco, Nuthmann, and Dimigen
575
D
o
w
n
l
o
a
d
e
d
l
l
/
/
/
/
j
f
/
t
t
i
t
.
:
/
/
f
r
o
m
D
o
h
w
t
n
t
p
o
:
a
/
d
/
e
d
m
i
f
r
t
o
p
m
r
c
h
.
p
s
i
l
d
v
i
r
e
e
r
c
t
c
.
m
h
a
i
e
r
d
.
u
c
o
o
m
c
n
/
j
a
o
r
t
c
i
c
n
e
/
-
a
p
r
d
t
i
3
2
c
l
4
e
5
-
7
p
1
d
f
2
0
/
1
3
3
2
2
/
4
4
7
/
5
o
7
c
1
n
/
_
a
1
_
8
0
6
1
1
5
2
0
7
4
6
p
/
d
j
o
b
c
y
n
g
_
u
a
e
_
s
0
t
1
o
5
n
0
0
4
8
.
p
S
d
e
f
p
e
b
m
y
b
e
g
r
u
2
e
0
s
2
t
3
/
j
t
/
.
f
o
n
0
5
M
a
y
2
0
2
1
Table 1. Eye Movement Behavior in the Task and Properties of the Target Object
Eye movement behavior
Ordinal fixation number of first target fixation
6.7 ± 6.0
5.2 ± 5.3
Consistent
Inconsistent
Mean ± SD
Mean ± SD
Fixation duration (t − 2), in msec
Fixation duration (t − 1), in msec
Fixation duration (t), in msec
Gaze duration on target, in msec
Number of refixations on target
220.7 ± 105
212.9 ± 95
207.6 ± 96
197 ± 91
261.6 ± 146
263.3 ± 136
408.5 ± 367.1
519.1 ± 373.6
1.7 ± 2
2.2 ± 2.1
Duration of refixations on target, in msec
238.9 ± 121.8
250.2 ± 135.7
Fixation duration (t + 1), in msec
Incoming saccade amplitude to t − 1 (°)
Incoming saccade amplitude to t (°)
Incoming saccade amplitude to t + 1 (°)
Distance of fixation t − 1 from the closest
edge of target (°)
Number of fixations after first encountering
target object until end of study phase
Duration of fixations after first encountering
target object (until end of study phase)
245.3 ± 148
243.7 ± 146
6.1 ± 5.2
8.5 ± 5.2
9.5 ± 5.9
6.8 ± 5.8
6 ± 4.8
8.3 ± 4.8
10.2 ± 5.8
6.3 ± 5.3
7.3 ± 2.1
7.3 ± 1.7
254.6 ± 120.4
251.7 ± 118.8
Target object properties
Distance of target object center from screen center (°)
12.1 ± 2.8
11.7 ± 3
Mean visual saliency (AWS model)
0.36 ± 0.16
0.37 ± 0.16
Width (°)
Height (°)
6.1 ± 2
5.1 ± 1.8
6.1 ± 2.1
5.4 ± 2.2
Area (degrees of visual angle squared)
16.1 ± 8.7
17.3 ± 11.4
Target object size and distance to target are based on the bounding box around the object. The fixation t + 1 is the first fixation after leaving the
bounding box of the target object.
change trials were distributed across 12 different lists, im-
plementing the different types of change. This implies
that each participant was exposed to an equal number
of consistent and inconsistent change trials. The 96 no-
change trials also were composed of an equal number
of consistent and inconsistent scenes and were the same
for each participant. During the experiment, all 192 trials
were presented in a randomized order. They were pre-
ceded by four practice trials at the start of the session.
Written instructions were given to explain the task, which
took 20–40 min to complete. The experiment was im-
plemented using the SR Research Experiment Builder
software.
Data Preprocessing
Eye-movement Events and Data Exclusion
Fixations and saccade events were extracted from the raw
gaze data using the SR Research Data Viewer software,
which performs saccade detection based on velocity and
acceleration thresholds of 30° sec−1 and 9500° sec−2,
respectively. To provide directly comparable results for
eye-movement behavior and FRP analyses, we discarded
all trials on which we did not have clean data from both re-
cordings. Specifically, from 4608 trials (24 participants × 192
trials), we excluded 10 trials (0.2%) because of machine
error (i.e., no data were recorded for those trials), 689 tri-
als (15.0%) because the participant responded incorrectly
after the recognition phase, and 494 trials (10.7%) be-
cause the target object was not fixated during the study
phase. Finally, we removed an additional 97 trials (2.1%)
for which the target fixation overlapped with intervals of
the EEG that contained nonocular artifacts (see below).
The final data set for the behavioral and FRP analyses
therefore was composed of 3318 unique trials: 1567 for
the consistent condition and 1751 for the inconsistent
condition. Per participant, this corresponded to an aver-
age of 65.3 trials (± 6.9, range = 48–78) for consistent
and 73.0 trials (± 6.9, range = 59–82) for inconsistent
576
Journal of Cognitive Neuroscience
Volume 32, Number 4
D
o
w
n
l
o
a
d
e
d
l
l
/
/
/
/
j
f
/
t
t
i
t
.
:
/
/
f
r
o
m
D
o
h
w
t
n
t
p
o
:
a
/
d
/
e
d
m
i
f
r
t
o
p
m
r
c
h
.
p
s
i
l
d
v
i
r
e
e
r
c
t
c
.
m
h
a
i
e
r
d
.
u
c
o
o
m
c
n
/
j
a
o
r
t
c
i
c
n
e
/
-
a
p
r
d
t
i
3
2
c
l
4
e
5
-
7
p
1
d
f
2
0
/
1
3
3
2
2
/
4
4
7
/
5
o
7
c
1
n
/
_
a
1
_
8
0
6
1
1
5
2
0
7
4
6
p
/
d
j
o
b
c
y
n
g
_
u
a
e
_
s
0
t
1
o
5
n
0
0
4
8
.
p
S
d
e
f
p
e
b
m
y
b
e
g
r
u
2
e
0
s
2
t
3
/
j
f
t
/
.
o
n
0
5
M
a
y
2
0
2
1
items. Because of the fixation check, participants were al-
ways fixating at the screen center when the scene appeared
on the display. This ongoing central fixation was removed
from all analyses.
EEG Ocular Artifact Correction
EEG recordings during free viewing are contaminated by
three types of ocular artifacts (Plöchl, Ossandón, & König,
2012) that need to be removed to get at the genuine brain
activity. Here, we applied an optimized variant (Dimigen,
2020) of independent component analysis (ICA; Jung
et al., 1998), which uses the information provided by the
eye tracker to objectively identify ocular ICA components
(Plöchl et al., 2012).
In a first step, we created optimized ICA training data
by high-pass filtering a copy of the EEG at 2 Hz (Dimigen,
2020; Winkler, Debener, Müller, & Tangermann, 2015)
and segmenting it into epochs lasting from scene onset
until 3 sec thereafter. These high-pass-filtered training
data were entered into an extended Infomax ICA using
EEGLAB, and the resulting unmixing weights were then
transferred to the original (i.e., less strictly filtered) re-
cording (Debener, Thorne, Schneider, & Viola, 2010).
From this original EEG data set, we then removed all inde-
pendent components whose time course varied more
strongly during saccade intervals (defined as lasting from
−20 msec before saccade onset until 20 msec after
saccade offset) than during fixations, with the threshold
for the variance ratio (saccade/fixation; see Plöchl et al.,
2012) set to 1.3. Finally, the artifact-corrected continuous
EEG was back-projected to the sensor space. For a valida-
tion of the ICA procedure, please refer to Supplementary
Figure S1.
In a next step, intervals with residual nonocular ar-
tifacts (e.g., EMG bursts) were detected by shifting a
2000-msec moving window in steps of 100 msec across
the continuous recording. Whenever the voltages within
the window exceeded a peak-to-peak threshold of 100 μV
in at least one of the channels, all data within the window
were marked as “bad” and subsequently excluded from
analysis. Within the linear deconvolution framework
(see below), this can easily be done by setting all predic-
tors to zero during these bad EEG intervals (Smith &
Kutas, 2015b), meaning that the data in these intervals
will not affect the computation.
Analysis
Eye-movement Data
Dependent measures. Behavioral analyses focused on
four eye-movement measures commonly reported in
the semantic consistency literature: (a) the cumulative
probability of having fixated the target object as a func-
tion of the ordinal fixation number, (b) the probability
of immediate object fixation, (c) the latency to first
fixation on the target object, and (d) the gaze duration
on the target object (cf. Võ & Henderson, 2009).
Linear mixed-effects modeling. Eye-movement data
were analyzed using linear mixed-effects models
(LMMs) and generalized LMMs (GLMM) as implemented
in the lme4 package in R (Bates, Mächler, Bolker, &
Walker, 2015). The only exception was the cumulative
probability of first fixations on the target for which a gen-
eralized linear model (GLM) was used. One advantage of
(G)LMM modeling is that it allows one to simultaneously
model the intrinsic variability of both participants and
scenes (e.g., Nuthmann & Einhäuser, 2015).
In all analyses, the main predictor was the consistency
of the critical object (contrast coding: consistent = −0.5,
inconsistent = 0.5) in the study scene. In the (G)LMMs,
Participant (24) and Scene (192) were included as ran-
dom intercepts.1 The cumulative probability of object fix-
ation was analyzed using a GLM with a binomial (probit)
link. This model included the Ordinal Number of Fixation
on the scene as a predictor; it was entered as a continu-
ous variable ranging from 1 to a maximum of 28 (the 99th
quantile).
In the tables of results, we report the beta coefficients,
t values (LMM), z values (GLMM), and p values for each
model. For LMMs, the level of significance was calculated
from an F test based on the Satterthwaite approximation
to the effective degrees of freedom (Satterthwaite, 1946),
whereas p values in GLMMs are based on asymptotic
Wald tests.
Electrophysiological Data
Linear deconvolution modeling (first level of analysis).
EEG measurements during active vision are associated
with two major methodological problems: overlapping
potentials and low-level signal variability (Dimigen &
Ehinger, 2019). Overlapping potentials arise from the rapid
pace of active information sampling through eye move-
ments, which causes the neural responses that are evoked
by subsequent fixations on the stimulus to overlap with
each other. Because the average fixation duration usually
varies between conditions, this changing overlap can eas-
ily confound the measured waveforms. A related issue is
the mutual overlap between the ERP elicited by the initial
presentation of the stimulus and the FRPs evoked by the
subsequent fixations on it. This second type of overlap is
especially important in experiments like ours, in which
the critical fixations occurred at different latencies after
scene onset in the two experimental conditions.
The problem of signal variability refers to the fact that
low-level visual and oculomotor variables can also influence
the morphology of the predominantly visually evoked
fixation-related neural responses (e.g., Kristensen et al.,
2017; Nikolaev et al., 2016; Dimigen et al., 2011). The most
relevant of these variables, which is known to modulate
the entire FRP waveform, is the amplitude of the saccade
Coco, Nuthmann, and Dimigen
577
D
o
w
n
l
o
a
d
e
d
l
l
/
/
/
/
j
f
/
t
t
i
t
.
:
/
/
f
r
o
m
D
o
h
w
t
n
t
p
o
:
a
/
d
/
e
d
m
i
f
r
t
o
p
m
r
c
h
.
p
s
i
l
d
v
i
r
e
e
r
c
t
c
.
m
h
a
i
e
r
d
.
u
c
o
o
m
c
n
/
j
a
o
r
t
c
i
c
n
e
/
-
a
p
r
d
t
i
3
2
c
l
4
e
5
-
7
p
1
d
f
2
0
/
1
3
3
2
2
/
4
4
7
/
5
o
7
c
1
n
/
_
a
1
_
8
0
6
1
1
5
2
0
7
4
6
p
/
d
j
o
b
c
y
n
g
_
u
a
e
_
s
0
t
1
o
5
n
0
0
4
8
.
p
S
d
e
f
p
e
b
m
y
b
e
g
r
u
2
e
0
s
2
t
3
/
j
t
f
/
.
o
n
0
5
M
a
y
2
0
2
1
that precedes fixation onset (e.g., Dandekar et al., 2012;
Thickbroom, Knezevič, Carroll, & Mastaglia, 1991). One
option for controlling the effect of saccade amplitude is
to include it as a continuous covariate in a massive uni-
variate regression model (Smith & Kutas, 2015a, 2015b),
in which a separate regression model is computed for
each EEG time point and channel ( Weiss, Knakker, &
Vidnyánszky, 2016; Hauk, Davis, Ford, Pulvermüller, &
Marslen-Wilson, 2006). However, this method does not
account for overlapping potentials.
An approach that allows one to simultaneously control
for overlapping potentials and low-level covariates is de-
convolution within the linear model (for tutorial reviews,
see Dimigen & Ehinger, 2019; Smith & Kutas, 2015a,
2015b), sometimes also called “continuous-time regres-
sion” (Smith & Kutas, 2015b). Initially developed to sepa-
rate overlapping BOLD responses (e.g., Serences, 2004),
linear deconvolution has also been applied to separate
overlapping potentials in ERP (Smith & Kutas, 2015b) and
FRP (Cornelissen et al., 2019; Ehinger & Dimigen, 2019;
Kristensen et al., 2017; Dandekar et al., 2012) paradigms.
Another elegant property of this approach is that the
ERPs elicited by scene onset and the FRPs elicited by
fixations on the scene can be disentangled and simulta-
neously estimated in the same regression model. The ben-
efits of deconvolution are illustrated in more detail in
Supplementary Figures S2 and S3.
Here, we applied this technique by using the new
unfold toolbox (Ehinger & Dimigen, 2019), which repre-
sents the first-level analysis and provides us with the par-
tial effects (i.e., the beta coefficients or “regression ERPs”;
Smith & Kutas, 2015a, 2015b) for each predictor of inter-
est. In a first step, both stimulus onset events and fixation
onset events were included as stick functions (also called
“finite impulse responses”) in the design matrix of the re-
gression model. To account for overlapping activity from
adjacent experimental events, the design matrix was then
time-expanded in a time window between −300 and
+800 msec around each stimulus and fixation onset
event. Time expansion means that the time points within
this window are added as predictors to the regression
model. Because the temporal distance between subse-
quent events in the experiment is variable, it is possible
to disentangle their overlapping responses. Time expan-
sion with stick functions is explained in Serences (2004)
and Ehinger and Dimigen (2019; see their Figure 2).
The model was run on EEG data sampled at the original
512 Hz; that is, no down-sampling was performed.
Using Wilkinson notation, the model formula for scene
onset events was defined as
ERP ∼ 1 þ Consistency
In this formula, the beta coefficients for the intercept (1)
capture the shape of the overall waveform of the stimulus
ERP in the consistent condition, which was used as the
reference level, whereas those for Consistency capture
the differential effect of presenting an inconsistent object
in the scene (relative to a consistent object) on the ERP.
The coefficients for the predictor Consistency are there-
fore analogous to a difference waveform in a traditional
ERP analysis (Smith & Kutas, 2015a, 2015b) and would
reveal if semantic processing already occurs immediately
after the initial presentation of the scene.
In the same regression model, we also included the
onsets of all fixations made on the scene. Fixation onsets
were modeled with the formula
FRP ∼ 1 þ Consistency * Type þ Sacc Amplitude
Thus, we predicted the FRP for each time point as a func-
tion of the semantic Consistency of the target object
(consistent vs. inconsistent; consistent as the reference
level) in interaction with the Type of fixation (critical
fixation vs. nontarget fixation; nontarget fixation as the
reference level). In this model, any FRP consistency
effects elicited by the pretarget or target fixation would
appear as an interaction between Consistency and
Fixation Type. In addition, we included the incoming
Saccade Amplitude (in degrees of visual angle) as a con-
tinuous linear covariate to control for the effect of sac-
cade size on the FRP waveform.2 Thus, the full model
was as follows:
fERP ∼1 þ Consistency;
FRP ∼ 1 þ Consistency * Type þ Sacc Amplitudeg
This regression model was then solved for the betas using
the LSMR algorithm in MATLAB (without regularization).
The deconvolution model specified by the formula
above was run twice: In one version, we treated the pre-
target fixation (t − 1) as the critical fixation; in the other
version, we treated the target fixation (t) as the critical
fixation. In a given model, all fixations but the critical
ones were defined as nontarget fixations. FRPs for fixa-
tion t − 1 and for fixation t were estimated in two sepa-
rate runs of the model, rather than simultaneously within
the same model, because the estimation of overlapping
activity was much more stable in this case. In other
words, although the deconvolution method allowed us
to control for much of the overlapping brain activity from
other fixations, we were not able to use the model to di-
rectly separate the (two) N400 consistency effects elicited
by the fixations t − 1 and t.3
Both runs of the model (the one for t − 1 and t) also
yield an estimate for the scene-onset ERP, but because
the results for the scene-onset ERP were virtually identi-
cal, we present the betas from the first run of the model.
The average number of events entering the model per
participant was 65.3 and 73.0 for scene onsets (consistent
and inconsistent conditions, respectively), 883.5 and 912.4
for nontarget fixations (nt), 59.8 and 61.8 for pretarget fix-
ations (t − 1), and 65.3 and 73.0 for target fixations (t).
578
Journal of Cognitive Neuroscience
Volume 32, Number 4
D
o
w
n
l
o
a
d
e
d
l
l
/
/
/
/
j
t
t
f
/
i
t
.
:
/
/
f
r
o
m
D
o
h
w
t
n
t
p
o
:
a
/
d
/
e
d
m
i
f
r
t
o
p
m
r
c
h
.
p
s
i
l
d
v
i
r
e
e
r
c
t
c
.
m
h
a
i
e
r
d
.
u
c
o
o
m
c
n
/
j
a
o
r
t
c
i
c
n
e
/
-
a
p
r
d
t
i
3
2
c
l
4
e
5
-
7
p
1
d
f
2
0
/
1
3
3
2
2
/
4
4
7
/
5
o
7
c
1
n
/
_
a
1
_
8
0
6
1
1
5
2
0
7
4
6
p
/
d
j
o
b
c
y
n
g
_
u
a
e
_
s
0
t
1
o
5
n
0
0
4
8
.
p
S
d
e
f
p
e
b
m
y
b
e
g
r
u
2
e
0
s
2
t
3
/
j
f
.
t
/
o
n
0
5
M
a
y
2
0
2
1
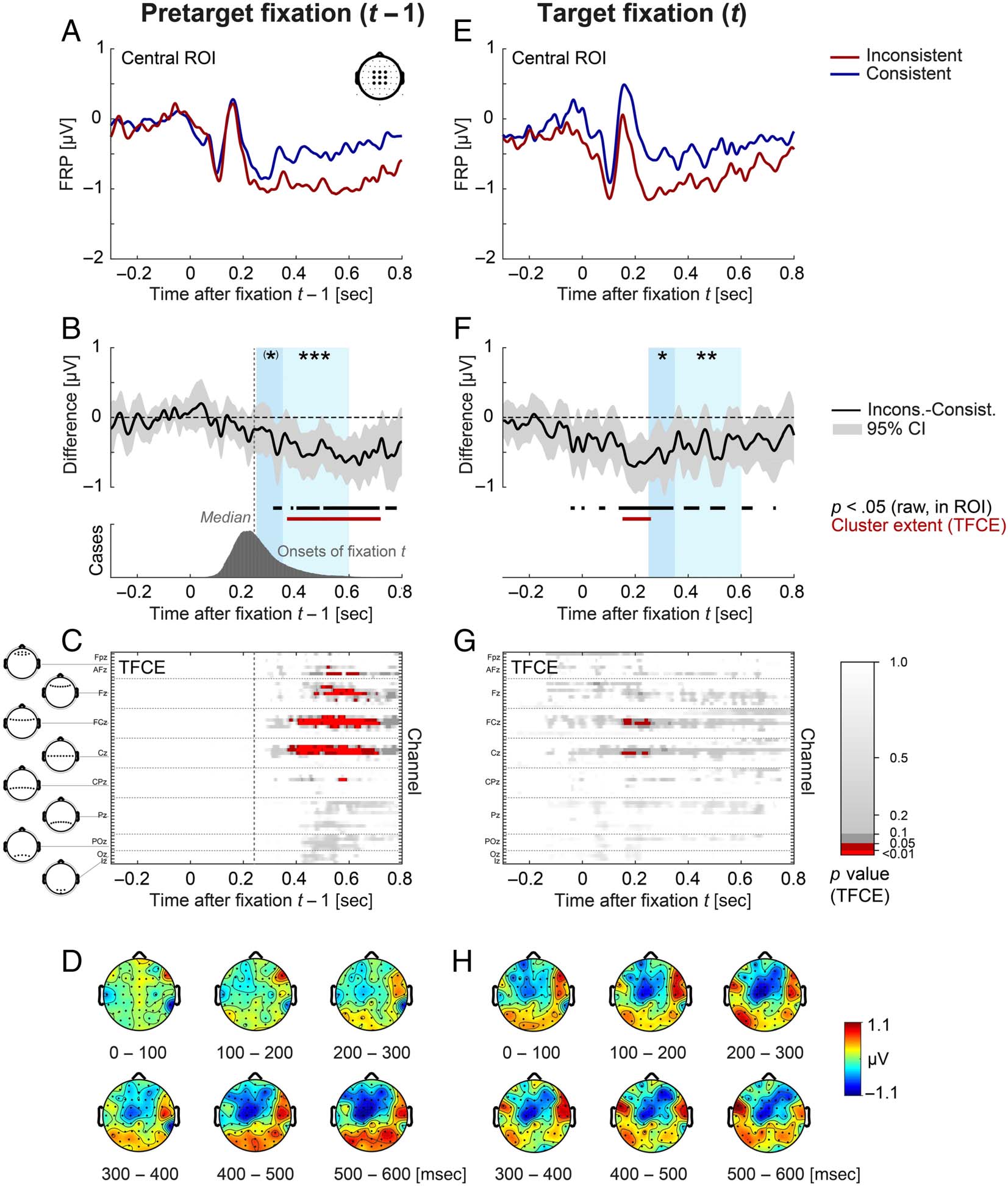
Baseline placement for FRPs. Another challenging issue
for free-viewing EEG experiments is the choice of an ap-
propriate neutral baseline interval for the FRP waveforms
(Nikolaev et al., 2016). Baseline placement is particularly
relevant for experiments on extrafoveal processing where
we do not know in advance when EEG differences will
arise and whether they may already develop before fixation
onset.
For the pretarget fixation t − 1 and nontarget fixations nt,
we used a standard baseline interval by subtracting the mean
channel voltages between −200 and 0 msec before the
event (note that the saccadic spike potential ramping up
at the end of this interval was almost completely removed
by our ICA procedure; see Supplementary Figure S1). For
fixation t, we cannot use such a baseline because semantic
processing may already be ongoing by the time the target
object is fixated. Thus, to apply a neutral baseline to
fixation t, we subtracted the mean channel voltages in
the 200-msec interval before the preceding fixation t − 1
also from the FRP aligned to the target fixations t (see
Nikolaev et al., 2016, for similar procedures). The scene-
onset ERP was corrected with a standard prestimulus
baseline (−200 to 0 msec).
Group statistics for EEG (second level of analysis). To
perform second-level group statistics, averaged EEG
waveforms at the single-participant level (“regression
ERPs”) were reconstructed from the beta coefficients of
the linear deconvolution model. These regression-based
ERPs are directly analogous to participant-level averages
in a traditional ERP analysis (Smith & Kutas, 2015a). We
then used two complementary statistical approaches to
examine consistency effect in the EEG at the group level:
linear mixed models and a cluster-based permutation test.
LMMs were
LMM in a priori defined time windows.
used to provide hypothesis-based testing motivated by exist-
ing literature. Specifically, we adopted the spatio-temporal
definitions by Võ and Wolfe (2013) and compared the con-
sistent and inconsistent conditions in the time windows
from 250 to 350 msec (early effect) and 350 to 600 msec
(late effect) at a midcentral ROI of nine electrodes (compris-
ing FC1, FCz, FC2, C1, Cz, C2, CP1, CPz, and CP2). Because
the outputs provided by the linear deconvolution model
(the first-level analysis) are already aggregated at the level
of participant averages, the only predictor included in these
LMMs was the Consistency of the object. Furthermore, to
minimize the risk of Type I error (Barr, Levy, Scheepers,
& Tily, 2013), we started with a random effects structure
with Participant as random intercept and slope for the
Consistency predictor. This random effects structure was
then evaluated and backwards-reduced using the step func-
tion of the lmerTest package (Kuznetsova, Brockhoff, &
Christensen, 2017) to retain the model that was justified
by the data; that is, it converged, and it was parsimonious
in the number of parameters (Matuschek, Kliegl, Vasishth,
Baayen, & Bates, 2017).
It is still largely unknown to
Cluster permutation tests.
what extent the topography of traditional ERP effects trans-
lates to natural viewing. Therefore, to test for consistency
effects across all channels and time points, we additionally
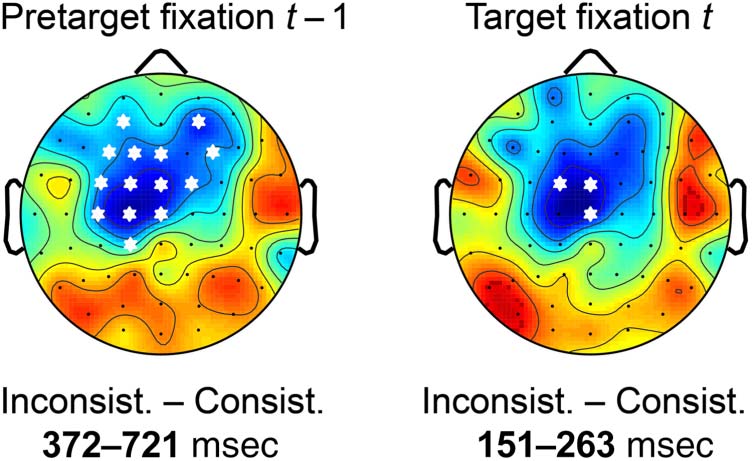
applied the Threshold-Free Cluster Enhancement (TFCE)
procedure developed by Smith and Nichols (2009) and
adapted to EEG data by Mensen and Khatami (2013;
http://github.com/Mensen/ept_TFCE-matlab). In a nutshell,
TFCE is a nonparametric permutation test that controls for
multiple comparisons across time and space, while maintain-
ing relatively high sensitivity (e.g., compared with a
Bonferroni correction). Its advantage over previous cluster
permutation tests (e.g., Maris & Oostenveld, 2007) is that
it does not require the experimenter to set an arbitrary
cluster-forming threshold. In the first stage of the TFCE pro-
cedure, a raw statistical measure (here, t values) is weighted
according to the support provided by clusters of similar
values at surrounding electrodes and time points. In the
second stage, these cluster-enhanced t values are then
compared with the maximum cluster-enhanced values ob-
served under the null hypotheses (based on n = 2000
random permutations of the data). In the present article
(Figures 4 and 5), we not only report the global result of
the test but also plot the spatio-temporal extent of the
first-stage clusters, because they provide some indication
about which time points and electrodes likely contributed
to the overall significant effect established by the test.
D
o
w
n
l
o
a
d
e
d
l
l
/
/
/
/
j
f
/
t
t
i
t
.
:
/
/
f
r
o
m
D
o
h
w
t
n
t
p
o
:
a
/
d
/
e
d
m
i
f
r
t
o
p
m
r
c
h
.
p
s
i
l
d
v
i
r
e
e
r
c
t
c
.
m
h
a
i
e
r
d
.
u
c
o
o
m
c
n
/
j
a
o
r
t
c
i
c
n
e
/
-
a
p
r
d
t
i
3
2
c
l
4
e
5
-
7
p
1
d
f
2
0
/
1
3
3
2
2
/
4
4
7
/
5
o
7
c
1
n
/
_
a
1
_
8
0
6
1
1
5
2
0
7
4
6
p
/
d
j
o
b
c
y
n
g
_
u
a
e
_
s
0
t
1
o
5
n
0
0
4
8
.
p
S
d
e
f
p
e
b
m
y
b
e
g
r
u
2
e
0
s
2
t
3
/
j
/
.
t
f
o
n
0
5
M
a
y
2
0
2
1
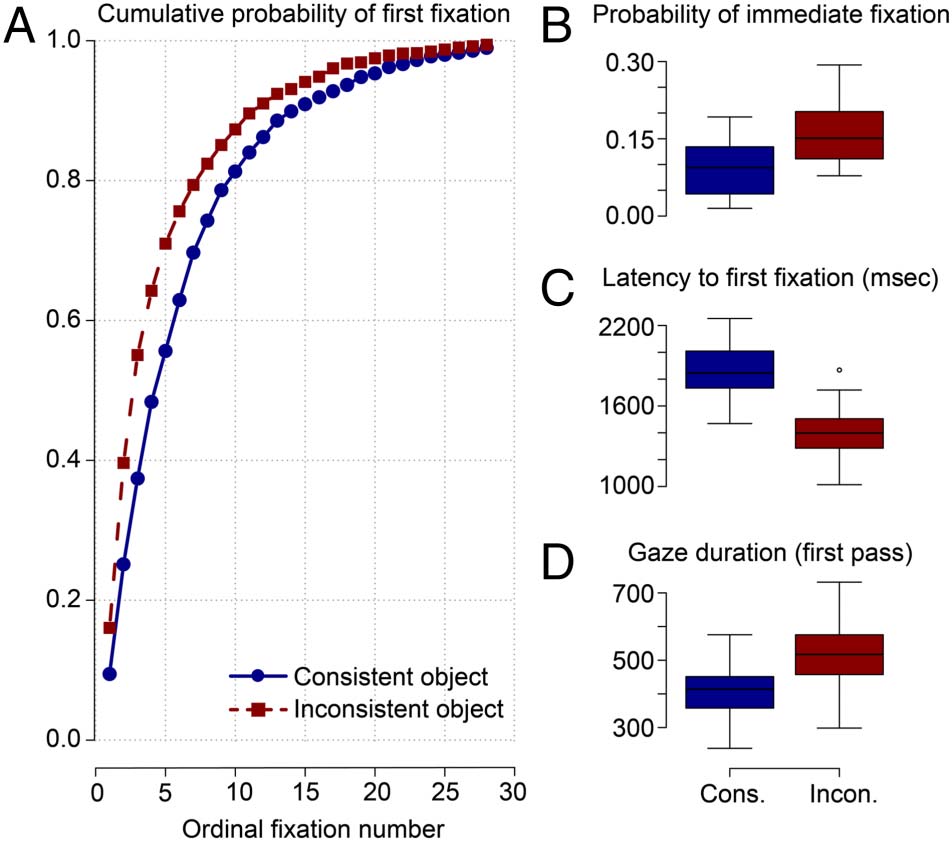
Figure 3. Eye-movement correlates of early overt attention toward
consistent and inconsistent critical objects. (A) Cumulative probability
of fixating the critical object as a function of the ordinal fixation number
on the scene. Blue solid line = consistent object; red dashed line =
inconsistent object. (B) Probability of fixating the critical object
immediately, that is, with the first fixation after scene onset. (C) Latency
until fixating the critical object for the first time. (D) First-pass gaze
duration for the critical object, that is, the sum of all fixation durations
from first entry to first exit. The size of the boxplots (B–D) represent
the 25th and 75th percentiles of the measure (lower and upper
quartiles). Dots indicate observations lying beyond the extremes of the
whiskers. Cons. = consistent; Incon. = inconsistent.
Coco, Nuthmann, and Dimigen
579
Table 2. Cumulative Probability of Having Fixated the Critical Object as a Function of the Ordinal Number of Fixations on the Scene
(Binomial Probit)
Predictor
Intercept
Nr. Fixation
Consistency
Consistency × Nr. Fixation
β
−1.04
−2.02
0.17
−0.72
Cumulative Probability of First Fixation
SE
0.02
0.06
0.03
0.09
z Value
−50.2
−35.5
5.9
−8.1
Pr (>|z|)
.00001
.00001
.00001
.00001
The centered predictors are Consistency (konsistent: −0.5, inconsistent: 0.5) and Number of Fixation (Nr. Fixation).
D
Ö
w
N
l
Ö
A
D
e
D
l
l
/
/
/
/
J
T
T
F
/
ich
T
.
:
/
/
F
R
Ö
M
D
Ö
H
w
T
N
T
P
Ö
:
A
/
D
/
e
D
M
ich
F
R
T
Ö
P
M
R
C
H
.
P
S
ich
l
D
v
ich
R
e
e
R
C
T
C
.
M
H
A
ich
e
R
D
.
u
C
Ö
Ö
M
C
N
/
J
A
Ö
R
T
C
ich
C
N
e
/
–
A
P
R
D
T
ich
3
2
C
l
4
e
5
–
7
P
1
D
F
2
0
/
1
3
3
2
2
/
4
4
7
/
5
Ö
7
C
1
N
/
_
A
1
_
8
0
6
1
1
5
2
0
7
4
6
P
/
D
J
Ö
B
C
j
N
G
_
u
A
e
_
S
0
T
1
Ö
5
N
0
0
4
8
.
P
S
D
e
F
P
e
B
M
j
B
e
G
R
u
2
e
0
S
2
T
3
/
J
.
/
T
F
Ö
N
0
5
M
A
j
2
0
2
1
Please note, Jedoch, that unlike the global test result,
these first-stage values are not stringently controlled for
false positives and do not establish precise effect onsets
or offsets (Sassenhagen & Draschkow, 2019). We report
them here as a descriptive statistic.
Endlich, for purely descriptive purposes and to provide a
priori information for future studies, we also plot the 95%
between-participant confidence interval for the consis-
tency effects at the central ROI (corresponding to sample-
by-sample paired t testing without correction for multiple
comparisons; see also Mudrik et al., 2014) in Figures 4
Und 5.
ERGEBNISSE
Task Performance (Change Detection Task)
After the recognition phase, participants pressed a button
to indicate whether or not a change had taken place within
the scene. Response accuracy in this task was high (M =
85.0 ± 5.16%) and did not differ as a function of whether
the study scene contained a consistent (84.6 ± 5.28%) oder
an inconsistent (85.3 ± 5.12%) target object.
Eye-movement Behavior
Figure 3A shows the cumulative probability of having fixat-
ed the target object as a function of the ordinal number of
fixation and semantic consistency, und Tisch 2 reports the
corresponding GLM coefficients. We found a significant
main effect of Consistency; overall, inconsistent objects
were looked at with a higher probability than consistent
Objekte. Wie erwartet, the cumulative probability of look-
ing at the critical object increased as a function of the
Ordinal Number of Fixation. There was also a significant
interaction between the two variables.
Complementing this global analysis, we analyzed the
very first eye movement during scene exploration to as-
sess whether observers had immediate extrafoveal access
to object–scene semantics (Loftus & Mackworth, 1978).
The mean probability of immediate object fixation was
12.93%; we observed a numeric advantage of inconsistent
objects over consistent objects (Abbildung 3B), but this differ-
ence was not significant (Tisch 3). The latency to first fix-
ation on the target object is another measure to capture
the potency of an object in attracting early attention in ex-
trafoveal vision (z.B., Võ & Henderson, 2009; Underwood
& Foulsham, 2006). This measure is defined as the time
elapsed between the onset of the scene image and the
first fixation on the critical object. Wichtig, this latency
was significantly shorter for inconsistent as compared with
consistent objects (Figure 3C, Tisch 3).
Darüber hinaus, we analyzed gaze duration as a measure of
foveal object processing time (z.B., Henderson et al.,
1999). First-pass gaze duration for a critical object is de-
fined as the sum of all fixation durations from first entry
to first exit. On average, participants looked longer at in-
konsistent (519 ms) than consistent (409 ms) Objekte
Tisch 3. Probability of Immediate Fixation, Latency to First Fixation, and Gaze Duration
Probability of Immediate Fixation
Latency to First Fixation
Gaze Duration
Predictor
Intercept
Consistency
β
−2.82
0.22
SE
0.18
0.16
z
−15.36***
1.38
β
1,774.4
−246.4
SE
77.2
64.0
T
β
SE
T
23.0***
455.5
36.55
23.33***
−3.85***
105.0
14.83
7.08***
The simple coded predictor is Consistency (consistent = −0.5, inconsistent = 0.5). We report the β, standard error, z value (for binomial link),
and t value. Asterisks indicate significant predictors.
***P < .001.
580
Journal of Cognitive Neuroscience
Volume 32, Number 4
D
o
w
n
l
o
a
d
e
d
l
l
/
/
/
/
j
t
t
f
/
i
t
.
:
/
/
f
r
o
m
D
o
h
w
t
n
t
p
o
:
a
/
d
/
e
d
m
i
f
r
t
o
p
m
r
c
h
.
p
s
i
l
d
v
i
r
e
e
r
c
t
c
.
m
h
a
i
e
r
d
.
u
c
o
o
m
c
n
/
j
a
o
r
t
c
i
c
n
e
/
-
a
p
r
d
t
i
3
2
c
l
4
e
5
-
7
p
1
d
f
2
0
/
1
3
3
2
2
/
4
4
7
/
5
o
7
c
1
n
/
_
a
1
_
8
0
6
1
1
5
2
0
7
4
6
p
/
d
j
o
b
c
y
n
g
_
u
a
e
_
s
0
t
1
o
5
n
0
0
4
8
.
p
S
d
e
f
p
e
b
m
y
b
e
g
r
u
2
e
0
s
2
t
3
/
j
/
t
.
f
o
n
0
5
M
a
y
2
0
2
1
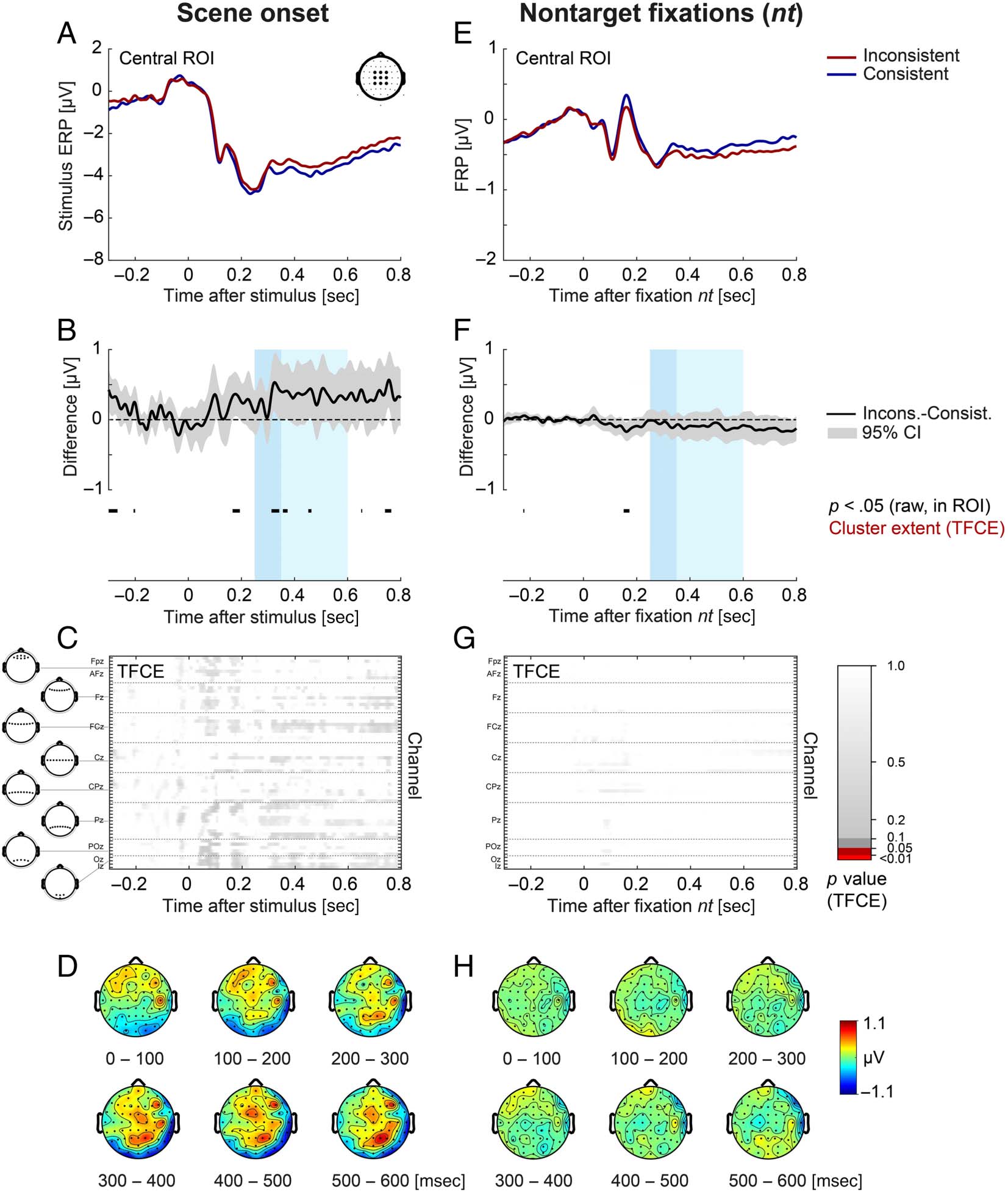
Figure 4. Stimulus ERP aligned to scene onset (left) and FRP aligned to nontarget fixations (right) as a function of object–scene consistency. (A, E)
Grand-averaged ERP/FRP at the central ROI (composed of electrodes FC1, FCz, FC2, C1, Cz, C2, CP1, CPz, and CP2). Red lines represent the
inconsistent condition, and blue lines represent the consistent condition. (B, F) Corresponding difference waves (inconsistent minus consistent) at
the central ROI. Gray shading illustrates the 95% confidence interval (without correction for multiple comparisons) of the difference wave, with
values outside the confidence interval also marked in black below the curve. The two windows used for LMM statistics (250–350 and 350–600 msec)
are indicated in light blue. (C, G) Extent of the spatio-temporal clusters underlying the cluster-based permutation statistic (TFCE) computed across all
electrodes/time points. There were no significant ( p < .05) effects. (D, H) Scalp topographies of the consistency effect (inconsistent minus
consistent) averaged across successive 100-msec time windows. Object–scene consistency had no significant effects on the stimulus ERP or on the
FRP elicited by nontarget fixations, neither in the LMM statistic nor in the cluster permutation test. Consist. = consistent; Incons. = inconsistent.
before leaving the target object for the first time, and this
difference was significant (Table 3). Table 1 summarizes
additional oculomotor characteristics in the two condi-
tions of object consistency.
Supplementary Figures S4 and S5 visualize the locations
of the pretarget, target, and posttarget fixations for two
example scene stimuli.
Electrophysiological Results
Figures 4 and 5 depict the ERP evoked by the presenta-
tion of the scene as well as the FRPs for the three types of
fixation that were analyzed. Results focus on the midcen-
tral ROI for which effects of object–scene consistency
have been reported. Waveforms for other scalp sites
are depicted in Supplementary Figures S6–S9.
Coco, Nuthmann, and Dimigen
581
D
o
w
n
l
o
a
d
e
d
l
l
/
/
/
/
j
t
t
f
/
i
t
.
:
/
/
f
r
o
m
D
o
h
w
t
n
t
p
o
:
a
/
d
/
e
d
m
i
f
r
t
o
p
m
r
c
h
.
p
s
i
l
d
v
i
r
e
e
r
c
t
c
.
m
h
a
i
e
r
d
.
u
c
o
o
m
c
n
/
j
a
o
r
t
c
i
c
n
e
/
-
a
p
r
d
t
i
3
2
c
l
4
e
5
-
7
p
1
d
f
2
0
/
1
3
3
2
2
/
4
4
7
/
5
o
7
c
1
n
/
_
a
1
_
8
0
6
1
1
5
2
0
7
4
6
p
/
d
j
o
b
c
y
n
g
_
u
a
e
_
s
0
t
1
o
5
n
0
0
4
8
.
p
S
d
e
f
p
e
b
m
y
b
e
g
r
u
2
e
0
s
2
t
3
/
j
f
t
.
/
o
n
0
5
M
a
y
2
0
2
1
Figure 5. Grand-averaged FRP elicited by pretarget fixation (left) and target fixation (right) as a function of object–scene consistency. (A, E) Grand-
averaged FRPs at the central ROI. (B, F) Difference waves at the central ROI. In B, the gray distribution shows the onset of fixation t relative to the onset of
the pretarget fixation t − 1, with the vertical dotted line indicating the median latency (260 msec). (C, G) Results of cluster-based permutation testing
(TFCE). The extent of the clusters from the first stage of the permutation test (marked in red) provides some indication which spatio-temporal features
of the waveforms likely contributed to the overall significant effect of consistency. The temporal extent of the clusters is also illustrated by the red bars in B
and F. (D, H) Scalp topographies of the consistency effect (inconsistent minus consistent) across successive 100-msec time windows. A frontocentral
N400 effect emerged in the FRP time-locked to fixation t − 1 and reached significance shortly after the eyes had moved on to fixation t. This effect then
continued during fixation t reaching a maximum of 200 msec after the start of the target fixation. Consist. = consistent; Incons. = inconsistent.
Scene-onset ERP
The left panels of Figure 4 show the grand-averaged ERP
aligned to scene onset. Although inspection of the scalp
maps indicated slightly more positive amplitudes over
central right-hemispheric electrodes in the inconsistent
condition, these differences were not statistically signifi-
cant. Specifically, no effect of Consistency was found with
the LMM analysis in the early or late time window (see
Table 4 for detailed LMM results). Similarly, the TFCE test
across all channels and time points yielded no significant
Consistency effect (all ps > .2; see Figure 4C). Daher, Wir
found no evidence that the semantic consistency of the
target object influences the neural response to the initial
presentation of the scene.
582
Zeitschrift für kognitive Neurowissenschaften
Volumen 32, Nummer 4
Tisch 4. Mixed-Effects Models for the ERPs/FRPs at the Mid-central ROI for Two Temporal Windows of Interest as Predicted by
Consistency
Type of Event
Scene onset
nt
t − 1
T
T (control analysis with baseline before fixation t)
Analysis Window
Early (250–350 msec)
Late (350–600 msec)
Early (250–350 msec)
Late (350–600 msec)
Early (250–350 msec)
Late (350–600 msec)
Early (250–350 msec)
Late (350–600 msec)
Early (250–350 msec)
Late (350–600 msec)
β
0.28
0.34
−0.06
−0.09
−0.28
−0.46
−0.52
−0.38
−0.34
−0.20
SE
0.39
0.39
0.07
0.08
0.15
0.12
0.17
0.15
0.16
0.17
t Value
0.71
0.37
−0.79
−1.10
−1.77(*)
−3.76***
−3.03**
−2.43*
−2.20*
−1.14
Temporal windows of interest: Early = 250–350 msec; Late = 350–600 msec. Consistency is defined as: Consistent = −0.5, Inconsistent = 0.5.
(*)P < .1. *p < .05. **p < .01. ***p < .001. Nontarget Fixations, nt Next, we tested whether fixations on scenes with an inconsistent object evoke a globally different neural re- sponse than those on scenes containing a consistent ob- ject. As the right panels of Figure 4 show, this was not the case: Consistency had no effect on the FRP for nontarget (nt) fixations, neither in the LMM analysis (see Table 4) nor in the TFCE statistic (all ps > .2; see Figure 4G).
Pretarget Fixation, t − 1
Figur 5 depicts the FRPs aligned to the pretarget and tar-
get fixations. Wichtig, in the FRP aligned to the pre-
target fixation t − 1, waveforms began to clearly diverge
between the two consistency conditions, developing into
a long-lasting frontocentral negativity in the inconsistent as
compared with the consistent condition (Figure 5A and B;
see also Supplementary Figure S8). The scalp distribution
of this difference, shown in Figure 6, closely resembled the
frontocentral N400 (and N300) previously reported in ERP
studies on object–scene consistency (z.B., Mudrik et al.,
2014; Võ & Wolfe, 2013). In the LMM analyses conducted
on the midcentral ROI, this effect was marginally signifi-
kippen ( P < .1) for the early time window (250–350 msec)
but became highly significant between 350 and 600 msec
( p < .001; Table 4). The TFCE test across all channels and
time points also revealed a significant effect of consistency
on the pretarget FRP ( p < .05). Figure 5C also shows the
extents of the underlying spatio-temporal clusters, com-
puted in the first stage of the TFCE procedure. Between
372 and 721 msec after fixation onset, we observed a cluster
of 14 frontocentral electrodes that was shifted slightly to
the left hemisphere. This N400 modulation on the pretar-
get fixation could be seen even in traditionally averaged
FRP waveforms without any control of overlapping poten-
tials (see Supplementary Figure S3). In summary, we were
able to measure a significant frontocentral N400 modula-
tion during natural scene viewing that already emerged in
FRPs aligned to the pretarget fixation.
On average, the target fixation t occurred at a median
latency of 240 msec (± 18 msec) after fixation t − 1, as
marked by the vertical dashed line in Figure 5B. If we take
the extent of the cluster from the TFCE test as a rough ap-
proximation for the likely onset of the effect in the FRP, this
means that, on average, at the time when the electrophys-
iological consistency effect started (372 msec), the eyes
had been looking at the target object for only 132 msec
(372 minus 240 msec).
Target Fixation, t
An anterior N400 effect was also clearly visible in the FRP
aligned to fixation t. In the LMM analysis at the central
ROI, the effect was significant in both the early (250–
350 msec, p < .01) and late (350–600 msec, p < .05) win-
dows (see Table 4). However, compared with the effect
aligned to the pretarget fixation, this N400 was significant
at only a few electrodes in the TFCE statistic (Cz, FCz,
and FC1; see Figure 6). Aligned to the target fixation t,
the N400 also peaked extremely early, with the maximum
of the difference curve already observed at 200 msec after
fixation onset (Figure 5F). Qualitatively, a frontocentral
negativity was already visible much earlier than that, within
Coco, Nuthmann, and Dimigen
583
D
o
w
n
l
o
a
d
e
d
l
l
/
/
/
/
j
t
t
f
/
i
t
.
:
/
/
f
r
o
m
D
o
h
w
t
n
t
p
o
:
a
/
d
/
e
d
m
i
f
r
t
o
p
m
r
c
h
.
p
s
i
l
d
v
i
r
e
e
r
c
t
c
.
m
h
a
i
e
r
d
.
u
c
o
o
m
c
n
/
j
a
o
r
t
c
i
c
n
e
/
-
a
p
r
d
t
i
3
2
c
l
4
e
5
-
7
p
1
d
f
2
0
/
1
3
3
2
2
/
4
4
7
/
5
o
7
c
1
n
/
_
a
1
_
8
0
6
1
1
5
2
0
7
4
6
p
/
d
j
o
b
c
y
n
g
_
u
a
e
_
s
0
t
1
o
5
n
0
0
4
8
.
p
S
d
e
f
p
e
b
m
y
b
e
g
r
u
2
e
0
s
2
t
3
/
j
/
f
t
.
o
n
0
5
M
a
y
2
0
2
1
underlying object recognition (e.g., Loftus & Mackworth,
1978; Biederman, 1972). At the core of this debate are
the type of object features that are accessed (e.g., low-
level vs. high-level), the time course of their processing
(e.g., preattentive vs. attentive), and the region of the vi-
sual field in which these features can be acquired (e.g.,
foveal vs. extrafoveal). A particularly controversial topic is
whether and how quickly semantic properties of objects
are available outside foveal vision.
In the current study, we approached these questions
from a new perspective by coregistering eye movements
and EEG while participants freely inspected images of
real-world scenes in which a critical object was either
consistent or inconsistent with the scene context. As a
novel finding, we demonstrate a fixation-related N400 ef-
fect during natural scene viewing. Moreover, behavioral
and electrophysiological measures converge to suggest
that the extraction of object–scene semantics can already
begin in extrafoveal vision, before the critical object is
fixated.
It is a rather undisputed finding that inconsistent ob-
jects, such as a flashlight in a bathroom, require increased
processing when selected as targets of overt attention.
Accordingly, several eye-movement studies have re-
ported longer gaze durations on inconsistent than consis-
tent objects, probably reflecting the greater effort required
to resolve the conflict between object meaning and scene
context (e.g., Cornelissen & Võ, 2017; Henderson et al.,
1999; De Graef et al., 1990). In addition, a number of
traditional ERP studies using steady-fixation paradigms
have found that inconsistent objects elicit a larger negative
brain response at frontocentral channels (an N300/N400
complex) as compared with consistent objects (e.g., Coco
et al., 2017; Mudrik et al., 2010; Ganis & Kutas, 2003).
However, previous research with eye movements re-
mained inconclusive on whether semantic processing can
take place before foveal inspection of the object. Evi-
dence in favor of extrafoveal processing of object–scene se-
mantics comes from studies in which inconsistent objects
were selected for fixation earlier than consistent ones
(e.g., Borges et al., 2019; LaPointe & Milliken, 2016;
Underwood et al., 2008). However, other studies have
not found evidence for earlier selection of inconsistent ob-
jects (e.g., Võ & Henderson, 2009, 2011; Henderson et al.,
1999; De Graef et al., 1990). Parafoveal and peripheral vi-
sion are known to be crucial for saccadic programming
(e.g., Nuthmann, 2014). Therefore, any demonstration that
semantic information can act as a source of guidance for
fixation selection in scenes implies that some semantic pro-
cessing must have occurred prior to foveal fixation, that is,
in extrafoveal vision.
ERPs are highly sensitive to semantic processing (Kutas
& Federmeier, 2011) and provide an excellent temporal
resolution to investigate the time course of object process-
ing. However, an obvious limitation of existing ERP studies
is that observers were not allowed to explore the scene
with saccadic eye movements, thereby constraining their
Figure 6. Scalp distribution of frontocentral N400 effects in the time
windows significant in the TFCE statistic (see also Figure 5). White asterisks
highlight the spatial extent of the clusters observed in the first stage of
the TFCE permutation test for both intervals. In the FRP aligned to the
pretarget fixation (left), clusters extended from 372 to 721 msec and across
14 frontocentral channels. In the FRP aligned to the target fixation
(right), clusters extended from 151 to 263 msec at three frontocentral
channels. Consist. = consistent; Inconsist. = inconsistent.
the first 100 msec after fixation onset (Figure 5H). The
TFCE permutation test confirmed an overall effect of con-
sistency ( p < .05) on the target-locked FRP. Figure 5G also
shows the extents of the underlying first-stage clusters.
For the target fixation, clusters only extended across a
brief interval between 151 and 263 msec after fixation on-
set, an interval during which the N400 effect also reached
its peak.
Figure 5F shows that, numerically, voltages at the cen-
tral ROI were more negative in the inconsistent condition
during the baseline interval already, that is, before the
critical object was fixated. To understand the role of ac-
tivity already present before fixation onset, we repeated
the FRP analyses for fixation t after applying a standard
baseline correction, with the baseline placed immedi-
ately before the target fixation itself (−200 to 0 msec).
This way, we eliminate any weak N400-like effects that
may have already been ongoing before target fixation
onset. Interestingly, in the resulting FRP waveforms,
the target-locked N400 effects were weakened: The
N400 effect now failed to reach significance in the
TFCE statistic and in the LMM analysis for the second
window (350–600 msec; see the last row of Table 4)
and only remained significant for the early window
(250–350 msec). This indicates that some N400-like neg-
ativity was already ongoing before target fixation onset.
To summarize, we found no immediate influences of
object–scene consistency in ERPs time-locked to scene
onset. However, N400 consistency effects were found
in FRPs aligned to the target fixation (t) and in those
aligned to the pretarget fixation (t − 1).
DISCUSSION
Substantial research in vision science has been devoted
to understanding the behavioral and neural mechanisms
584
Journal of Cognitive Neuroscience
Volume 32, Number 4
D
o
w
n
l
o
a
d
e
d
l
l
/
/
/
/
j
f
/
t
t
i
t
.
:
/
/
f
r
o
m
D
o
h
w
t
n
t
p
o
:
a
/
d
/
e
d
m
i
f
r
t
o
p
m
r
c
h
.
p
s
i
l
d
v
i
r
e
e
r
c
t
c
.
m
h
a
i
e
r
d
.
u
c
o
o
m
c
n
/
j
a
o
r
t
c
i
c
n
e
/
-
a
p
r
d
t
i
3
2
c
l
4
e
5
-
7
p
1
d
f
2
0
/
1
3
3
2
2
/
4
4
7
/
5
o
7
c
1
n
/
_
a
1
_
8
0
6
1
1
5
2
0
7
4
6
p
/
d
j
o
b
c
y
n
g
_
u
a
e
_
s
0
t
1
o
5
n
0
0
4
8
.
p
S
d
e
f
p
e
b
m
y
b
e
g
r
u
2
e
0
s
2
t
3
/
j
.
f
/
t
o
n
0
5
M
a
y
2
0
2
1
normal attentional dynamics. Instead, the critical object
was usually large and/or placed near the point of fixation.
Hence, these studies were unable to establish whether
semantic processing can take place before foveal inspec-
tion of the critical object.
In the current study, we addressed this problem by si-
multaneously recording behavioral and brain-electric cor-
relates of object processing. Specifically, we analyzed
different eye-movement responses that tap into extra-
foveal and foveal processing along with FRPs time-locked
to the first fixation on the critical object (t) and the fixa-
tion preceding it (t − 1). We also analyzed the scene-
onset ERP evoked by the trial-initial presentation of the
image. Recent advances in linear deconvolution methods
for EEG (e.g., Ehinger & Dimigen, 2019) allowed us to
disentangle the overlapping brain potentials produced
by the scene onset and the subsequent fixations and to
control for the modulating influence of saccade ampli-
tude on the FRP.
The eye-movement behavior showed no evidence for
Hypothesis A, as outlined in the Introduction, according
to which semantic information can exert an immediate
effect on eye-movement control (Loftus & Mackworth,
1978). Specifically, the mean probability of immediate
object fixation was fairly low (12.9%) and not modulated
by Consistency. Instead, the data lend support to
Hypothesis B, according to which extrafoveal processing
of object–scene semantics is possible but takes some
time to unfold. In particular, the results for the latency
to first fixation of the critical object show that inconsis-
tent objects were, on average, looked at sooner than con-
sistent objects (cf. Bonitz & Gordon, 2008; Underwood
et al., 2008). At the same time, we observed longer gaze
durations on inconsistent objects, replicating previous
findings (e.g., Võ & Henderson, 2009; Henderson et al.,
1999; De Graef et al., 1990). Thus, we found not only
behavioral evidence for the extrafoveal processing of
object–scene (in)consistencies but also differences in
the subsequent foveal processing.
The question then remains why existing eye-movement
studies have provided very different results, ranging from
rapid processing of semantic information in peripheral vi-
sion to a complete lack of evidence for extrafoveal se-
mantic processing. Researchers have suggested that the
outcome may depend on factors related to the critical ob-
ject or the scene in which it is located. Variables that may
(or may not) facilitate the appearance of the incongruency
effect include visual saliency (e.g., Underwood &
Foulsham, 2006; Henderson et al., 1999), image clutter
(Henderson & Ferreira, 2004), and the critical object’s size
and eccentricity (Gareze & Findlay, 2007). Therefore, an
important question for future research is to identify the
specific conditions under which extrafoveal semantic in-
formation can be extracted or when the three outlined hy-
potheses and/or outcomes would prevail.
Returning to the present data, the FRP waveforms
showed a negative shift over frontal and central scalp sites
when participants fixated a scene-inconsistent object. This
result is in agreement with traditional ERP studies that
have shown a frontocentral N300/N400 complex after pas-
sive foveal stimulation (e.g., Coco et al., 2017; Mudrik
et al., 2014; Võ & Wolfe, 2013; Ganis & Kutas, 2003) and
extends this finding for the first time to a natural viewing
situation with eye movements. Regarding the time course,
the present data suggest that the effect was already initi-
ated during the preceding fixation (t − 1) but then car-
ried on through fixation (t) on the target object.
As a cautionary note, we emphasize that it is not trivial
to unambiguously ascribe typical N400 (and N300) effects
in the EEG to either extrafoveal or foveal processing. The
reason is that these canonical congruency effects only be-
gin 200–250 msec after stimulus onset (Draschkow et al.,
2018; Mudrik et al., 2010). This means that even a purely
extrafoveal effect would be almost impossible to measure
during the pretarget fixation (t − 1) itself, because it
would only emerge at a time when the eyes are already
moving to the target object. That being said, three prop-
erties of the observed FRP consistency effect suggest that
it was already initiated during the pretarget fixation.
First, because of the temporal jitter introduced by var-
iable fixation durations, an effect that only arises in foveal
vision should be the most robust in the FRP averages
aligned to fixation t but latency-jittered and attenuated
in those aligned to fixation t − 1. However, the opposite
was the case: At least qualitatively, a frontocentral N400
effect was seen at more electrodes (Figure 6) and for
longer time intervals (Figure 5) in the FRP aligned to
the pretarget fixation as compared with the actual target
fixation. The second argument for extrafoveal contribu-
tions to the effect is the forward shift in its time course.
Relative to fixation t, the observed N400 occurred almost
instantly: As the effect topographies in Figure 5H show,
the frontocentral negativity for inconsistent objects was
qualitatively visible within the first 100 msec after fixation
onset, and the effect reached its peak after only 200 msec.
Clusters underlying the TFCE test were also restricted to
an early time range between 151 and 263 msec after fixa-
tion onset and therefore to a much earlier interval to what
we would expect from the canonical N300 or N400 effect
elicited by foveal stimulation.
Of course, it is possible that even purely foveal N400
effects may emerge earlier during active scene explora-
tion with eye movements as compared with the latencies
established in traditional ERP research. For example, it is
reasonable to assume that, during natural vision, ob-
servers preprocess some low-level (nonsemantic) fea-
tures of the soon-to-be fixated object in extrafoveal
vision (cf. Nuthmann, 2017). This nonsemantic preview
benefit might then speed up the timeline of foveal pro-
cessing (including the latency of semantic access) once
the object is fixated (cf. Dimigen, Kliegl, & Sommer,
2012, for reading). Moreover, if eye movements are per-
mitted, observers have more time to build a representa-
tion of the scene before they foveate the target, and this
Coco, Nuthmann, and Dimigen
585
D
o
w
n
l
o
a
d
e
d
l
l
/
/
/
/
j
f
/
t
t
i
t
.
:
/
/
f
r
o
m
D
o
h
w
t
n
t
p
o
:
a
/
d
/
e
d
m
i
f
r
t
o
p
m
r
c
h
.
p
s
i
l
d
v
i
r
e
e
r
c
t
c
.
m
h
a
i
e
r
d
.
u
c
o
o
m
c
n
/
j
a
o
r
t
c
i
c
n
e
/
-
a
p
r
d
t
i
3
2
c
l
4
e
5
-
7
p
1
d
f
2
0
/
1
3
3
2
2
/
4
4
7
/
5
o
7
c
1
n
/
_
a
1
_
8
0
6
1
1
5
2
0
7
4
6
p
/
d
j
o
b
c
y
n
g
_
u
a
e
_
s
0
t
1
o
5
n
0
0
4
8
.
p
S
d
e
f
p
e
b
m
y
b
e
g
r
u
2
e
0
s
2
t
3
/
j
t
.
f
/
o
n
0
5
M
a
y
2
0
2
1
increased contextual constraint may also affect the N400
timing (but see Kutas & Hillyard, 1984). Importantly,
however, neither of these two accounts could explain
why the N400 effect is stronger—rather than much
weaker—in the waveforms aligned to fixation t − 1 as
compared with fixation t. The fact that the eye movement
data also provided clear evidence in favor of extrafoveal
processing further strengthens our interpretation of the
N400 timing.
Finally, we found that the N400 consistency effect
aligned to the target fixation (t) became weaker (and
nonsignificant in two of the three statistical measures
considered) if the baseline interval for the FRP analysis
was placed directly before this target fixation. Again, this
indicates that at least a weak frontocentral negativity in
the inconsistent condition was already present during
the baseline period before the target was fixated.
Together, these results are difficult to reconcile with a
pure foveal processing account and are more consistent
with the notion that semantic processing of the object
was at least initiated in extrafoveal vision (and then con-
tinued after it was foveated).
Crucially, we did not find any effect of target consis-
tency in the traditional ERP aligned to scene onset. In
line with the behavioral results, this goes against the
most extreme Hypothesis A postulating that object se-
mantics can be extracted from peripheral vision already at
the first glance of a scene (Loftus & Mackworth, 1978).
Similarly, there was no effect of consistency on the FRPs
evoked by the nontarget fixations on the scene (Figure 4);
this was also the case in a control analysis that only
included nontarget fixations that occurred earlier than
t − 1 and at an extrafoveal distance between 3° and 7°
from the target object (see Supplementary Figure S10).
All these analyses suggest that the semantic informa-
tion of the critical object started during fixation t − 1.
However, from any given fixation, there are many can-
didate locations that could potentially be chosen for the
next saccade (cf. Tatler, Brockmole, & Carpenter, 2017).
Thus, it is conceivable that observers may have partially
acquired semantic information of the critical object out-
side foveal vision before fixation t − 1, but without select-
ing it as a saccade target. Such reasoning leaves open the
possibility that observers may have already picked up
some information about the target object’s semantics dur-
ing these occasions.
Taken together, our behavior and electrophysiolog-
ical findings are consistent with the claim formulated in
Hypothesis B that objects can be recognized outside the fo-
vea or even in the visual periphery, at least to some degree.
Indirectly, our results also speak to the debate about the
unit of saccade targeting and, by inference, attentional selec-
tion during scene viewing. Finding effects of object–scene
semantics on eye guidance is evidence in favor of object-
and meaning-based, rather than image-based, guidance of
attention in scenes (e.g., Henderson, Hayes, Peacock, &
Rehrig, 2019; Hwang, Wang, & Pomplun, 2011).
In summary, our findings converge to suggest that the
visual system is capable of accessing semantic features of
objects in extrafoveal vision to guide attention toward ob-
jects that do not fit to the scene’s overall meaning. They
also highlight the utility of investigating attentional and
neural mechanisms in parallel to uncover the mecha-
nisms underlying object recognition during the uncon-
strained exploration of naturalistic scenes.
Acknowledgments
This research was supported by the Leverhulme Trust (grant
ECF-014-205) and Fundação para a Ciência e Tecnologia (grant
PTDC/PSI-ESP/30958/2017) to M. I. C., while he was a Research
Fellow at the University of Edinburgh. The authors thank
Benedikt Ehinger for helpful discussions on EEG deconvolution
techniques.
Reprint requests should be sent to Moreno I. Coco, School of
Psychology, The University of East London, Water Lane,
London E16 2RD, United Kingdom, or Olaf Dimigen, Institut
für Psychologie, Humboldt-Universität zu Berlin, Unter den
Linden 6, 10099 Berlin, or via e-mail: moreno.cocoi@gmail.com
or olaf.dimigen@hu-berlin.de.
Notes
1. We did not include random slopes for two reasons: For
Participant, the inclusion of a random slope led to a small var-
iance and a perfect correlation between intercept and slope.
For the random effect Scene, only the change trials were fully
counterbalanced in terms of location and consistency, meaning
that the slope for Consistency could not be estimated for the
no-change trials.
2. Other low-level variables, such as local image features in
the currently foveated image region (e.g., luminance, spatial fre-
quency), are also known to modulate the FRP waveform. In the
model presented here, we did not include these other covari-
ates because (1) their influence on the FRP waveform is small
compared with that of saccade amplitude and (2) the properties
of the target object (such as its visual saliency) did not differ
between the two levels of object consistency (see Materials
and Rating section). For reasons of simplicity, saccade ampli-
tude was included as a linear predictor in the current model,
although its influence on the FRP becomes nonlinear for large
saccades (e.g., Dandekar et al., 2012). However, virtually iden-
tical results were obtained when we included it as a nonlinear
(spline) predictor instead (Dimigen & Ehinger, 2019).
3.
In theory, a more elegant model would include Type as a
three-level predictor, with the levels of pretarget, target, and
nontarget fixation. In principle, this would allow us to dissociate
which parts of the N400 consistency effects are elicited by fixa-
tion t − 1 versus fixation t. The practical disadvantage of this
approach is that the overlapping activities from both t − 1
and t would then be estimated on comparatively fewer observa-
tions (compared with the extremely stable estimate for the nu-
merous nontarget fixations). This is critical because, compared
with the limited amount of jitter in natural fixation durations,
N400 effects are a long-lasting response, which makes the de-
convolution more challenging. Specifically, we found that, with
the three-level model, model outputs became extremely noisy
and did not yield significant consistency effects for any EEG
time-locking point. By defining either fixation t − 1 or fixation t
as the critical fixation in two separate runs of the model and by
treating all other fixations as nontarget fixations, the estimation be-
comes very robust. This simpler model still removes most of
586
Journal of Cognitive Neuroscience
Volume 32, Number 4
D
o
w
n
l
o
a
d
e
d
l
l
/
/
/
/
j
f
/
t
t
i
t
.
:
/
/
f
r
o
m
D
o
h
w
t
n
t
p
o
:
a
/
d
/
e
d
m
i
f
r
t
o
p
m
r
c
h
.
p
s
i
l
d
v
i
r
e
e
r
c
t
c
.
m
h
a
i
e
r
d
.
u
c
o
o
m
c
n
/
j
a
o
r
t
c
i
c
n
e
/
-
a
p
r
d
t
i
3
2
c
l
4
e
5
-
7
p
1
d
f
2
0
/
1
3
3
2
2
/
4
4
7
/
5
o
7
c
1
n
/
_
a
1
_
8
0
6
1
1
5
2
0
7
4
6
p
/
d
j
o
b
c
y
n
g
_
u
a
e
_
s
0
t
1
o
5
n
0
0
4
8
.
p
S
d
e
f
p
e
b
m
y
b
e
g
r
u
2
e
0
s
2
t
3
/
j
/
.
f
t
o
n
0
5
M
a
y
2
0
2
1
the overlapping activity from other fixations. However, the
consistency-specific activity evoked by fixation t − 1 (i.e., the
N400 effect) will not be removed from the FRP aligned to
the fixation t and vice versa.
REFERENCES
Andrews, S., & Veldre, A. (2019). What is the most plausible
account of the role of parafoveal processing in reading?
Language and Linguistics Compass, 13, e12344.
Antes, J. R. (1974). The time course of picture viewing. Journal
of Experimental Psychology, 103, 62–70.
Barr, D. J., Levy, R., Scheepers, C., & Tily, H. J. (2013).
Random effects structure for confirmatory hypothesis testing:
Keep it maximal. Journal of Memory and Language, 68,
255–278.
Bates, D., Mächler, M., Bolker, B. M., & Walker, S. C. (2015).
Fitting linear mixed-effects models using lme4. Journal of
Statistical Software, 67, 1–48.
Belke, E., Humphreys, G. W., Watson, D. G., Meyer, A. S., &
Telling, A. L. (2008). Top–down effects of semantic knowledge
in visual search are modulated by cognitive but not perceptual
load. Perception & Psychophysics, 70, 1444–1458.
Biederman, I. (1972). Perceiving real-world scenes. Science,
177, 77–80.
Bonitz, V. S., & Gordon, R. D. (2008). Attention to smoking-
related and incongruous objects during scene viewing. Acta
Psychologica, 129, 255–263.
Borges, M. T., Fernandes, E. G., & Coco, M. I. (2019). Age-
related differences during visual search: The role of
contextual expectations and cognitive control mechanisms.
Aging, Neuropsychology, and Cognition. https://doi.org/
10.1080/13825585.2019.1632256.
Brouwer, A.-M., Reuderink, B., Vincent, J., van Gerven, M. A. J.,
& van Erp, J. B. F. (2013). Distinguishing between
target and nontarget fixations in a visual search task
using fixation-related potentials. Journal of Vision, 13, 17.
Cimminella, F., Della Sala, S., & Coco, M. I. (in press). Parallel
and extra-foveal processing of object semantics during
visual search. Attention, Perception, & Psychophysics.
https://doi.org/10.3758/s13414-019-01906-1.
Coco, M. I., Araujo, S., & Petersson, K. M. (2017). Disentangling
stimulus plausibility and contextual congruency: Electro-
physiological evidence for differential cognitive dynamics.
Neuropsychologia, 96, 150–163.
Cornelissen, T. H. W., Sassenhagen, J., & Võ, M. L.-H. (2019).
Improving free-viewing fixation-related EEG potentials with
continuous-time regression. Journal of Neuroscience
Methods, 313, 77–94.
Cornelissen, T. H. W., & Võ, M. L.-H. (2017). Stuck on semantics:
Processing of irrelevant object–scene inconsistencies
modulates ongoing gaze behavior. Attention, Perception, &
Psychophysics, 79, 154–168.
Dandekar, S., Privitera, C., Carney, T., & Klein, S. A. (2012).
Neural saccadic response estimation during natural viewing.
Journal of Neurophysiology, 107, 1776–1790.
Davenport, J. L., & Potter, M. C. (2004). Scene consistency in
object and background perception. Psychological Science,
15, 559–564.
Debener, S., Thorne, J., Schneider, T. R., & Viola, F. C. (2010).
Using ICA for the analysis of multi-channel EEG data. In M.
Ullsperger & S. Debener (Eds.), Simultaneous EEG and
fMRI: Recording, analysis, and application (pp. 121–133).
New York: Oxford University Press.
De Graef, P., Christiaens, D., & d’Ydewalle, G. (1990). Perceptual
effects of scene context on object identification. Psychological
Research, 52, 317–329.
Delorme, A., & Makeig, S. (2004). EEGLAB: An open sorce
toolbox for analysis of single-trial EEG dynamics including
independent component analysis. Journal of Neuroscience
Methods, 134, 9–21.
Devillez, H., Guyader, N., & Guérin-Dugué, A. (2015). An eye
fixation-related potentials analysis of the P300 potential for
fixations onto a target object when exploring natural scenes.
Journal of Vision, 15, 20.
Dimigen, O. (2020). Optimizing the ICA-based removal of ocular
EEG artifacts from free viewing experiments. Neuroimage,
207, 116117.
Dimigen, O., & Ehinger, B. V. (2019). Analyzing combined eye-
tracking/EEG experiments with (non)linear deconvolution
models. BioRxiv. https://doi.org/10.1101/735530.
Dimigen, O., Kliegl, R., & Sommer, W. (2012). Trans-saccadic
parafoveal preview benefits in fluent reading: A study
with fixation-related brain potentials. Neuroimage, 62,
381–393.
Dimigen, O., Sommer, W., Hohlfeld, A., Jacobs, A. M., & Kliegl,
R. (2011). Coregistration of eye movements and EEG in
natural reading: Analyses and review. Journal of
Experimental Psychology: General, 140, 552–572.
Draschkow, D., Heikel, E., Võ, M. L.-H., Fiebach, C. J., &
Sassenhagen, J. (2018). No evidence from MVPA for
different processes underlying the N300 and N400 incongruity
effects in object–scene processing. Neuropsychologia,
120, 9–17.
Dyck, M., & Brodeur, M. B. (2015). ERP evidence for the
influence of scene context on the recognition of ambiguous
and unambiguous objects. Neuropsychologia, 72, 43–51.
Ehinger, B. V., & Dimigen, O. (2019). Unfold: An integrated
toolbox for overlap correction, non-linear modeling,
and regression-based EEG analysis. PeerJ, 7, e7838.
Feldman, J. (2003). What is a visual object? Trends in Cognitive
Sciences, 7, 252–256.
Fenske, M. J., Aminoff, E., Gronau, N., & Bar, M. (2006). Top–
down facilitation of visual object recognition: Object-based
and context-based contributions. Progress in Brain
Research, 155, 3–21.
Ganis, G., & Kutas, M. (2003). An electrophysiological study of
scene effects on object identification. Cognitive Brain
Research, 16, 123–144.
Garcia-Diaz, A., Fdez-Vidal, X. R., Pardo, X. M., & Dosil, R.
(2012). Saliency from hierarchical adaptation through
decorrelation and variance normalization. Image and Vision
Computing, 30, 51–64.
Gareze, L., & Findlay, J. M. (2007). Absence of scene context
effects in object detection and eye gaze capture. In R. P. G.
van Gompel, M. H. Fischer, W. S. Murray, & R. W. Hill
(Eds.), Eye movements: A window on mind and brain
(pp. 617–637). Oxford, UK: Elsevier.
Hauk, O., Davis, M. H., Ford, M., Pulvermüller, F., & Marslen-
Wilson, W. D. (2006). The time course of visual word
recognition as revealed by linear regression analysis of ERP
data. Neuroimage, 30, 1383–1400.
Henderson, J. M., & Ferreira, F. (2004). Scene perception for
psycholinguists. In J. M. Henderson & F. Ferreira (Eds.), The
interface of language, vision, and action: Eye movements
and the visual world (pp. 1–58). New York: Psychology
Press.
Henderson, J. M., Hayes, T. R., Peacock, C. E., & Rehrig, G.
(2019). Meaning and attentional guidance in scenes:
A review of the meaning map approach. Vision, 3, 19.
Henderson, J. M., Weeks, P. A., Jr., & Hollingworth, A. (1999).
The effects of semantic consistency on eye movements
during complex scene viewing. Journal of Experimental
Psychology: Human Perception and Performance, 25,
210–228.
Coco, Nuthmann, and Dimigen
587
D
o
w
n
l
o
a
d
e
d
l
l
/
/
/
/
j
f
/
t
t
i
t
.
:
/
/
f
r
o
m
D
o
h
w
t
n
t
p
o
:
a
/
d
/
e
d
m
i
f
r
t
o
p
m
r
c
h
.
p
s
i
l
d
v
i
r
e
e
r
c
t
c
.
m
h
a
i
e
r
d
.
u
c
o
o
m
c
n
/
j
a
o
r
t
c
i
c
n
e
/
-
a
p
r
d
t
i
3
2
c
l
4
e
5
-
7
p
1
d
f
2
0
/
1
3
3
2
2
/
4
4
7
/
5
o
7
c
1
n
/
_
a
1
_
8
0
6
1
1
5
2
0
7
4
6
p
/
d
j
o
b
c
y
n
g
_
u
a
e
_
s
0
t
1
o
5
n
0
0
4
8
.
p
S
d
e
f
p
e
b
m
y
b
e
g
r
u
2
e
0
s
2
t
3
/
j
.
t
f
/
o
n
0
5
M
a
y
2
0
2
1
Hohenstein, S., & Kliegl, R. (2014). Semantic preview benefit
Mudrik, L., Shalgi, S., Lamy, D., & Deouell, L. Y. (2014).
during reading. Journal of Experimental Psychology:
Learning, Memory, and Cognition, 40, 166–190.
Hwang, A. D., Wang, H.-C., & Pomplun, M. (2011). Semantic
guidance of eye movements in real-world scenes. Vision
Research, 51, 1192–1205.
Itti, L., Koch, C., & Niebur, E. (1998). A model of saliency-based
visual attention for rapid scene analysis. IEEE Transaction
on Pattern Analysis and Machine Intelligence, 20,
1254–1259.
Jung, T.-P., Humphries, C., Lee, T.-W., Makeig, S., McKeown,
M. J., Iragui, V., et al. (1998). Extended ICA removes artifacts
from electroencephalographic recordings. Advances in
Neural Information Processing Systems, 10, 894–900.
Kamienkowski, J. E., Ison, M. J., Quiroga, R. Q., & Sigman, M.
(2012). Fixation-related potentials in visual search: A combined
EEG and eye tracking study. Journal of Vision, 12, 4.
Kaunitz, L. N., Kamienkowski, J. E., Varatharajah, A., Sigman, M.,
Quiroga, R. Q., & Ison, M. J. (2014). Looking for a face in the
crowd: Fixation-related potentials in an eye-movement visual
search task. Neuroimage, 89, 297–305.
Kliegl, R., Dambacher, M., Dimigen, O., Jacobs, A. M., &
Sommer, W. (2012). Eye movements and brain electric
potentials during reading. Psychological Research, 76,
145–158.
Kretzschmar, F., Bornkessel-Schlesewsky, I., & Schlesewsky,
M. (2009). Parafoveal versus foveal N400s dissociate
spreading activation from contextual fit. NeuroReport, 20,
1613–1618.
Kristensen, E., Rivet, B., & Guérin-Dugué, A. (2017). Estimation
of overlapped eye fixation related potentials: The general
linear model, a more flexible framework than the ADJAR
algorithm. Journal of Eye Movement Research, 10, 1–27.
Kutas, M., & Federmeier, K. D. (2011). Thirty years and
counting: Finding meaning in the N400 component of the
event-related brain potential (ERP). Annual Review of
Psychology, 62, 621–647.
Kutas, M., & Hillyard, S. A. (1984). Brain potentials during
reading reflect word expectancy and semantic association.
Nature, 307, 161–163.
Kuznetsova, A., Brockhoff, P. B., & Christensen, R. H. B. (2017).
lmerTest package: Tests in linear mixed effects models.
Journal of Statistical Software, 82, 1–26.
LaPointe, M. R. P., & Milliken, B. (2016). Semantically
incongruent objects attract eye gaze when viewing scenes for
change. Visual Cognition, 24, 63–77.
Loftus, G. R., & Mackworth, N. H. (1978). Cognitive
determinants of fixation location during picture viewing.
Journal of Experimental Psychology: Human Perception
and Performance, 4, 565–572.
Mackworth, N. H., & Morandi, A. J. (1967). The gaze selects
information details within pictures. Perception &
Psychophysics, 2, 547–552.
Maris, E., & Oostenveld, R. (2007). Nonparametric statistical
testing of EEG- and MEG-data. Journal of Neuroscience
Methods, 164, 177–190.
Matuschek, H., Kliegl, R., Vasishth, S., Baayen, H., & Bates, D.
(2017). Balancing Type I error and power in linear mixed
models. Journal of Memory and Language, 94, 305–315.
Mensen, A., & Khatami, R. (2013). Advanced EEG analysis using
threshold-free cluster-enhancement and non-parametric
statistics. Neuroimage, 67, 111–118.
Moores, E., Laiti, L., & Chelazzi, L. (2003). Associative
knowledge controls deployment of visual selective attention.
Nature Neuroscience, 6, 182–189.
Mudrik, L., Lamy, D., & Deouell, L. Y. (2010). ERP evidence for
context congruity effects during simultaneous object–scene
processing. Neuropsychologia, 48, 507–517.
Synchronous contextual irregularities affect early scene
processing: Replication and extension. Neuropsychologia,
56, 447–458.
Niefind, F., & Dimigen, O. (2016). Dissociating parafoveal
preview benefit and parafovea-on-fovea effects during
reading: A combined eye tracking and EEG study.
Psychophysiology, 53, 1784–1798.
Nikolaev, A. R., Meghanathan, R. N., & van Leeuwen, C. (2016).
Combining EEG and eye movement recording in free
viewing: Pitfalls and possibilities. Brain and Cognition, 107,
55–83.
Nuthmann, A. (2013). On the visual span during object search
in real-world scenes. Visual Cognition, 21, 803–837.
Nuthmann, A. (2014). How do the regions of the visual field
contribute to object search in real-world scenes? Evidence
from eye movements. Journal of Experimental Psychology:
Human Perception and Performance, 40, 342–360.
Nuthmann, A. (2017). Fixation durations in scene viewing:
Modeling the effects of local image features, oculomotor
parameters, and task. Psychonomic Bulletin & Review, 24,
370–392.
Nuthmann, A., de Groot, F., Huettig, F., & Olivers, C. N. L.
(2019). Extrafoveal attentional capture by object semantics.
PLoS One, 14, e0217051.
Nuthmann, A., & Einhäuser, W. (2015). A new approach to
modeling the influence of image features on fixation
selection in scenes. Annals of the New York Academy of
Sciences, 1339, 82–96.
Nuthmann, A., & Henderson, J. M. (2010). Object-based attentional
selection in scene viewing. Journal of Vision, 10, 20.
Plöchl, M., Ossandón, J. P., & König, P. (2012). Combining EEG
and eye tracking: Identification, characterization, and correction
of eye movement artifacts in electroencephalographic data.
Frontiers in Human Neuroscience, 6, 278.
Rämä, P., & Baccino, T. (2010). Eye fixation-related potentials
(EFRPs) during object identification. Visual Neuroscience,
27, 187–192.
Rayner, K. (2014). The gaze-contingent moving window in
reading: Development and review. Visual Cognition, 22,
242–258.
Rayner, K., Balota, D. A., & Pollatsek, A. (1986). Against
parafoveal semantic preprocessing during eye fixations in
reading. Canadian Journal of Psychology, 40, 473–483.
Sassenhagen, J., & Draschkow, D. (2019). Cluster-based
permutation tests of MEG/EEG data do not establish
significance of effect latency or location. Psychophysiology,
56, e13335.
Satterthwaite, F. E. (1946). An approximate distribution of
estimates of variance components. Biometrics Bulletin, 2,
110–114.
Serences, J. T. (2004). A comparison of methods for
characterizing the event-related BOLD timeseries in rapid
fMRI. Neuroimage, 21, 1690–1700.
Simola, J., Le Fevre, K., Torniainen, J., & Baccino, T. (2015).
Affective processing in natural scene viewing: Valence and
arousal interactions in eye-fixation-related potentials.
Neuroimage, 106, 21–33.
Smith, N. J., & Kutas, M. (2015a). Regression-based estimation
of ERP waveforms: I. The rERP framework. Psychophysiology,
52, 157–168.
Smith, N. J., & Kutas, M. (2015b). Regression-based estimation
of ERP waveforms: II. Nonlinear effects, overlap correction,
and practical considerations. Psychophysiology, 52, 169–181.
Smith, S. M., & Nichols, T. E. (2009). Threshold-free cluster
enhancement: Addressing problems of smoothing, threshold
dependence and localisation in cluster inference.
Neuroimage, 44, 83–98.
588
Journal of Cognitive Neuroscience
Volume 32, Number 4
D
o
w
n
l
o
a
d
e
d
l
l
/
/
/
/
j
f
/
t
t
i
t
.
:
/
/
f
r
o
m
D
o
h
w
t
n
t
p
o
:
a
/
d
/
e
d
m
i
f
r
t
o
p
m
r
c
h
.
p
s
i
l
d
v
i
r
e
e
r
c
t
c
.
m
h
a
i
e
r
d
.
u
c
o
o
m
c
n
/
j
a
o
r
t
c
i
c
n
e
/
-
a
p
r
d
t
i
3
2
c
l
4
e
5
-
7
p
1
d
f
2
0
/
1
3
3
2
2
/
4
4
7
/
5
o
7
c
1
n
/
_
a
1
_
8
0
6
1
1
5
2
0
7
4
6
p
/
d
j
o
b
c
y
n
g
_
u
a
e
_
s
0
t
1
o
5
n
0
0
4
8
.
p
S
d
e
f
p
e
b
m
y
b
e
g
r
u
2
e
0
s
2
t
3
/
j
.
/
t
f
o
n
0
5
M
a
y
2
0
2
1
Stoll, J., Thrun, M., Nuthmann, A., & Einhäuser, W. (2015).
Overt attention in natural scenes: Objects dominate features.
Vision Research, 107, 36–48.
Tatler, B. W., Brockmole, J. R., & Carpenter, R. H. S. (2017).
LATEST: A model of saccadic decisions in space and time.
Psychological Review, 124, 267–300.
Thickbroom, G. W., Knezevič, W., Carroll, W. M., & Mastaglia,
F. L. (1991). Saccade onset and offset lambda waves: Relation
to pattern movement visually evoked potentials. Brain
Research, 551, 150–156.
Underwood, G., & Foulsham, T. (2006). Visual saliency and
semantic incongruency influence eye movements when
inspecting pictures. Quarterly Journal of Experimental
Psychology, 59, 1931–1949.
Underwood, G., Templeman, E., Lamming, L., & Foulsham, T.
(2008). Is attention necessary for object identification?
Evidence from eye movements during the inspection of
real-world scenes. Consciousness and Cognition, 17,
159–170.
Ušćumlić, M., & Blankertz, B. (2016). Active visual search in
non-stationary scenes: Coping with temporal variability and
uncertainty. Journal of Neural Engineering, 13, 016015.
Võ, M. L.-H., & Henderson, J. M. (2009). Does gravity matter?
Effects of semantic and syntactic inconsistencies on the
allocation of attention during scene perception. Journal of
Vision, 9, 24.
Võ, M. L.-H., & Henderson, J. M. (2011). Object–scene
inconsistencies do not capture gaze: Evidence from the
flash-preview moving-window paradigm. Attention, Perception,
& Psychophysics, 73, 1742–1753.
Võ, M. L.-H., & Wolfe, J. M. (2013). Differential
electrophysiological signatures of semantic and syntactic
scene processing. Psychological Science, 24, 1816–1823.
Weiss, B., Knakker, B., & Vidnyánszky, Z. (2016). Visual
processing during natural reading. Scientific Reports, 6,
26902.
Winkler, I., Debener, S., Müller, K.-R., & Tangermann, M.
(2015). On the influence of high-pass filtering on ICA-based
artifact reduction in EEG-ERP. Paper presented at the 2015
37th Annual International Conference of the IEEE
Engineering in Medicine and Biology Society (EMBC)
(pp. 4101–4105). Milan, Italy: IEEE.
Wolfe, J. M., Alvarez, G. A., Rosenholtz, R., Kuzmova, Y. I., &
Sherman, A. M. (2011). Visual search for arbitrary objects in
real scenes. Attention, Perception, & Psychophysics, 73,
1650–1671.
Wu, C.-C., Wick, F. A., & Pomplun, M. (2014). Guidance of visual
attention by semantic information in real-world scenes.
Frontiers in Psychology, 5, 54.
Yan, M., Richter, E. M., Shu, H., & Kliegl, R. (2009). Readers of
Chinese extract semantic information from parafoveal words.
Psychonomic Bulletin & Review, 16, 561–566.
D
o
w
n
l
o
a
d
e
d
l
l
/
/
/
/
j
f
/
t
t
i
t
.
:
/
/
f
r
o
m
D
o
h
w
t
n
t
p
o
:
a
/
d
/
e
d
m
i
f
r
t
o
p
m
r
c
h
.
p
s
i
l
d
v
i
r
e
e
r
c
t
c
.
m
h
a
i
e
r
d
.
u
c
o
o
m
c
n
/
j
a
o
r
t
c
i
c
n
e
/
-
a
p
r
d
t
i
3
2
c
l
4
e
5
-
7
p
1
d
f
2
0
/
1
3
3
2
2
/
4
4
7
/
5
o
7
c
1
n
/
_
a
1
_
8
0
6
1
1
5
2
0
7
4
6
p
/
d
j
o
b
c
y
n
g
_
u
a
e
_
s
0
t
1
o
5
n
0
0
4
8
.
p
S
d
e
f
p
e
b
m
y
b
e
g
r
u
2
e
0
s
2
t
3
/
j
f
/
.
t
o
n
0
5
M
a
y
2
0
2
1
Coco, Nuthmann, and Dimigen
589